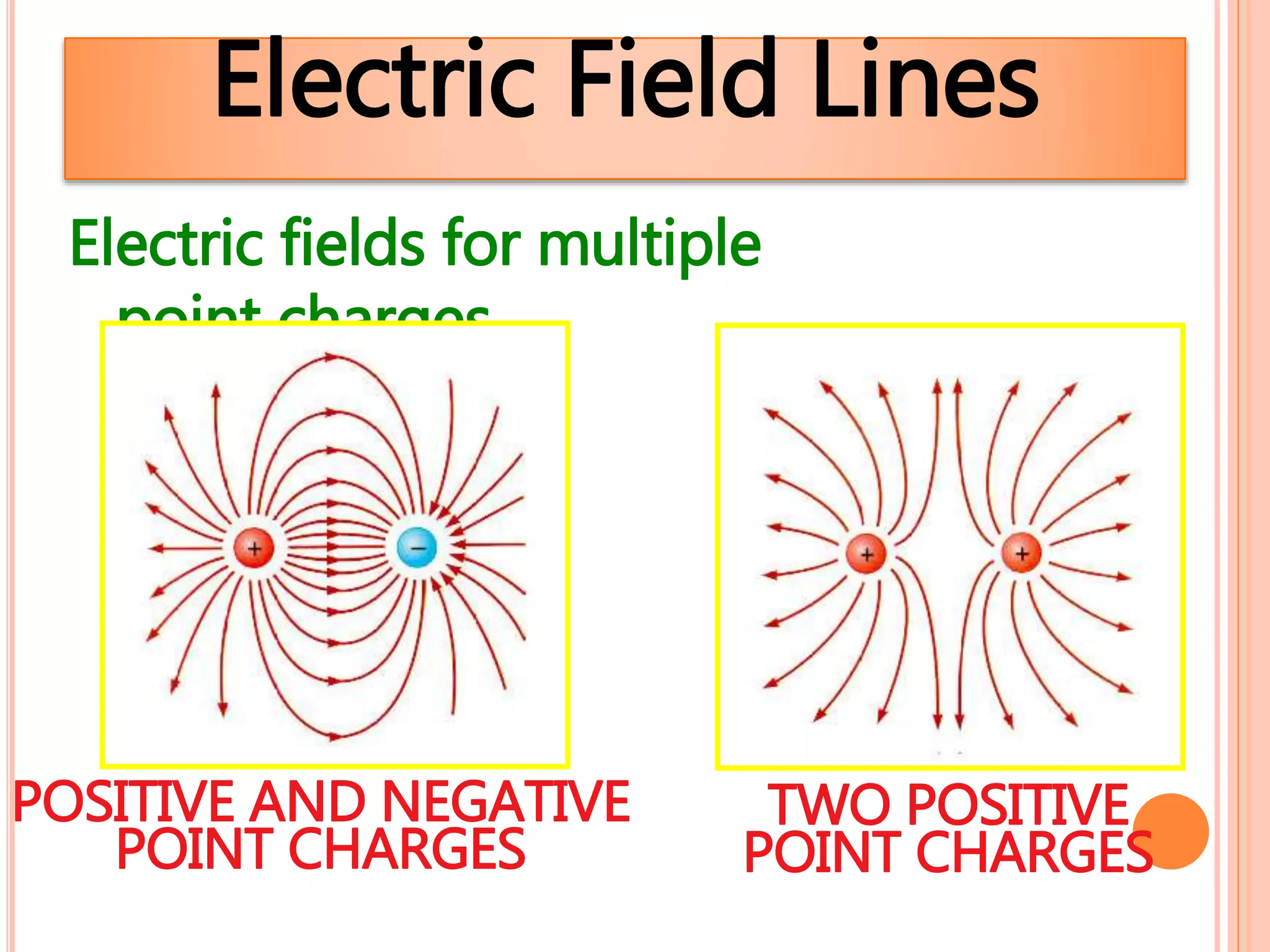
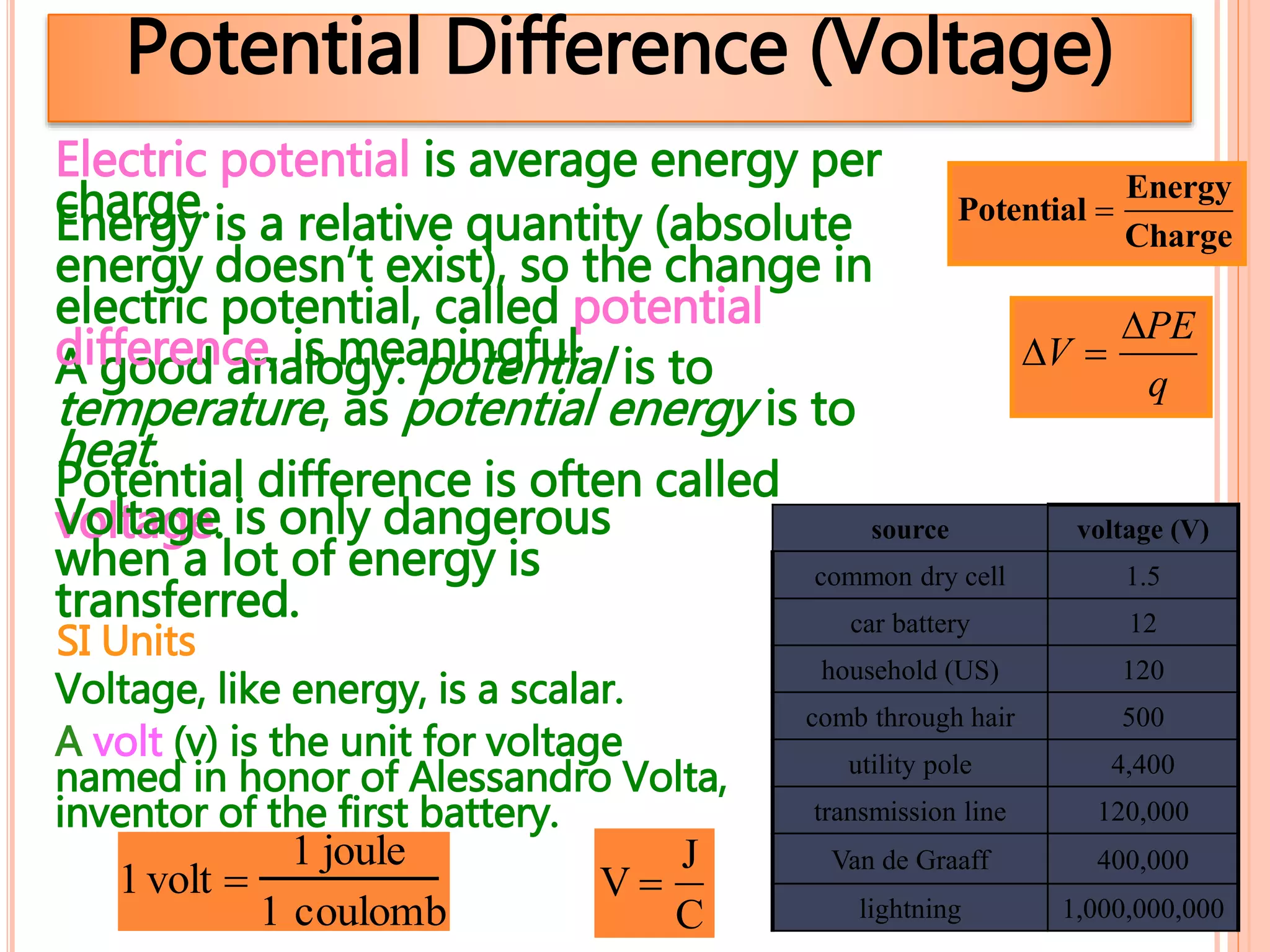
1. Electrostatics is the study of stationary or static electricity and its behavior in and between objects.
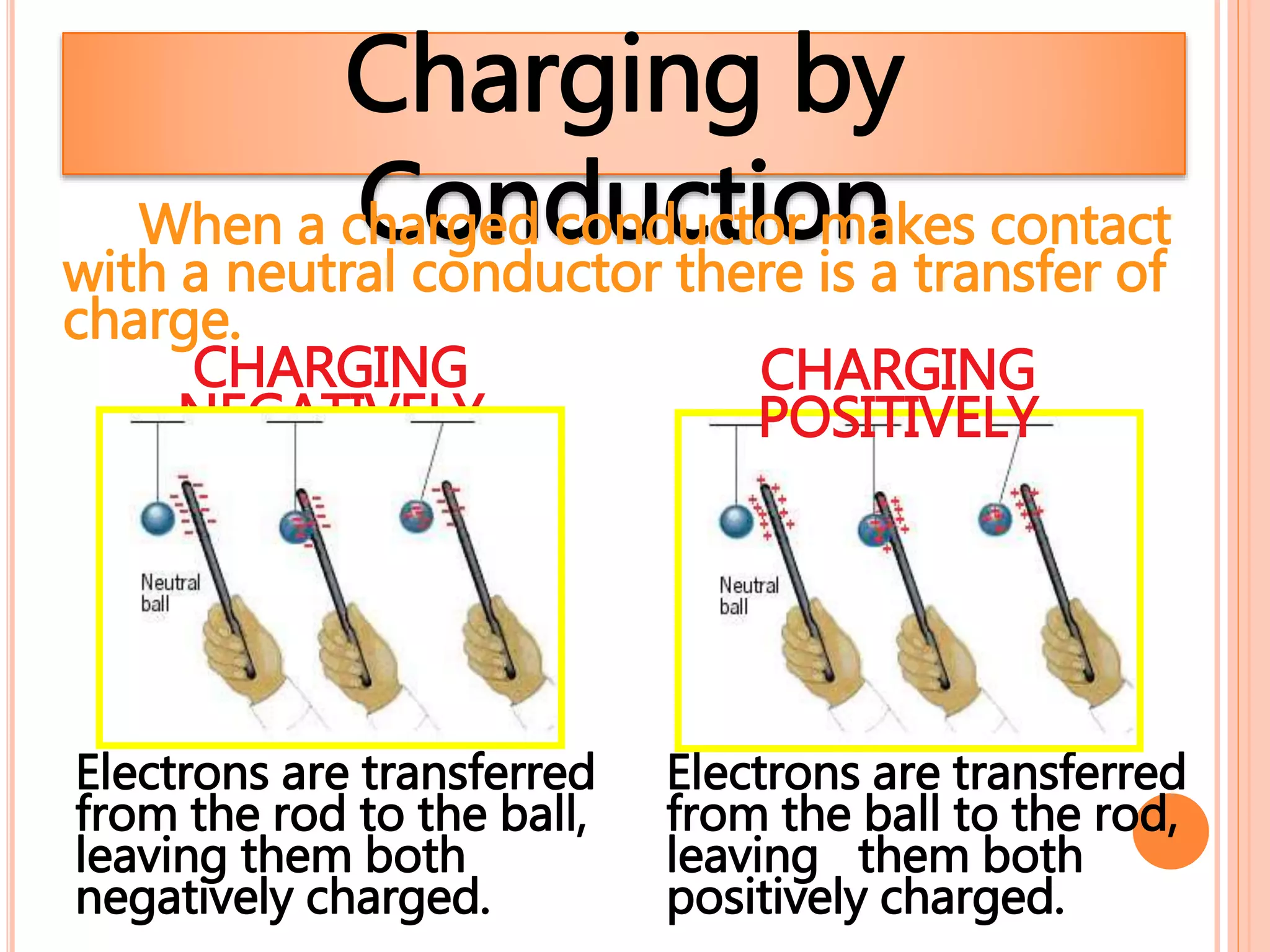
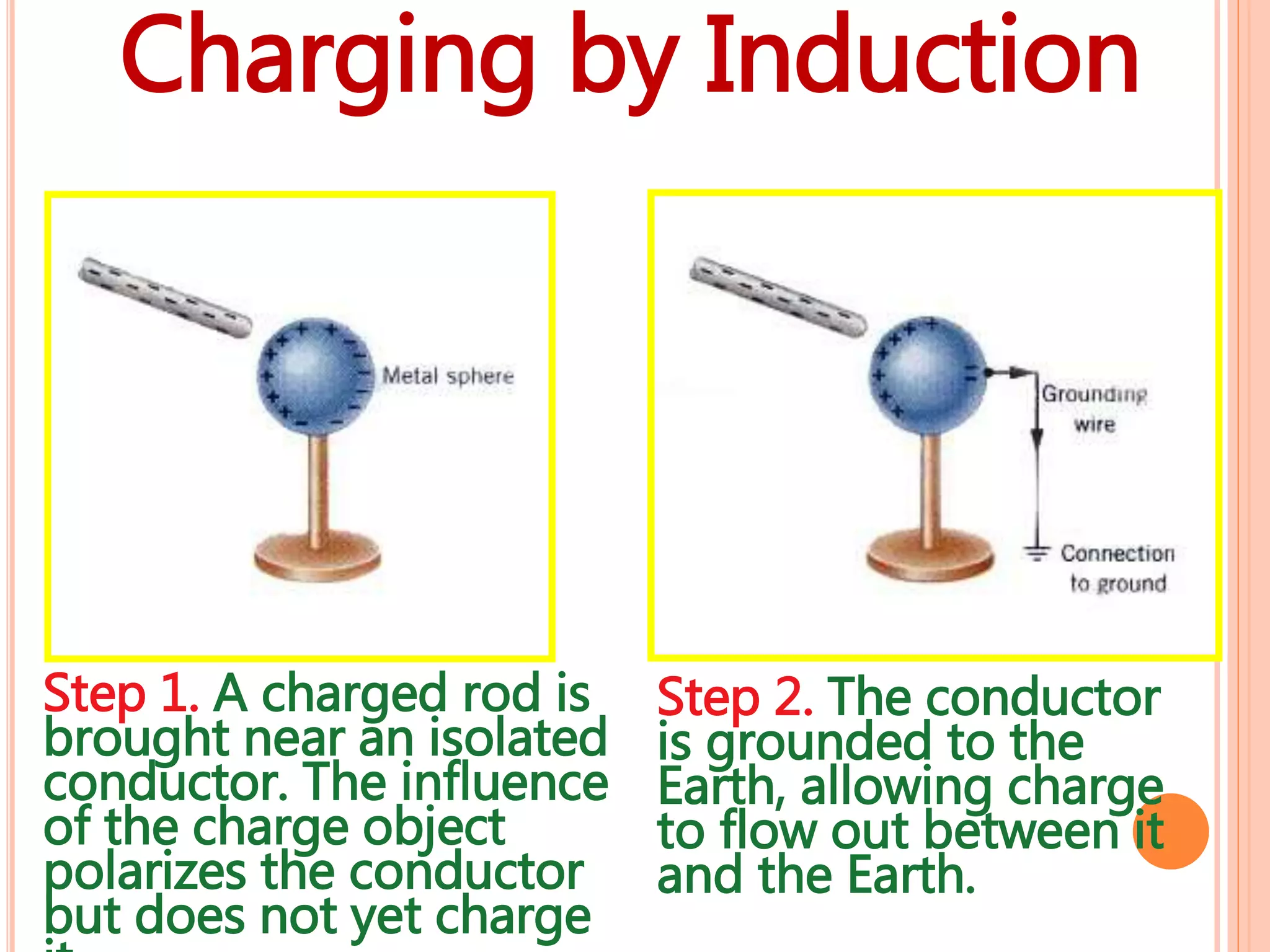
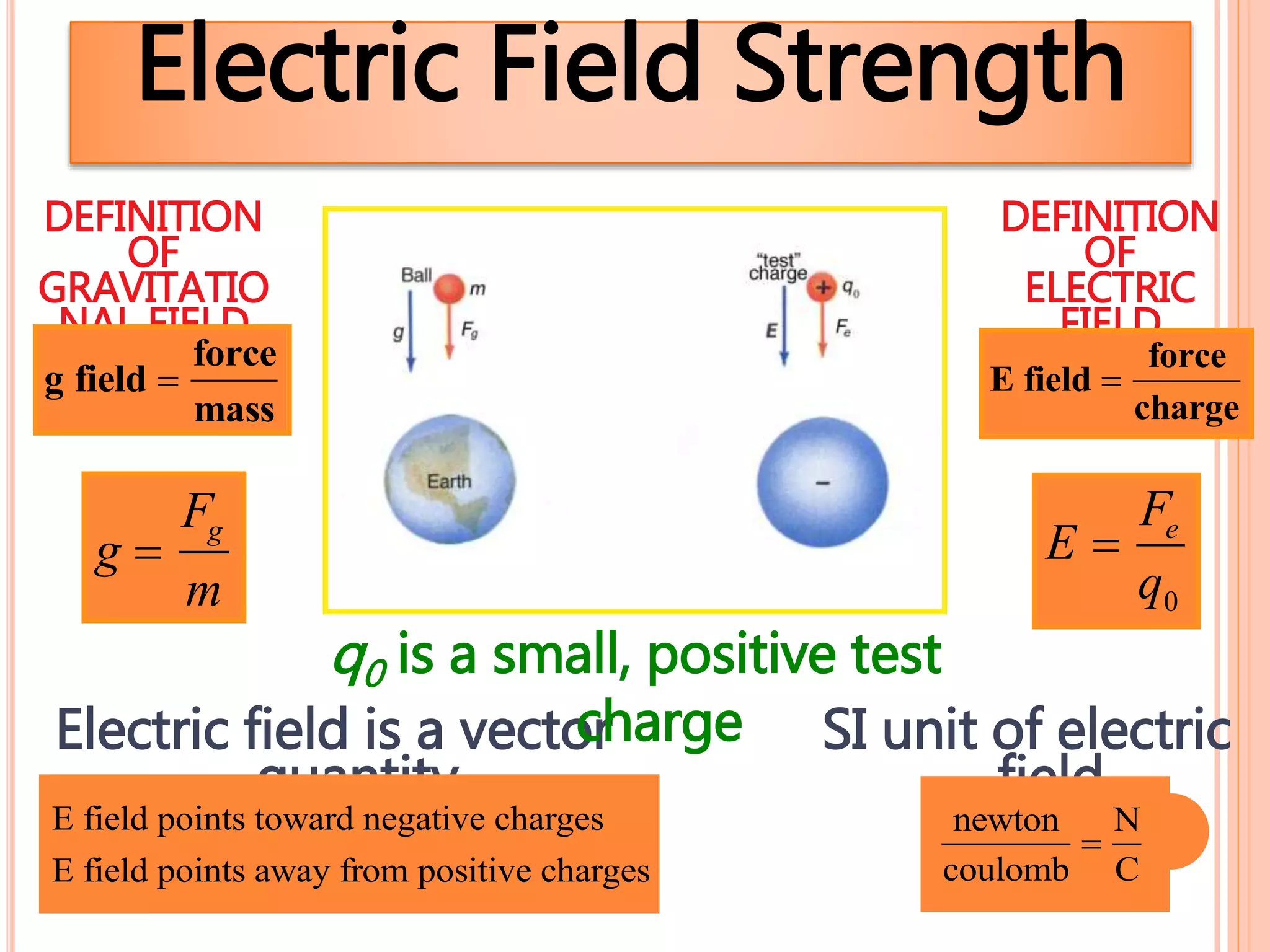
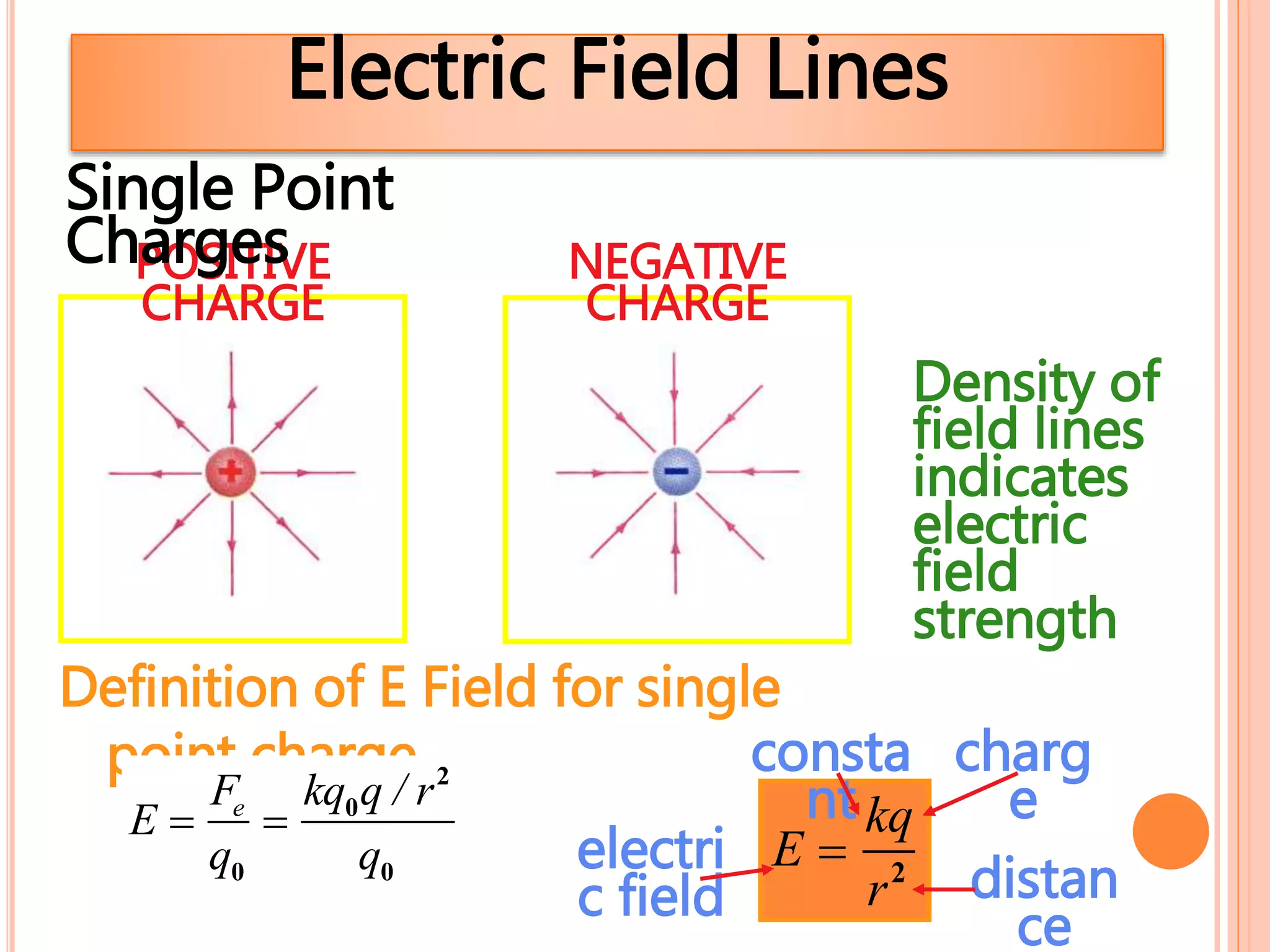
2. There are two types of charge - positive and negative - which attract each other but causes like charges to repel. Charge is conserved and cannot be created or destroyed.
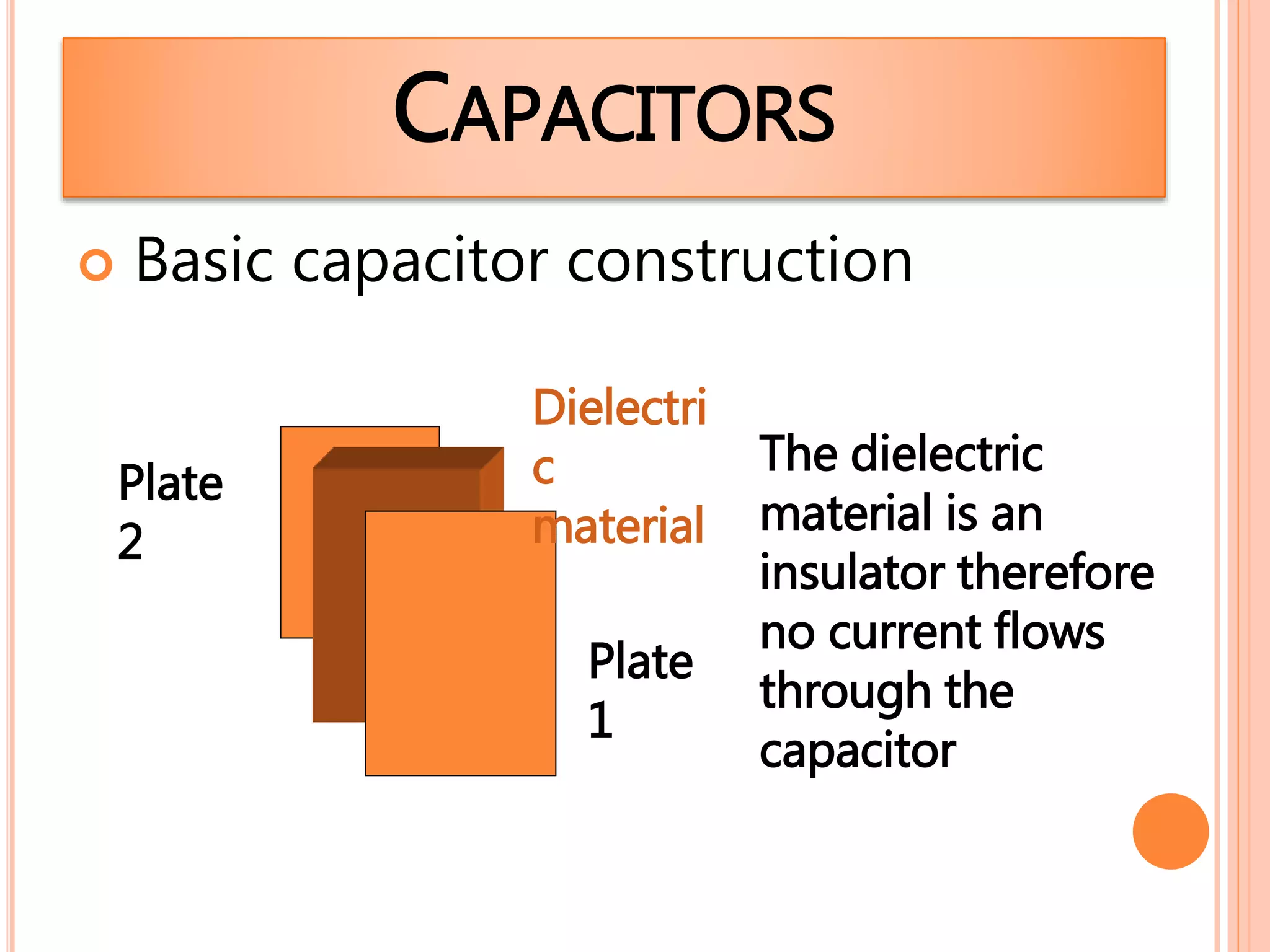
3. Conductors allow charge to flow through them, while insulators do not. Common conductors include metals, and common insulators include wood, plastic and glass.