Report
Share
Download to read offline
Recommended
Recommended
Marc i Lluis / Si has begut o consumit drogues, no agafis la moto

Marc i Lluis / Si has begut o consumit drogues, no agafis la motoConcurs Joves i Conducció 5ª edició (RACC)
More Related Content
Viewers also liked
Marc i Lluis / Si has begut o consumit drogues, no agafis la moto

Marc i Lluis / Si has begut o consumit drogues, no agafis la motoConcurs Joves i Conducció 5ª edició (RACC)
Viewers also liked (19)
Marc i Lluis / Si has begut o consumit drogues, no agafis la moto

Marc i Lluis / Si has begut o consumit drogues, no agafis la moto
El humo de pipa reaviva los recuerdos (jack hick)s

El humo de pipa reaviva los recuerdos (jack hick)s
Kitengesa Community Lilbrary newsletter March 2012

Kitengesa Community Lilbrary newsletter March 2012
Similar to Tot d lieb
Similar to Tot d lieb (20)
Thermoelastic Damping of Vibrations in a Transversely Isotropic Hollow Cylinder 

Thermoelastic Damping of Vibrations in a Transversely Isotropic Hollow Cylinder
ATOICV1-1-3-Bent-Rule-and-Energetic-of-Hybridization.pptx

ATOICV1-1-3-Bent-Rule-and-Energetic-of-Hybridization.pptx
Numerical Analysis of heat transfer in a channel Wit in clined baffles

Numerical Analysis of heat transfer in a channel Wit in clined baffles
More from Σωκράτης Ρωμανίδης
More from Σωκράτης Ρωμανίδης (20)
Ακολουθία και Παράκληση Οσίου Παϊσίου του Αγιορείτου

Ακολουθία και Παράκληση Οσίου Παϊσίου του Αγιορείτου
Ceb1cebacebfcebbcebfcf85ceb8ceb9ceb1 cebfcf83ceb9cebfcf85-ceb4ceb9cebfcebdcf8...

Ceb1cebacebfcebbcebfcf85ceb8ceb9ceb1 cebfcf83ceb9cebfcf85-ceb4ceb9cebfcebdcf8...
Recently uploaded
💉💊+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHABI}}+971581248768
+971581248768 Mtp-Kit (500MG) Prices » Dubai [(+971581248768**)] Abortion Pills For Sale In Dubai, UAE, Mifepristone and Misoprostol Tablets Available In Dubai, UAE CONTACT DR.Maya Whatsapp +971581248768 We Have Abortion Pills / Cytotec Tablets /Mifegest Kit Available in Dubai, Sharjah, Abudhabi, Ajman, Alain, Fujairah, Ras Al Khaimah, Umm Al Quwain, UAE, Buy cytotec in Dubai +971581248768''''Abortion Pills near me DUBAI | ABU DHABI|UAE. Price of Misoprostol, Cytotec” +971581248768' Dr.DEEM ''BUY ABORTION PILLS MIFEGEST KIT, MISOPROTONE, CYTOTEC PILLS IN DUBAI, ABU DHABI,UAE'' Contact me now via What's App…… abortion Pills Cytotec also available Oman Qatar Doha Saudi Arabia Bahrain Above all, Cytotec Abortion Pills are Available In Dubai / UAE, you will be very happy to do abortion in Dubai we are providing cytotec 200mg abortion pill in Dubai, UAE. Medication abortion offers an alternative to Surgical Abortion for women in the early weeks of pregnancy. We only offer abortion pills from 1 week-6 Months. We then advise you to use surgery if its beyond 6 months. Our Abu Dhabi, Ajman, Al Ain, Dubai, Fujairah, Ras Al Khaimah (RAK), Sharjah, Umm Al Quwain (UAQ) United Arab Emirates Abortion Clinic provides the safest and most advanced techniques for providing non-surgical, medical and surgical abortion methods for early through late second trimester, including the Abortion By Pill Procedure (RU 486, Mifeprex, Mifepristone, early options French Abortion Pill), Tamoxifen, Methotrexate and Cytotec (Misoprostol). The Abu Dhabi, United Arab Emirates Abortion Clinic performs Same Day Abortion Procedure using medications that are taken on the first day of the office visit and will cause the abortion to occur generally within 4 to 6 hours (as early as 30 minutes) for patients who are 3 to 12 weeks pregnant. When Mifepristone and Misoprostol are used, 50% of patients complete in 4 to 6 hours; 75% to 80% in 12 hours; and 90% in 24 hours. We use a regimen that allows for completion without the need for surgery 99% of the time. All advanced second trimester and late term pregnancies at our Tampa clinic (17 to 24 weeks or greater) can be completed within 24 hours or less 99% of the time without the need surgery. The procedure is completed with minimal to no complications. Our Women's Health Center located in Abu Dhabi, United Arab Emirates, uses the latest medications for medical abortions (RU-486, Mifeprex, Mifegyne, Mifepristone, early options French abortion pill), Methotrexate and Cytotec (Misoprostol). The safety standards of our Abu Dhabi, United Arab Emirates Abortion Doctors remain unparalleled. They consistently maintain the lowest complication rates throughout the nation. Our Physicians and staff are always available to answer questions and care for women in one of the most difficult times in their lives. The decision to have an abortion at the Abortion Cl+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...?#DUbAI#??##{{(☎️+971_581248768%)**%*]'#abortion pills for sale in dubai@
Recently uploaded (20)
ICT role in 21st century education and its challenges

ICT role in 21st century education and its challenges
AWS Community Day CPH - Three problems of Terraform

AWS Community Day CPH - Three problems of Terraform
Apidays New York 2024 - The value of a flexible API Management solution for O...

Apidays New York 2024 - The value of a flexible API Management solution for O...
Repurposing LNG terminals for Hydrogen Ammonia: Feasibility and Cost Saving

Repurposing LNG terminals for Hydrogen Ammonia: Feasibility and Cost Saving
+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...

+971581248768>> SAFE AND ORIGINAL ABORTION PILLS FOR SALE IN DUBAI AND ABUDHA...
Boost Fertility New Invention Ups Success Rates.pdf

Boost Fertility New Invention Ups Success Rates.pdf
Connector Corner: Accelerate revenue generation using UiPath API-centric busi...

Connector Corner: Accelerate revenue generation using UiPath API-centric busi...
Why Teams call analytics are critical to your entire business

Why Teams call analytics are critical to your entire business
Spring Boot vs Quarkus the ultimate battle - DevoxxUK

Spring Boot vs Quarkus the ultimate battle - DevoxxUK
Strategies for Landing an Oracle DBA Job as a Fresher

Strategies for Landing an Oracle DBA Job as a Fresher
Apidays New York 2024 - The Good, the Bad and the Governed by David O'Neill, ...

Apidays New York 2024 - The Good, the Bad and the Governed by David O'Neill, ...
Apidays New York 2024 - Accelerating FinTech Innovation by Vasa Krishnan, Fin...

Apidays New York 2024 - Accelerating FinTech Innovation by Vasa Krishnan, Fin...
2024: Domino Containers - The Next Step. News from the Domino Container commu...

2024: Domino Containers - The Next Step. News from the Domino Container commu...
Tot d lieb
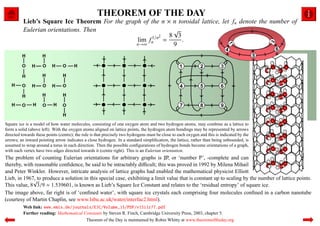
- 1. THEOREM OF THE DAY Lieb’s Square Ice Theorem For the graph of the n × n toroidal lattice, let fn denote the number of Eulerian orientations. Then √ 1/n2 8 3 lim fn = . n→∞ 9 Square ice is a model of how water molecules, consisting of one oxygen atom and two hydrogen atoms, may combine as a lattice to form a solid (above left). With the oxygen atoms aligned on lattice points, the hydrogen atom bondings may be represented by arrows directed towards these points (centre); the rule is that precisely two hydrogens must be close to each oxygen and this is indicated by the arrows; an inward pointing arrow indicates a close hydrogen. In a standard simplification, the lattice, rather than being unbounded, is assumed to wrap around a torus in each direction. Then the possible configurations of hydrogen bonds become orientations of a graph, with each vertex have two edges directed towards it (centre right). This is an Eulerian orientation. The problem of counting Eulerian orientations for arbitrary graphs is ♯P, or ‘number P’, -complete and can thereby, with reasonable confidence, be said to be intractably difficult; this was proved in 1992 by Milena Mihail and Peter Winkler. However, intricate analysis of lattice graphs had enabled the mathematical physicist Elliott Lieb, in 1967, to produce a solution in this special case, exhibiting a limit value that is constant up to scaling by the number of lattice points. √ This value, 8 3/9 ≈ 1.539601, is known as Lieb’s Square Ice Constant and relates to the ‘residual entropy’ of square ice. The image above, far right is of ‘confined water’, with square ice crystals each comprising four molecules confined in a carbon nanotube (courtesy of Martin Chaplin, see www.lsbu.ac.uk/water/interfac2.html). Web link: www.emis.de/journals/EJC/Volume_15/PDF/v15i1r77.pdf Further reading: Mathematical Constants by Steven R. Finch, Cambridge University Press, 2003, chapter 5. Theorem of the Day is maintained by Robin Whitty at www.theoremoftheday.org
