Case record...Intramedullary cavernoma
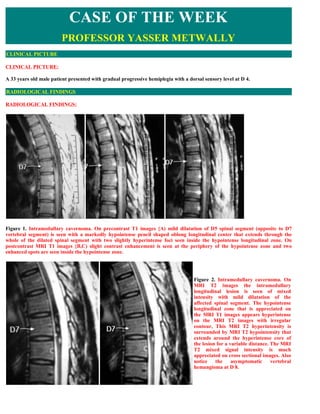
- 1. CASE OF THE WEEK PROFESSOR YASSER METWALLY CLINICAL PICTURE CLINICAL PICTURE: A 33 years old male patient presented with gradual progressive hemiplegia with a dorsal sensory level at D 4. RADIOLOGICAL FINDINGS RADIOLOGICAL FINDINGS: Figure 1. Intramedullary cavernoma. On precontrast T1 images {A) mild dilatation of D5 spinal segment (opposite to D7 vertebral segment) is seen with a markedly hypointense pencil shaped oblong longitudinal center that extends through the whole of the dilated spinal segment with two slightly hyperintense foci seen inside the hypointense longitudinal zone. On postcontrast MRI T1 images {B,C) slight contrast enhancement is seen at the periphery of the hypointense zone and two enhanced spots are seen inside the hypointense zone. Figure 2. Intramedullary cavernoma. On MRI T2 images the intramedullary longitudinal lesion is seen of mixed intensity with mild dilatation of the affected spinal segment. The hypointense longitudinal zone that is appreciated on the MRI T1 images appears hyperintense on the MRI T2 images with irregular contour, This MRI T2 hyperintensity is surrounded by MRI T2 hypointensity that extends around the hyperintense core of the lesion for a variable distance. The MRI T2 mixed signal intensity is much appreciated on cross sectional images. Also notice the asymptomatic vertebral hemangioma at D 8.
- 2. Figure 3. Intramedullary cavernoma. On MRI T2 images (A,B) the intramedullary longitudinal lesion is seen of mixed intensity with mild dilatation of the affected spinal segment. The hypointense longitudinal zone that is appreciated on the MRI T1 (C,D) images appears hyperintense on the MRI T2 images with irregular contour, This MRI T2 hyperintensity is surrounded by MRI T2 hypointensity that extends around the hyperintense core of the lesion for a variable distance. The MRI T2 mixed signal intensity is much appreciated on cross sectional images. 44 Figure 4. Intramedullary cavernoma. On MRI T2 images (B) the intramedullary longitudinal lesion is seen of mixed intensity with mild dilatation of the affected spinal segment. The hypointense longitudinal zone that is appreciated on the MRI T1 images (A) appears hyperintense on the MRI T2 images with irregular contour, This MRI T2 hyperintensity is surrounded by MRI T2 hypointensity that extends around the hyperintense core of the lesion for a variable distance. The MRI T2 mixed signal intensity is much appreciated on cross sectional images. Also notice the asymptomatic vertebral hemangioma at D 8.
- 3. Figure 5. Intramedullary cavernoma. MRI T2 images (A,B) and precontrast MRI T1 images showing a mixed signal intensity intramedullary lesion, The MRI mixed signal intensity of the precontrast MRI T1 images and the MRI T2 images is due to the presence of altered blood of different ages. Conclusion MRI picture of intramedullary cavernoma. The MRI mixed signal intensity of the precontrast MRI T1 images and the MRI T2 images is due to the presence of altered blood of different ages. 1. The MRI T1 precontrast hyperintensity is due to methemoglobin 2. The MRI T2 hypointensity is due to hemosiderin pigments. Table 1. Evolution of MR imaging signal characteristics Iron-containing Iron oxidation Red cell Phase Time T1 T2 T2* molecule state membranes Hyperacute Hours Oxyhemoglobin Fe2+ Intact Hypointense Hyperintense Acute Hours to days Deoxyhemoglobin Fe 2+ Intact iso/ Hypointense Hypointense Hypointense Early Days to 1 Methemoglobin Fe2+ Intact Hyperintense Hypointense subacute week Late 1 week to Methemoglobin Fe2+ Degraded Hyperintense Hyperintense subacute months Chronic >months Hemosiderin Fe2+ Degraded iso/ Hypointense Hypointense Hypointense Abbreviations: Fe, iron; iso, isointense relative to normal brain. DIAGNOSIS: DIAGNOSIS: INTRAMEDULLARY CAVERNOMA DISCUSSION DISCUSSION: Although they are not considered vascular lesions in the Anson and Spetzler grading system because they do not shunt blood and have no arterial feeders and draining veins, spinal cavernous malformations (spinal cavernoma) (SCM) are another cause of treatable myelopathy. 1 The frequency of spinal cavernomas is difficult to ascertain because of the low number of spinal cord autopsies. In 1976, Voigt and Yasargil could find only 164 cases of cavernous malformations (cavernoma) in the literature. 13 In anther review, Ghogawala and Ogilvy reported only 67 intramedullary spinal cavernomas in the English-language
- 4. literature. 18 With the development of CT scanning and particularly MR imaging, these lesions are being reported more often. It is estimated that cavernomas have an incidence of between 0.02% and 4% in the population 8 and that 3% to 5% of cavernomas occur in the spinal cord. 15 Spinal cavernous malformations are considered to be congenital lesions that may enlarge with time. 8 They are slow-flow lesions without arteriovenous shunting. They are found throughout the cord and are often intramedullary Unlike their intracranial counterparts, which have no gender preponderance, 70% of spinal cavernoma occur in women. 56 Sporadic forms and familial forms with autosomal dominant inheritance with variable penetrance have been described. 4,6,7 Clinical features of spinal cavernomas vary from acute to progressive to asymptomatic. 10 Ogilvy et al described four types of clinical presentations: (1) acute episodes with stepwise neurologic decline, (2) acute onset of a neurologic deficit with rapid neurologic deterioration, (3) acute onset of a mild neurologic deficit with gradual neurologic deterioration, and (4) slowly progressive neurologic decline. 16 Acute symptoms are caused by hemorrhage, which may cause enlargement of the spinal cavernoma or hematomyelia. Patients often present with sudden back pain, often at the level of the lesion. Neurologic defects may follow many hours after the attack of pain. This delayed neurologic deterioration may help differentiate spinal cavernoma hemorrhages from those of spinal AVMS. The progressive course of spinal cavernomas could be caused by small microhemorrhages, which may cause enlargement of the spinal cavernoma. Small hemorrhages with organization, capillary budding, and propagation and repetitive hemorrhages are two possible reasons for progression. 8 Other theories for progressive neurologic decline include a neurotoxic effect from red blood cell degradation or an effect on the spinal cord microcirculation. 8 On gross inspection, cavernomas are well-defined, red-to-purple, lobulated lesions often described as looking like a mulberry. Microscopically, cavernomas are made up of enlarged capillaries or sinusoidal vascular spaces with a simple endothelial lining, thin adventitia, and no elastic or smooth muscle fibers in its walls. Evidence of prior hemorrhage is commonly seen. The surrounding tissue usually has gliosis and hemosiderin. Calcification of spinal cavernoma is rare. 5 Spinal cavernomas are not pathologically distinguishable from their intracranial counterparts. 9 Because so few spinal cavernomas have been studied, the natural history of these lesions is not completely known. Management of spinal cavernoma depends on the patient's clinical symptoms and age and on the characteristics of the lesion. 5,8 For asymptomatic lesions, no treatment is currently advised. 5,8 Symptomatic lesions are often surgically explored because of the risk of future neurologic deterioration. The hemorrhage rate for cerebral cavernomas has been estimated to be between 0.5% and 1.0% per year. 16,17 Whether this rate is valid for spinal cavernomas is unclear. In a review of 57 symptomatic patients with spinal cavernomas, Canavero et al, 3 suggested a hemorrhage rate of 1.6% per year. 13 As with any spinal vascular malformation, postoperative outcome depends largely on the level of preoperative neurologic function. 12 Residual tumor is often difficult to distinguish from postoperative changes. Recurrence of spinal cavernoma after total removal and negative postoperative MR imaging has been mentioned in only one review. 8,17 Imaging of Spinal Cavernous Malformations As with other vascular lesions of the spine, MR imaging should be the first modality used to look for spinal cavernomas. Cavernous malformations are considered to be one of the angiographically occult lesions, and spinal angiography has no role in its diagnosis or treatment. Although spinal cavernoma can enlarge the cord, this enlargement is often not detected with myelographic studies. 12 MR imaging appearances of spinal cavernoma are quite similar to cerebral cavernomas. On MR imaging spinal cavernomas are round or oblong lesions with mixed signal intensity from different stages of red blood cell breakdown. Cavernous malformations are often encircled by a dark hemosiderin ring that shows characteristic blooming on gradient- recalled images. Labauge et al, 7 found gradient-echo sequences to be the most sensitive for detecting small cavernomas. There is usually no or minimal enhancement with the administration of contrast. Surrounding edema can often be seen on T2-weighted images. Whereas the MR imaging appearance is characteristic, it is not pathognomonic. Other vascular malformations or hemorrhagic neoplasms may have a similar appearance. Also, some spinal cavernomas may be hidden by hemorrhage and not visualized on MR imaging.
- 5. Figure 6. Spinal cavernous malformation. A, Sagittal Tl -weighted MR image of the cervical spine shows an area of heterogeneous signal intensity (arrow) at the level of C6 with some increased signal seen extending caudally (open arrow). B, Sagittal T2-weighted MR image demonstrates heterogeneous signal (arrow) with some decreased signal seen extending caudally (open arrow), representing subacute hemorrhage. C, Axial Tl -weighted MR image at the level of C6 shows intramedullary mixed signal in the left aspect of the spinal cord (arrow). D, Axial T2*-weighted gradient-recalled MR image at the level of C6 demonstrates low signal with a small area of high signal (arrow). SUMMARY SUMMARY Table 2. Pathological characteristics of cavernomas that differentiate them from arteriovenous malformation Cavernous angiomas (cavernomas) consist of enlarged capillaries with the following characteristics: Sinusoidal Single layer of endothelium Thin collagenous wall
- 6. Lack of smooth muscle fibers and elastic fibers Capillaries are immediately adjacent to each other, with no intervening neural tissue. They typically are not associated with enlarged feeding arteries or draining veins, and blood flow is low or even stagnated (cavernomas are angiographically occult). The surrounding parenchyma contains hemosiderin-laden macrophages and iron-laden glial cells. Grossly, they may range from soft to hard. Thrombosis, calcification, or ossification results in a harder lesion. Adjacent neural tissue may be affected, and small hemorrhages result in hemosiderin staining (iron salts may incite an epileptogenic focus). Tissues also may be gliotic. Figure 7. Histopathology. (A) Microscopic section (trichrome stain) shows a mass of closely apposed vessels of varying size. The vessel walls have a variable thickness, and there is no intervening brain. (B) Whole mount (trichrome stain) of a CM of the third ventricle shows a plexus of vessels with some intervening brain at the periphery. Figure 8. Example of cavernomas, notice absence of feeding arteries or draining veins
- 7. Figure 9. Pontine cavernoma, notice absence of feeding arteries or draining veins Figure 10. For comparison, an arteriovenous malformation, notice the feeding arteries and the draining veins Addendum A new version of this PDF file (with a new case) is uploaded in my web site every week (every Saturday and remains available till Friday.) To download the current version follow the link quot;http://pdf.yassermetwally.com/case.pdfquot;. You can also download the current version from my web site at quot;http://yassermetwally.comquot;. To download the software version of the publication (crow.exe) follow the link: http://neurology.yassermetwally.com/crow.zip The case is also presented as a short case in PDF format, to download the short case follow the link: http://pdf.yassermetwally.com/short.pdf At the end of each year, all the publications are compiled on a single CD-ROM, please contact the author to know more details.
- 8. Screen resolution is better set at 1024*768 pixel screen area for optimum display. For an archive of the previously reported cases go to www.yassermetwally.net, then under pages in the right panel, scroll down and click on the text entry quot;downloadable case records in PDF formatquot; REFERENCES References 1. Anson JA, Spetzler RF: Classification of spinal arteriovenous malformations and implications for treatment. Barrow Neurological Institute Quarterly 8:2-8, 1992 2. Awad IA: Patients with spinal cord cavernous malformations are at an increased risk for multiple neuraxis cavernous malformations [comment]. Neurosurgery 45:33,1999 3. Canavero S, Pagni C, Kornfield M, et al: Spinal intramedullary cavernous angiomas: A literature meta- analysis. Surg Neurol 43:448-452,1995 4. Halbach VV, Higashida RT, Hiehima GB: Treatment of vertebral arteriovenous fistulas. AJR Am J Roentgenol 150:405- 412,1988 5. Hurst RW: Spinal vascular disorders. In Atlas SW (ed): Magnetic Resonance Imaging of the Brain and Spine, ed 2. Philadelphia. Lippincott-Raven, 1996 pp 1387- 1412 6. Johnson PC, Wascher TM, Golfinos J, et al: Definition and pathologic features. In Awad IA, Barrow DL (eds): Cavernous Malformations. Park Ridge, IL, American Association of Neurological Surgeons, 1993, pp 1-11 7. Labauge P, Labauge S, Levy C, et al: Hereditary cerebral cavernous angiomas: Clinical and genetic features in 57 French families. Lancet 352:1892-1897,1998 8. McCormick WF, Nofzinger JD: quot;Cryptickquot; vascular malformations of the central nervous system. J Neurosurg 24:865- 875, 1966 9. Mehmet KY, Wecht DA, Awad IA: Clinical spectrum and natural history of spinal vascular malformations. In Barrow DL, Awad IA (eds): Spinal Vascular Malformations. Park Ridge, IL, American Association of Neurological Surgeons, 1999, pp 45-55 10. Ogilvy C, Louis D, Ojemann R: Intramedullary cavernous angiomas of the spinal cord: Clinical presentation, pathological features, and surgical management. Neurosurgery 31:219-230,1992 11. Oldfield EH: Spinal vascular malformations. In Wilkins RH, Rengachary SS (eds): Neurosurgery, ed 2, vol 2. New York, McGraw-Hill, 1996, pp 2541-2558 12. Vishteh GA, Zabramski JM, Spetzler RF: Patients with spinal cord cavernous malformations are at an increased risk for multiple neuraxis cavernous malformations. Neurosurgery 45:30-32, 1999 13. Voigt K, Yasargil MG: Cerebral cavernous hemangiomas or cavernomas: Incidence, pathology, localization, diagnosis, clinical features and treatment: Review of the literature and report of an unusual case. Neurochirurgia 19:59,1976 14. Wong JH, Awad IA: Spinal vascular malformations. In Jafar jj, Awad IA, Rosenwasser RH (eds): Vascular Malformations of the Central Nervous System. Philadelphia, Lippincott Williams & Wilkins, 1999, pp 155-160 15. Zimmerman R, Spetzler R, Lee K, et al: Cavernous malformations of the brainstem. J Neurosurg 75:32- 39,1991 16. Ogilvy C, Louis D, Ojemann R: Intramedullary cavernous angiomas of the spinal cord: Clinical presentation, pathological features, and surgical management. Neurosurgery 31:219-230,1992 17. Metwally, MYM: Textbook of neuroimaging, A CD-ROM publication, (Metwally, MYM editor) WEB-CD agency for electronic publication, version 9.2a April 2008 18. Ghogawala Z, Ogilvy OS: Intramedullary cavernous malformations of the spinal cord. Neurosurg Clin North Am 10:101-109,1999
