Intrototitrations 140617143055-phpapp01
•Download as PPT, PDF•
0 likes•51 views
Harris Quantitative Analysis, Quantitative Analysis
Report
Share
Report
Share
Recommended
Recommended
More Related Content
What's hot
What's hot (20)
Total Nitrogen Determination - Traditional and Modern Methods

Total Nitrogen Determination - Traditional and Modern Methods
IA on effectiveness of different types of catalysts MnO2 vs Fe(NO3)3 on the r...

IA on effectiveness of different types of catalysts MnO2 vs Fe(NO3)3 on the r...
A New Method for Determination of Total Nitrogen in Aqueous Samples

A New Method for Determination of Total Nitrogen in Aqueous Samples
IA on effect of temperature of NaOH on the rate of hydrogen production, and f...

IA on effect of temperature of NaOH on the rate of hydrogen production, and f...
Flue gas analisys in industry-Practical guide for Emission and Process Measur...

Flue gas analisys in industry-Practical guide for Emission and Process Measur...
Similar to Intrototitrations 140617143055-phpapp01
Similar to Intrototitrations 140617143055-phpapp01 (20)
Titration methods for quantification of pharmaceutical products

Titration methods for quantification of pharmaceutical products
Synthesizing nickle ammonium chloride chemistry two

Synthesizing nickle ammonium chloride chemistry two
Presentation of pharmaceutical analysis on kjeldhal method of nitrogen estima...

Presentation of pharmaceutical analysis on kjeldhal method of nitrogen estima...
Which titration technique is best fit to detect what type of analyte?

Which titration technique is best fit to detect what type of analyte?
To Determine the Enthalpy of H2O2 Decomposition in Aqueous Solution

To Determine the Enthalpy of H2O2 Decomposition in Aqueous Solution
More from Cleophas Rwemera
More from Cleophas Rwemera (20)
Chapter22 themolecularcellbiologyofdevelopment-140105100412-phpapp02

Chapter22 themolecularcellbiologyofdevelopment-140105100412-phpapp02
Chapter21 cellbirthlineageanddeath-140105095914-phpapp02

Chapter21 cellbirthlineageanddeath-140105095914-phpapp02
Chapter20 regulatingtheeukaryoticcellcycle-140105095738-phpapp01

Chapter20 regulatingtheeukaryoticcellcycle-140105095738-phpapp01
Chapter19 integratingcellsintotissues-140105095535-phpapp02

Chapter19 integratingcellsintotissues-140105095535-phpapp02
Chapter18 cellorganizationandmovementiimicrotubulesandintermediatefilaments-1...

Chapter18 cellorganizationandmovementiimicrotubulesandintermediatefilaments-1...
Chapter17 cellorganizationandmovementimicrofilaments-140105094810-phpapp02

Chapter17 cellorganizationandmovementimicrofilaments-140105094810-phpapp02
Chapter16 cellsignalingiisignalingpathwaysthatcontrolgeneactivity-14010509451...

Chapter16 cellsignalingiisignalingpathwaysthatcontrolgeneactivity-14010509451...
Chapter15 cellsignalingisignaltransductionandshort-termcellularresponses-1401...

Chapter15 cellsignalingisignaltransductionandshort-termcellularresponses-1401...
Chapter14 vesiculartrafficsecretionandendocytosis-140105094215-phpapp01

Chapter14 vesiculartrafficsecretionandendocytosis-140105094215-phpapp01
Chapter13 movingproteinsintomembranesandorganelles-140105094005-phpapp01

Chapter13 movingproteinsintomembranesandorganelles-140105094005-phpapp01
Chapter12 cellularenergetics-140105093734-phpapp01

Chapter12 cellularenergetics-140105093734-phpapp01
Chapter11 transmembranetransportofionsandsmallmolecules-140105092904-phpapp02

Chapter11 transmembranetransportofionsandsmallmolecules-140105092904-phpapp02
Chapter10 biomembranestructure-140105093829-phpapp02

Chapter10 biomembranestructure-140105093829-phpapp02
Chapter9 visualizingfractionatingandculturingcells-140105092245-phpapp01

Chapter9 visualizingfractionatingandculturingcells-140105092245-phpapp01
Recently uploaded
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"National Information Standards Organization (NISO)
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
Recently uploaded (20)
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
TataKelola dan KamSiber Kecerdasan Buatan v022.pdf

TataKelola dan KamSiber Kecerdasan Buatan v022.pdf
Mattingly "AI & Prompt Design: The Basics of Prompt Design"

Mattingly "AI & Prompt Design: The Basics of Prompt Design"
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Beyond the EU: DORA and NIS 2 Directive's Global Impact

Beyond the EU: DORA and NIS 2 Directive's Global Impact
18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf

18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf
Separation of Lanthanides/ Lanthanides and Actinides

Separation of Lanthanides/ Lanthanides and Actinides
Intrototitrations 140617143055-phpapp01
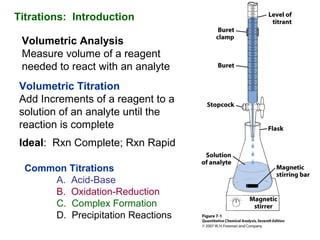
- 1. Titrations: Introduction Volumetric Analysis Measure volume of a reagent needed to react with an analyte Volumetric Titration Add Increments of a reagent to a solution of an analyte until the reaction is complete Ideal: Rxn Complete; Rxn Rapid Common Titrations A. Acid-Base B. Oxidation-Reduction C. Complex Formation D. Precipitation Reactions
- 2. Definitions Indicator - Compd whose color changes abruptly near the equiv pt Equivalence Point - pt in the titration where the quantity of titrant added is stoich. equiv to amt. of analyte present. End Point - Typically what is measured; marked by sudden change in prop. of soln Titration Error - diff btwn end pt & equiv pt Back Titration - Add excess of titrant then determine the amount excess
- 3. Example 5H2C2O4 + 2 MnO4 - + 6H+ 10 CO2 + 2 Mn+2 + 8H2O analyte titrant colorless purple Equiv point point when 2 mmol of MnO4 - has been added to 5 mmoles of H2C2O4 End point detect by monitoring color of solution better eyes, closer endpoint will be to equiv point Back titration add xs MnO4 - , back titrate with another standard to determine excess slow reactions, standard solutions not stable.
- 4. What’s Needed Solution of known composition Primary standard Standard Solution Analytical Balance Volumetric Flask Accurately measure volume Buret Volumetric Pipet
- 5. Mercuric Nitrate was standardized by titrating a solution containing 147.6 mg of NaCl. 28.06 mL of the Mercuric Nitrate solution was required to reach the end point. This solution was then used to titrate 2.000 mL of urine. It took 22.83 mL to reach the endpoint. Find the concentration of Cl- in Urine as mg/mL Hg2+ + 2 Cl- = HgCl2 Titration Calculations
- 7. The CO in a 20.3 L sample of gas was converted to CO2 by passage over iodine pentoxide heat to 150 C. I2O5 + 5CO = 5 CO2 + I2 The iodine distilled at this temperature was collected in an absorber containing 8.25 mL of 0.01101 M Na2S2O3 I2 + 2S2O3 2- = 2I- + S4O6 2- The excess Na2S2O3 was back titrated with 2.16 mL of a 0.00947 M I2 solution. Calculate the number of mgs of CO per liter of sample Titration Calculations
- 9. Applications of Acid-Base Titrations 1. Determination of Nitrogen Kjeldahl Analysis: Important method to accurately determine nitrogen in proteins and other nitrogen containing compounds
- 10. Steps Digest the material in sulfuric acid convert to NH4HSO4 Cool the solution and make it alkali to convert to NH3 (gaseous) Volatile ammonia distilled into known volume of acid Excess acid back titrated
- 11. A protein contains 16.2 wt% nitrogen. A 0.500 mL sample of the protein solution was digested and the liberated NH3 distilled into 10.00 mL of a 0.02140 M HCl solution. The unreacted HCl required 3.26 mL of a 0.0198 M NaOH. Calculate the concentration (mg/mL) of protein in the original sample. Example