CNS Toxoplasmosis Diagnosis and Treatment
- 1. CNS TOXOPLASMOSIS BY: DIANA TABANSI JAN 13TH 2018
- 2. INTRODUCTION Toxoplasmosis is the leading cause of focal central nervous system (CNS) disease in AIDS It is the most common cause of cerebral abscess in immunocompromised patients when the absolute CD4 count is less than 100 u/L The causative organism is Toxoplasma gondii (T. gondii), an obligate intracellular protozoan parasite
- 3. CNS toxoplasmosis in HIV-infected patients is usually a complication of the late phase of the disease. Typically, lesions are found in the brain and their effects dominate the clinical presentation.
- 4. The decision to treat a patient for CNS toxoplasmosis is usually empiric. Primary therapy is followed by long- term suppressive therapy, which is continued until antiretroviral therapy can raise CD4+ counts above 200 cells/µL.
- 5. EPIDEMIOLOGY Clinical CNS toxoplasmosis occurs in 3-15% of patients with AIDS in the United States. Some clinically silent lesions come to diagnosis only at autopsy. Clinical CNS toxoplasmosis occurs in as many as 50- 75% of patients in some European countries and in Africa. The probability of developing reactivated toxoplasmosis is as high as 30% among AIDS patients with a CD4 count <100 cells/microL who are toxoplasma seropositive and are not receiving effective prophylaxis or antiretroviral therapy.
- 6. The most common site of reactivation is the central nervous system (CNS). Recently in a epidemiologic study in Mexico city with 320 AIDS patients, the main conditions related to HIV/AIDS were brain toxoplasmosis (42%), cerebral cryptococcosis (28%), tuberculous meningitis (8.7%), lymphoma (non-Hodgkin) (3.75%), acute HIV infection (3.4%), and AIDS dementia complex (3%).
- 7. The incidence rate has decreased due to the use of highly active antiretroviral therapy (HAART) and prophylaxis against Pneumocystis jiroveci infections with trimethoprim-sulfamethoxazole, which is also effective against toxoplasmosis
- 10. LIFE CYCLE OF T. GONDII Toxoplasmosis is a disease caused by Toxoplasma gondii, an intracellular obligate protozoal parasite. T. gondii has three forms: and the sporozoite (contained in oocysts) the tachyzoite (the rapidly reproducing form), the bradyzoite (a slower reproducing form contained in tissue cysts)
- 13. PATHOPHYSIOLOGY CNS toxoplasmosis results from infection by the intracellular parasite Toxoplasma gondii. It is almost always due to reactivation of old CNS lesions or to hematogenous spread of a previously acquired infection. Occasionally, it results from primary infection.
- 14. CLINICAL PRESENTATIONS CNS toxoplasmosis begins with constitutional symptoms and headache. Later, confusion and drowsiness, seizures, focal weakness, and language disturbance develop. Without treatment, patients progress to coma in days to weeks. Extracerebral involvement can also be seen including but not limited to pneumonitis, chorioretinitis, etc.
- 15. On physical examination, personality and mental status changes may be observed. Seizures, hemiparesis, hemianopia, aphasia, ataxia, and cranial nerve palsies may be evident. Occasionally, symptoms and signs of a radiculomyelopathy predominate.
- 16. DIFFERENTIAL DIAGNOSIS The differential diagnosis of CNS toxoplasmosis includes the following: CNS Lymphoma in HIV Mycobacterial infection (eg, tuberculous abscess, tuberculoma) Fungal infection (eg, cryptococcosis, candidiasis) Chagas disease Bacterial abscess (eg, Nocardia) Neurosyphilis Cardioembolic Stroke Cytomegalovirus Encephalitis in HIV Progressive Polyradiculopathy in HIV Vacuolar Myelopathy in HIV Progressive Multifocal Leukoencephalopathy
- 17. SEROLOGIC STUDIES Serologic studies in patients with CNS toxoplasmosis may demonstrate rising titers of anti-toxoplasma immunoglobulin G (IgG) antibodies. An immunoglobulin M (IgM) antibody response is seen in cases of newly acquired toxoplasmosis or Toxoplasma encephalitis.
- 18. However, anti-Toxoplasmagondii IgG detection may be unreliable in immunodeficient individuals who fail to produce significant titers of specific antibodies. In one study, 16% of patients with a clinical diagnosis and 22% of patients with a histologic diagnosis of toxoplasmosis had undetectable anti-T gondii IgG levels. Causes of false-negative results also include recent infection and insensitive assays
- 19. DEFINITIVE DIAGNOSIS Definitive diagnosis of CNS toxoplasmosis requires the following : Compatible clinical findings Identification of one or more mass lesions by CT, MRI, or other radiographic testing Detection of T gondii in a clinical sample Detection of T gondii DNA on polymerase chain reaction (PCR) testing of cerebrospinal fluid (CSF) samples may facilitate the diagnosis and follow-up of toxoplasmosis in patients with AIDS. In a study using the B1 gene, rapid PCR showed a sensitivity of 83.3% and specificity of 95.7%.
- 20. CSF findings may also include elevated protein and variable glucose and WBC counts. The presence of Epstein-Barr virus DNA in the CSF favors the diagnosis of lymphoma Lumbar puncture may be contraindicated because of increased intracranial pressure. For many clinicians, therefore, CNS toxoplasmosis is an empiric diagnosis that relies on clinical and radiographic improvement in response to specific anti-T gondii therapy. In patients who fail to respond to specific therapy, brain biopsy can be used to secure a clinical sample for testing.
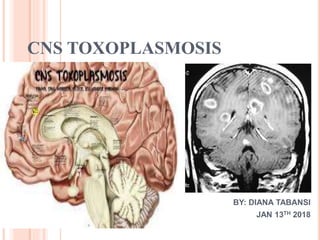
- 21. IMAGING A brain CT scan or MRI with and without contrast is indicated for all patients presenting with altered mental status, headaches, seizures, or focal neurologic signs. MRI clearly is the superior technique but is not available universally. Single or multiple hypodense or hypointense lesions in white matter and basal ganglia with mass effects may be observed on CT or MRI scans. Lesions may enhance in a homogeneous or ring pattern with contrast .
- 27. IMAGING Single lesions favor the diagnosis of lymphoma over that of toxoplasmosis. However, while multiple lesions are more common than single lesions in toxoplasmosis, in one study 27% of patients had a single lesion on CT scan. In the same study, 14% had a single lesion on MRI. MRI is more sensitive than CT scan in detecting multiple lesions.
- 28. IMAGING If the initial imaging study is normal or shows atrophy or focal signal abnormalities (but no mass lesion), diagnostic consideration should be given to meningitides, AIDS dementia complex, or progressive multifocal leukoencephalopathy. If imaging shows one or more focal mass lesions with impending herniation, an open biopsy with decompression is indicated. Treatment for lymphoma, toxoplasmosis, or other opportunistic infections and neoplasms is initiated, depending on biopsy results.
- 29. SCINTIGRAPHY Where available, thallium-201 single-photon emission computed tomography (201 TI SPECT) or 18fluorodeoxyglucose positron emission tomography (18FDG-PET) may be useful in distinguishing between lymphoma and toxoplasmosis. Lymphoma shows an increased uptake as compared with toxoplasmosis. These tests have high specificity but low sensitivity.
- 30. BRAIN BIOPSY Indications for brain biopsy include either of the following: Single mass lesion and negative serologic results No response to 14 days of empiric therapy Diagnostic yield of stereotactic biopsies increases with the number of specimens obtained.
- 31. S
- 32. Histologic findings include the following (also see the images below): Lymphocytic meningitis, individual cyst-containing lesions Astroglial and microglial nodules Associated lymphocytic vasculitis Diffuse encephalitis
- 35. PRIMARY PROPHYLAXIS Toxoplasma-seropositive patients who have CD4 counts <100 cells/μL should receive prophylaxis against TE (toxoplasma Encephalitis ) . The one double-strength-tablet daily dose of trimethoprimsulfamethoxazole (TMP-SMX), which is the preferred regimen for Pneumocystis jirovecii pneumonia (PCP) prophylaxis, is effective against TE and is recommended . TMP-SMX, one double-strength tablet three times weekly, is an alternative . dapsone-pyrimethamine plus leucovorin, which is also effective against PCP .
- 36. SECONDARY PROPHYLAXIS Start after acute treatment of toxoplasmosis . combination of pyrimethamine plus sulfadiazine plus leucovorin is highly effective as suppressive therapy for patients with TE and provides protection against PCP . Pyrimethamine plus clindamycin is commonly used as suppressive therapy for patients with TE who cannot tolerate sulfa drugs not provide protection against PCP . Pyrimethamine 25–50 mg PO daily + sulfadiazine 2000– 4000 mg PO daily (in 2 to 4 divided doses) + leucovorin 10–25 mg PO daily. Alternative Regimen: Clindamycin 600 mg PO q8h + (pyrimethamine 25–50 mg + leucovorin 10–25 mg) PO daily ; must add additional agent to prevent PCP
- 37. PREVENTION To prevent primary toxoplasmosis, patients should avoid eating undercooked meat and should wash their hands carefully after contact with soil or cats. Patients who are seropositive for Toxoplasma should be started on prophylaxis against CNS toxoplasmosis if their CD4+ count drops below 100 cells/μL
- 38. REFERENCES https://www.ncbi.nlm.nih.gov/pubmed/2585580 https://www.medscape.com/invitation/viewTracker.d o?src=house_6 https://www.sciencedirect.com/science/article/pii/S1 930043315300698 http://www.hivmanual.hk/content.html?gonum=34
- 39. S
Editor's Notes
- Of 511 young (1-19 years of age) Jamaicans sampled for IgG antibodies to Toxoplasma by the ELISA method, there was an overall prevalence of 45 per cent. Seropositivity ranged from 24-26 per cent in the 2-year-olds, up to 54 per cent in the 5-9-year-olds. There was no significant difference in prevalence between the sexes, but there was a statistically significant difference between the rates from rural uplands (58 per cent), urban uplands (46 per cent), rural lowlands (40 per cent), and urban lowlands (32 per cent).
- Unsporulated oocysts are shed in the cat’s feces . Although oocysts are usually only shed for 1-2 weeks, large numbers may be shed. Oocysts take 1-5 days to sporulate in the environment and become infective. Intermediate hosts in nature (including birds and rodents) become infected after ingesting soil, water or plant material contaminated with oocysts . • Oocysts transform into tachyzoites shortly after ingestion. These tachyzoites localize in neural and muscle tissue and develop into tissue cyst bradyzoites . • Cats become infected after consuming intermediate hosts harboring tissue cysts . Cats may also become infected directly by ingestion of sporulated oocysts. Animals bred for human consumption and wild game may also become infected with tissue cysts after ingestion of sporulated oocysts in the environment . Eating undercooked meat of animals harboring tissue cysts . • Consuming food or water contaminated with cat feces or by contaminated environmental samples (such as fecal-contaminated soil or changing the litter box of a pet cat) . • Blood transfusion or organ transplantation . • Transplacentally from mother to fetus . • In the human host, the parasites form tissue cysts, most commonly in skeletal muscle, myocardium, brain, and eyes; these cysts may remain throughout the life of the host. Diagnosis is usually achieved by serology, although tissue cysts may be observed in stained biopsy specimens . • Diagnosis of congenital infections can be achieved by detecting T. gondii DNA in amniotic fluid using molecular methods such as PCR
- Constitutional symptoms refers to a group of symptoms that can affect many different systems of the body. Examples include weight loss, fevers, fevers of unknown origin, hyperhidrosis, generalized hyperhidrosis, chronic pain, fatigue, dyspnea, and malaise.
- Like CNS toxoplasmosis, progressive multifocal leukoencephalopathy (PML) can cause mental status changes and focal signs. However, headache and seizures are unusual with PML, and PML lesions themselves cause no mass effect.
- Aerosolized pentamidine does not protect against TE and is not recommended for antitoxoplasma prophylaxis (AI). Prophylaxis against TE should be discontinued in adult and adolescent patients receiving ART whose CD4 counts increase to >200 cells/μL for more than 3 months Preferred Regimen: • TMP-SMX 1 DS PO daily (AII) Alternative Regimens: • TMP-SMX 1 DS PO TIW (BIII), or • TMP-SMX SS PO daily (BIII), or • Dapsone 50 mg PO daily + (pyrimethamine 50 mg + leucovorin 25 mg) PO weekly (BI), or • (Dapsone 200 mg + pyrimethamine 75 mg + leucovorin 25 mg) PO weekly (BI), or • Atovaquone 1500 mg PO daily (CIII), or aidsinfo.nih.gov/guidelines on 5/7/2013 Guidelines for Prevention and Treatment of Opportunistic
- Duration and discontinuation : Patient who remain asymptomatic with regard to signs and symptoms of TE, and have an increase in their CD4 counts to >200 cells/μL after ART that is sustained for more than 6 months (BI) . Secondary prophylaxis (chronic maintenance therapy) for TE should be reintroduced if the CD4 count decreases to <200 cells/μL (AIII). aidsinfo.nih.gov/guidelines on 5/7/2013 Guidelines for Prevention and Treatment of Opportunistic Infections in HIV-Infected Adults For many patients, we use TMP-SMX (TMP 5 mg/kg/day - SMX 25 mg/kg/day) for secondary prophylaxis to help reduce pill burden. This regimen is not recommended in standard guidelines because of the lack of sufficient clinical trial data. www