Nuclear Fission & Fusion: A Guide
- 1. Nuclear Fission & Fusion
- 2. History: Hahn & Strassman (1939) •Bombarded Uranium-235 samples with neutrons expecting the Uranium-235 to capture neutrons •Instead, the products showed different chemical properties that they could not explain
- 3. •Explained Hahn & Strassman results. •Instead of heavier Uranium, it had split into smaller elements = Nuclear Fission Meitner & Frisch
- 4. Nuclear Forces 1. Electric repulsion – ________ charge particles repel each other same 1. Strong Nuclear Force – causes protons and neutrons to ________ each otherattract •Stable Nuclei = strong nuclear force is ________ than repulsion force greater •Unstable Nuclei = strong nuclear force is ________ than repulsion force oHave too many or too few neutrons in nucleus oHave more than 83 protons in nucleus oWill decay into a more stable nucleus less
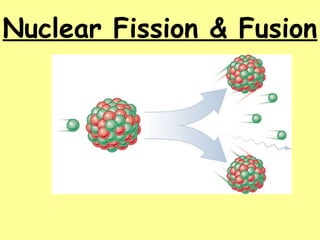
- 5. Nuclear Fission – splitting of heavier nuclei into lighter nuclei. U235 92 n1 0 Ba137 56 Xe84 36 n1 0 energy+ +15+ + How much energy? E=mc2 _______= _______ x (_____ _ _____)2 c=3.0x108 Energy mass speed of light E=mc2 explains _____ _____ (total mass of nucleus is less than sum of individual particles) mass defect
- 7. Nuclear Chain Reactions: •Nuclear fission releases more neutrons which trigger more fission reactions •The number of ________ released determines the success of a chain reaction neutrons
- 9. Applications of Controlling Chain Reactions • Must have a minimum amount of radioactive isotope to sustain a chain reaction =_________ _______ 1. Atomic Bomb (fission bomb) – Triggering a chain reaction in U-235 or Pu-239 CRITICAL MASS 1. Nuclear Power Plants – Convert heat energy from fission chain reaction into __________.electricity Control chain reaction with ________ _____ that absorb ________ emitted after fission reaction. control rods neutrons
- 10. Nuclear Fusion - Energy released when two light nuclei combine or fuse •However, a large amount of energy is required to start a fusion reaction: o Need this energy to overcome ________ forces of protons. o Extremely high temperatures can provide start-up energy. repulsion More energy in fusing hydrogen that fission of uranium
- 11. Nuclear Fusion
- 12. Stars energy is produced through fusion reactions Fusion occurs until Fe is produced because less energy is released than required to fuse Fe nuclei = _____ ____ ____ Star burns out
- 13. Difference between nuclear fission and fussion • Fission reaction does not occur naturaly. • It produces many highly radioactive particles. • Energy released is million times grater that that in chemical reactions, but lower that the energy released by nuclear fusion • Fusion occurs in stars such as sun. • Few radioactive particles are produced by fusion reaction, but requires fussiom trigger • Energy released is three to four times greater than the energy released by fission
- 14. • One class of nuclear weapon is a fission bomb • Definition • Takes little energy to split two atoms in a fission reaction • Hydrogen bomb, which used fission reaction to trigger a fusion reaction • Definition High density, high temperature environment is required.
- 15. Radius of Gyration Protons travel in the doughnut with constant radius. Proton are injected into the doughnut from a linear accelerator or van de Graff generatore and are recovered by magnetic deflection as a pulsed beam.When protons reach their maximum energy, the oscillator frequency is distorted so taht the orbit expands or contracts and the protons strike the target. It gives the proton 10 GeV energy
- 16. General features of Nuclear reactor
- 17. REACTOR
- 18. COMPONENTS OF A REACTOR 1. Control Rods Control rods made of a material that absorbs neutrons are inserted into the bundle using a mechanism that can rise or lower the control rods. The control rods essentially contain neutron absorbers like, boron, cadmium or indium. 2. Steam Generators Steam generators are heat exchangers used to convert water into steam from heat produced in a nuclear reactor core. Either ordinary water or heavy water is used as the coolant.
- 19. 3. Steam Turbine A steam turbine is a mechanical device that extracts thermal energy from pressurized steam, and converts it into useful mechanical. Various high-performance alloys and super alloys have been used for steam generator tubing. 4. Coolant Pump The coolant pump pressurizes the coolant to pressures of the order of 155 bar. The pressure of the coolant loop is maintained almost constant with the help of the pump and a pressurize unit.
- 20. 5. Feed Pump Steam coming out of the turbine, flows through the condenser for condensation and recalculated for the next cycle of operation. The feed pump circulates the condensed water in the working fluid loop. 6. Condenser Condenser is a device or unit which is used to condense vapour into liquid. The objective of the condenser are to reduce the turbine exhaust pressure to increase the efficiency and to recover high quality feed water in the form of condensate & feed back it to the steam generator without any further treatment
- 21. 7. Cooling Tower Cooling towers are heat removal devices used to transfer process waste heat to the atmosphere. Water circulating through the condenser is taken to the cooling tower for cooling and reuse.
- 22. Types of nuclear reactor An assembly of nuclear fuel and moderator which is able to sustain reaction is called a nuclear reaction. It is a controlled chain reacting system supplying nuclear energy. It is a nuclear furnace that burns fuels like U-235, U- 233, Pu-239.
- 23. All the reactors consists of following components 1. Reactor coil 2. Reflector 3. Reactor vessel 4. Radiation shield 5. Structural materials 6. Coolant loops 7. Heat exchangers
- 24. 1. Reactor core. The central region of a reactor where the fussion takes place is known as core. It consists of nuclear fuel. In fast reactors it consists of a coolant, control rods and structural materials. A moderator is also present in thermal reactors. Fuel is in the form of ceramic Fuel Rods –An ideal fuel should have high thermal conductivity, high melting point, high resistance to radiation demage and chemically inert. To prevent th e fission fragments into the coolant or the moderators, corrosion , the fuel rods are covered with protective materials – cladding. An ideal fuel clad material must be – highly resistant to corrosion, a poor neutron absorption, cheap, easily available, good mechanical strength, high melting point. Zirconum, steel Al, Mg, Ni. Zr is the best one, LWR- steel Resarch reactors- Al
- 25. 2. Coolant – To remove fission heat from the core, it is necessary to circulate a fluid – liquid or gas through the reactor. This fluid is referred to as coolant. It must have high thermal capacity, low cost, low neutron absorption,, good radiation and thermal stability and compatibility with fuel and clad. Water, liquid sodium, gases like CO2, He 3. Moderator: to slow down the fast neutrons, to increase their fission cross section with uranium. It must have low mass number and should not absorb neutrons. Heavy water, D2O, graphite, beryllium, paraffin which are reach in protons
- 26. When a reaction takes place in uranium rod, most of the fast neutrons enters in moderator. They collide with the protons of moderator by interchanigng their energies and are slowed down. 4. Control rods: for maintaining the desired state of fission reactions within a nuclear reactor. To control the chain reaction from becoming violent, rods of boron or cadmium are inserted in the holes of reactor core. As a result of which the desired number of neutrons are absorbed and only limited numbers of neutrons are left to produce fission.
- 27. Cold Fusion: Efforts are being made to start and sustain a fusion reaction at lower temperatures, in other words with a lower amount of input energy