What is Pfund series of the hydrogen spectrum?
•
0 likes•4 views
The electron transitions from n ≥6 to n=5 result in the emergence of a sequence of spectral lines in the Pfund series of the hydrogen spectrum. Consequently, the n1 and n2 values of the Brackett series vary from 5 to infinity.
Report
Share
Report
Share
Download to read offline
Recommended
de-Broglie equation.pdf

If everyone is thinking alike, we cannot solve the hurdles of a problem. It seems true if we observe these hypotheses' hierarchy once. Italian physicist Francesco Maria Grimaldi discovered the wave phenomenon of light in 1665. But the uncertainty about light's nature was finally solved by Einstein's explanation of the photoelectric effect. Similarly, Neil Bohr succeeded in describing the structure of an atom with quantized electron orbits. But his stipulation of allowed stationary orbits was only a supposition until the discovery of the de-Broglie equation.
Louis de-Broglie, a French physicist, presumed that moving microscopic and macroscopic objects are waves. He introduced a word called 'matter wave' to describe the waves of material objects in motion. As a result, matter exhibits a dual character of both particle and wave. Moreover, he derived an empirical formula to measure the wavelength of matter waves in 1923 called the de-Broglie equation.
Wien displacement law graph.pdf

It is a graphical explanation of a blackbody's emissive power and radiation wavelength at various temperatures. We know blackbody emissions are temperature specific. And a blackbody graph is an overall variation of a blackbody's emissive power as a function of wavelength measured at different temperatures. But the Wien displacement law graph focuses on spectral intensities of blackbody emissions as a function of wavelength at a peak wavelength state λmax at a specific temperature T.
MCQs of blackbody & its radiation .pdf

It is an e-book of MCQs & answers on blackbody. It is in PowerPoint format with 40 questions and detailed answer explanation that makes you clear every single point of blackbody and its radiation. The e-book has 54 beautifully designed slides. The contents of the e-book are below.
Multiple choice questions and answers - 20 no.s
True or false questions-10 no.s
Reasoning questions - 10 no.s
To disclose all the contents of the e-book, visit this link.
https://kameswariservices201831.myinstamojo.com/product/3556812/mcqs-answers-of-blackbody-and-its-radiation
Fatafat check list of blackbody.pdf

A blackbody is an imaginary object that absorbs all incident light without reflecting any, invented by German physicist Gustav Kirchhoff in 1860 to help understand thermal radiation. Examples of materials that approximate a blackbody include lamp black, graphite, and platinum black. The energy distribution of electromagnetic radiation emitted by a blackbody, known as blackbody radiation, depends only on the body's temperature according to Planck's law.
Wien displacement law .pdf

It discusses Wien displacement law. To go through the numerical problems of the Wien displacement law topic, visit our e-book at;
https://kameswariservices201831.myinstamojo.com/product/3493453/numerical-problems-on-wien-displacement-law-
Blackbody radiations infographic.pdf

The blackbody is a hollow enclosure with a pinhole to emit its radiations. The secure covering of the blackbody prevents the absorbed light from escaping.
A blackbody is a solid closed unreal body that is inexistent. But lamp black, platinum black, and graphite-coated surfaces are non-ideal black bodies for laboratory purposes. An object with above 0.95 emissivities is an approximate blackbody. Besides, the hotter bodies emitting electromagnetic radiation under thermal equilibrium conditions are also considered partial black bodies.
Blackbody mind map.pdf

An ideal black body is an imaginary concept developed by Gustav Kirchhoff that perfectly absorbs all electromagnetic radiation that falls on it and reflects none. It plays a significant role in quantum mechanics by being a perfect emitter and absorber of thermal radiation, releasing electromagnetic radiation when heated that follows specific laws like Planck's quantum law, Wien's displacement law, and Stefan-Boltzmann law. Real blackbodies have opaque, closed surfaces that absorb radiation based on factors like temperature, emissivity, and surface area, and their radiation is used in applications like thermal imaging, optical sensors, and burglar alarms.
Blackbody Vs. graybody.pdf

It explains the difference between a blackbody and a gray body following Kirchhoff's law of thermal radiation.
Recommended
de-Broglie equation.pdf

If everyone is thinking alike, we cannot solve the hurdles of a problem. It seems true if we observe these hypotheses' hierarchy once. Italian physicist Francesco Maria Grimaldi discovered the wave phenomenon of light in 1665. But the uncertainty about light's nature was finally solved by Einstein's explanation of the photoelectric effect. Similarly, Neil Bohr succeeded in describing the structure of an atom with quantized electron orbits. But his stipulation of allowed stationary orbits was only a supposition until the discovery of the de-Broglie equation.
Louis de-Broglie, a French physicist, presumed that moving microscopic and macroscopic objects are waves. He introduced a word called 'matter wave' to describe the waves of material objects in motion. As a result, matter exhibits a dual character of both particle and wave. Moreover, he derived an empirical formula to measure the wavelength of matter waves in 1923 called the de-Broglie equation.
Wien displacement law graph.pdf

It is a graphical explanation of a blackbody's emissive power and radiation wavelength at various temperatures. We know blackbody emissions are temperature specific. And a blackbody graph is an overall variation of a blackbody's emissive power as a function of wavelength measured at different temperatures. But the Wien displacement law graph focuses on spectral intensities of blackbody emissions as a function of wavelength at a peak wavelength state λmax at a specific temperature T.
MCQs of blackbody & its radiation .pdf

It is an e-book of MCQs & answers on blackbody. It is in PowerPoint format with 40 questions and detailed answer explanation that makes you clear every single point of blackbody and its radiation. The e-book has 54 beautifully designed slides. The contents of the e-book are below.
Multiple choice questions and answers - 20 no.s
True or false questions-10 no.s
Reasoning questions - 10 no.s
To disclose all the contents of the e-book, visit this link.
https://kameswariservices201831.myinstamojo.com/product/3556812/mcqs-answers-of-blackbody-and-its-radiation
Fatafat check list of blackbody.pdf

A blackbody is an imaginary object that absorbs all incident light without reflecting any, invented by German physicist Gustav Kirchhoff in 1860 to help understand thermal radiation. Examples of materials that approximate a blackbody include lamp black, graphite, and platinum black. The energy distribution of electromagnetic radiation emitted by a blackbody, known as blackbody radiation, depends only on the body's temperature according to Planck's law.
Wien displacement law .pdf

It discusses Wien displacement law. To go through the numerical problems of the Wien displacement law topic, visit our e-book at;
https://kameswariservices201831.myinstamojo.com/product/3493453/numerical-problems-on-wien-displacement-law-
Blackbody radiations infographic.pdf

The blackbody is a hollow enclosure with a pinhole to emit its radiations. The secure covering of the blackbody prevents the absorbed light from escaping.
A blackbody is a solid closed unreal body that is inexistent. But lamp black, platinum black, and graphite-coated surfaces are non-ideal black bodies for laboratory purposes. An object with above 0.95 emissivities is an approximate blackbody. Besides, the hotter bodies emitting electromagnetic radiation under thermal equilibrium conditions are also considered partial black bodies.
Blackbody mind map.pdf

An ideal black body is an imaginary concept developed by Gustav Kirchhoff that perfectly absorbs all electromagnetic radiation that falls on it and reflects none. It plays a significant role in quantum mechanics by being a perfect emitter and absorber of thermal radiation, releasing electromagnetic radiation when heated that follows specific laws like Planck's quantum law, Wien's displacement law, and Stefan-Boltzmann law. Real blackbodies have opaque, closed surfaces that absorb radiation based on factors like temperature, emissivity, and surface area, and their radiation is used in applications like thermal imaging, optical sensors, and burglar alarms.
Blackbody Vs. graybody.pdf

It explains the difference between a blackbody and a gray body following Kirchhoff's law of thermal radiation.
MCQs of blackbody & Kirchhoff's law.pdf

It is a PowerPoint presentation on multiple choice questions and answers on Blackbody & Kirchhoff's law topics. It discusses all practical examples and numerical problems of the said concepts.
Planck quantum theory.pdf

This PowerPoint presentation explains Planck's quantum theory and Planck's constant topics briefly. It is now available on Instamojo for just Rs.10/-. Save a personalized copy of this presentation by visiting Instamojo. The link is below.
https://kameswariservices201831.myinstamojo.com/product/3439450/planck-quantum-theory-postulates
Kirchhoff's law (1).pdf

It is a PowerPoint presentation of Kirchhoff's law of thermal radiation. And it gives a brief idea of Kirchhoff's law, its applications, and examples. Moreover, it explains the terms absorptivity, emissive power, thermal equilibrium, and emissivity.
Kirchhoff's law.pdf

It discusses Kirchhoff's law and its examples. It explains blackbody emissions in thermal equilibrium conditions.
We are sorry to inform you about a small error in Kirchhoff's formula section of the above infographic. Kirchhoff's formula states that the absorptivity and emissivity of the body are equal in thermal equilibrium conditions. 'a' is the absorbing power of the body at a wavelength λ. And 'e' is the emissivity of the body at the same wavelength. So, we will write aλ=e but not aλ=eλ. We request you read it correctly while going through the PDF.
Kirchhoff's law formula and its derivation. Numerical problems.pdf

It discusses Kirchhoff's formula and its derivation. And it includes numerical problems of Kirchhoff's law. For a colorful e-book for only 10/- rupees on Kirchhoff's law, visit our store at;
https://kameswariservices201831.myinstamojo.com/product/3467221/kirchhoffs-law-of-thermal-radiations
Planck's constant.pdf

It describes the definition of Planck's constant. Planck constant helps compute the discrete energy changes of a body by relating to the frequency of the photon. Planck constant explains the proportionality relationship between the photon's energy and the frequency.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/08/plancks-constant-chemistry-learners.html
Numerical problems of Planck's quantum theory.pdf

It is a PowerPoint presentation on numerical problems of Planck quantum theory topic.
It includes 12 numerical problems with solved answers. Besides, this PowerPoint presentation has a mind map to remember all formulas of Planck quantum law.
MCQ on Planck constant.pdf

This document contains 10 multiple choice questions about Planck's constant. It provides explanations for why Planck's constant was introduced, its value in different units, and that it explains both the quantum and particle nature of light. The questions cover topics like the relationship between photon energy and frequency, experimental methods used to determine Planck's constant, and why LED lights are used in these calculations. Additional resources on Planck's constant and quantum theory are provided at the end.
Detailed light theories.pdf

It explains both wave & particle theories of light. It also includes Maxwell's mathematical equations and postulates of Planck quantum theory.
Theories of light.pdf

It explains the theories of light, including electromagnetic wave theory and Planck's quantum theory.
MCQ's on hydrogen spectrum.pdf

The document contains 10 multiple choice questions about the hydrogen spectrum. It discusses that the essential condition to observe the hydrogen spectrum is high temperature and low pressure. The hydrogen spectrum appears as distinct lines, with the prominent line being red with a wavelength of 656 nm. It is an example of a line spectrum and can be recorded using a spectroscope. Robert Bunsen and Gustav Kirchhoff discovered the hydrogen spectrum and spectroscope, while Isaac Newton first discovered the word "spectrum".
Rydberg equation.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
Some important points of Rydberg formula.pdf

Rydberg formula calculates the wavenumber of spectral lines obtained in the spectra of single electron hydrogen-like atoms. Rydberg's envision to measure the wavenumber of spectral energies instead of wavelengths had brought a distinguishing variation in the spectral evaluations.
The applications of spectral studies.pdf

It discusses the importance of spectral studies. The Rydberg-Ritz combination principle deals with the identification of unknown spectral lines of the atomic spectrum. Besides, spectral studies have much significance in the analysis of the composition of astronomical matter. The study of emission and absorption spectra of the substance provides information on its structure. In particular, astronomers use this spectroscopic data to determine the constituents of stars and interstellar matter.
Rydberg-Ritz combination principle.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
For more information on this topic, kindly visit our blog at;
https://jayamchemistrylearners.blogspot.com/2022/05/rydberg-ritz-combination-principle.html
Rydberg formula.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/06/rydberg-formula-chemistry-learners.html
Comparison between the Rydberg formula and Balmer formula.pdf

In 1888, Rydberg put forward an empirical relation to calculate the wavelengths of spectral emission lines of the hydrogen spectral series. It is an empirical generalization of the Balmer formula. Here are the points of difference between the Rydberg formula and the Balmer formula.
Rydberg formula:
It determines the wavenumbers of spectral lines of all hydrogen spectral series.
It calculates the spectral energies in terms of wavenumbers.
Johannes Rydberg developed it.
It gave incorrect results for multi-electron systems due to the screening effect. So, it is only applicable to hydrogen and other single-electron atoms.
Balmer formula:
It determines the wavelengths of hydrogen spectral lines in the visible region only.
It calculates the spectral energies in terms of wavelengths.
Johann Jakob Balmer discovered it.
It is only applicable to the Balmer series of the hydrogen spectrum.
Tour plan.pdf

Go for free-cancellation accommodations in a central location between peak seasons to save money. Consider hostels and avoid peak weekends, when prices triple in big cities. Carefully choose accommodations based on your budget, travel style, comfort level, group size, and interests.
Wavelength & frequency relationship of an electromagnetic wave.pdf

1. The wavelength of an electromagnetic wave is the distance between identical points on adjacent waveforms, measured as the distance between crests or troughs for transverse waves and compressions or rarefactions for longitudinal waves.
2. The frequency is the number of waves that pass through a given point in a unit of time, inversely related to wavelength, and velocity equals wavelength multiplied by frequency.
3. Shorter waves have higher frequencies as wavelength and frequency are inversely proportional - when wavelength decreases, frequency increases.
Chemistry exam.pdf

The document provides tips for solving numerical chemistry problems in an upcoming exam. It advises to carefully read the question, identify the key information needed to solve it, recall the appropriate formula, and do the calculations to find the answer. The document also includes a link to a chemistry learning blog for additional help.
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশের অর্থনৈতিক সমীক্ষা ২০২৪ [Bangladesh Economic Review 2024 Bangla.pdf] কম্পিউটার , ট্যাব ও স্মার্ট ফোন ভার্সন সহ সম্পূর্ণ বাংলা ই-বুক বা pdf বই " সুচিপত্র ...বুকমার্ক মেনু 🔖 ও হাইপার লিংক মেনু 📝👆 যুক্ত ..
আমাদের সবার জন্য খুব খুব গুরুত্বপূর্ণ একটি বই ..বিসিএস, ব্যাংক, ইউনিভার্সিটি ভর্তি ও যে কোন প্রতিযোগিতা মূলক পরীক্ষার জন্য এর খুব ইম্পরট্যান্ট একটি বিষয় ...তাছাড়া বাংলাদেশের সাম্প্রতিক যে কোন ডাটা বা তথ্য এই বইতে পাবেন ...
তাই একজন নাগরিক হিসাবে এই তথ্য গুলো আপনার জানা প্রয়োজন ...।
বিসিএস ও ব্যাংক এর লিখিত পরীক্ষা ...+এছাড়া মাধ্যমিক ও উচ্চমাধ্যমিকের স্টুডেন্টদের জন্য অনেক কাজে আসবে ...
More Related Content
More from SaiKalyani11
MCQs of blackbody & Kirchhoff's law.pdf

It is a PowerPoint presentation on multiple choice questions and answers on Blackbody & Kirchhoff's law topics. It discusses all practical examples and numerical problems of the said concepts.
Planck quantum theory.pdf

This PowerPoint presentation explains Planck's quantum theory and Planck's constant topics briefly. It is now available on Instamojo for just Rs.10/-. Save a personalized copy of this presentation by visiting Instamojo. The link is below.
https://kameswariservices201831.myinstamojo.com/product/3439450/planck-quantum-theory-postulates
Kirchhoff's law (1).pdf

It is a PowerPoint presentation of Kirchhoff's law of thermal radiation. And it gives a brief idea of Kirchhoff's law, its applications, and examples. Moreover, it explains the terms absorptivity, emissive power, thermal equilibrium, and emissivity.
Kirchhoff's law.pdf

It discusses Kirchhoff's law and its examples. It explains blackbody emissions in thermal equilibrium conditions.
We are sorry to inform you about a small error in Kirchhoff's formula section of the above infographic. Kirchhoff's formula states that the absorptivity and emissivity of the body are equal in thermal equilibrium conditions. 'a' is the absorbing power of the body at a wavelength λ. And 'e' is the emissivity of the body at the same wavelength. So, we will write aλ=e but not aλ=eλ. We request you read it correctly while going through the PDF.
Kirchhoff's law formula and its derivation. Numerical problems.pdf

It discusses Kirchhoff's formula and its derivation. And it includes numerical problems of Kirchhoff's law. For a colorful e-book for only 10/- rupees on Kirchhoff's law, visit our store at;
https://kameswariservices201831.myinstamojo.com/product/3467221/kirchhoffs-law-of-thermal-radiations
Planck's constant.pdf

It describes the definition of Planck's constant. Planck constant helps compute the discrete energy changes of a body by relating to the frequency of the photon. Planck constant explains the proportionality relationship between the photon's energy and the frequency.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/08/plancks-constant-chemistry-learners.html
Numerical problems of Planck's quantum theory.pdf

It is a PowerPoint presentation on numerical problems of Planck quantum theory topic.
It includes 12 numerical problems with solved answers. Besides, this PowerPoint presentation has a mind map to remember all formulas of Planck quantum law.
MCQ on Planck constant.pdf

This document contains 10 multiple choice questions about Planck's constant. It provides explanations for why Planck's constant was introduced, its value in different units, and that it explains both the quantum and particle nature of light. The questions cover topics like the relationship between photon energy and frequency, experimental methods used to determine Planck's constant, and why LED lights are used in these calculations. Additional resources on Planck's constant and quantum theory are provided at the end.
Detailed light theories.pdf

It explains both wave & particle theories of light. It also includes Maxwell's mathematical equations and postulates of Planck quantum theory.
Theories of light.pdf

It explains the theories of light, including electromagnetic wave theory and Planck's quantum theory.
MCQ's on hydrogen spectrum.pdf

The document contains 10 multiple choice questions about the hydrogen spectrum. It discusses that the essential condition to observe the hydrogen spectrum is high temperature and low pressure. The hydrogen spectrum appears as distinct lines, with the prominent line being red with a wavelength of 656 nm. It is an example of a line spectrum and can be recorded using a spectroscope. Robert Bunsen and Gustav Kirchhoff discovered the hydrogen spectrum and spectroscope, while Isaac Newton first discovered the word "spectrum".
Rydberg equation.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
Some important points of Rydberg formula.pdf

Rydberg formula calculates the wavenumber of spectral lines obtained in the spectra of single electron hydrogen-like atoms. Rydberg's envision to measure the wavenumber of spectral energies instead of wavelengths had brought a distinguishing variation in the spectral evaluations.
The applications of spectral studies.pdf

It discusses the importance of spectral studies. The Rydberg-Ritz combination principle deals with the identification of unknown spectral lines of the atomic spectrum. Besides, spectral studies have much significance in the analysis of the composition of astronomical matter. The study of emission and absorption spectra of the substance provides information on its structure. In particular, astronomers use this spectroscopic data to determine the constituents of stars and interstellar matter.
Rydberg-Ritz combination principle.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
For more information on this topic, kindly visit our blog at;
https://jayamchemistrylearners.blogspot.com/2022/05/rydberg-ritz-combination-principle.html
Rydberg formula.pdf

The Rydberg formula helps to determine the wavenumber or wavelengths of hydrogen spectral lines obtained in the hydrogen spectrum. Previously, Johann Jakob Balmer discovered an empirical formula to determine the wavelengths of hydrogen spectral lines obtained in the visible region of the hydrogen spectrum. As we all know, the hydrogen spectrum is not limited to the visible zone only. It occupies the ultraviolet and infrared parts of the electromagnetic spectrum also. Hence, the scientists' quests to determine the spectral positions of various spectral lines of the hydrogen spectrum finally came to an end with the Rydberg formula.
For more information on this topic, kindly visit our blog article at;
https://jayamchemistrylearners.blogspot.com/2022/06/rydberg-formula-chemistry-learners.html
Comparison between the Rydberg formula and Balmer formula.pdf

In 1888, Rydberg put forward an empirical relation to calculate the wavelengths of spectral emission lines of the hydrogen spectral series. It is an empirical generalization of the Balmer formula. Here are the points of difference between the Rydberg formula and the Balmer formula.
Rydberg formula:
It determines the wavenumbers of spectral lines of all hydrogen spectral series.
It calculates the spectral energies in terms of wavenumbers.
Johannes Rydberg developed it.
It gave incorrect results for multi-electron systems due to the screening effect. So, it is only applicable to hydrogen and other single-electron atoms.
Balmer formula:
It determines the wavelengths of hydrogen spectral lines in the visible region only.
It calculates the spectral energies in terms of wavelengths.
Johann Jakob Balmer discovered it.
It is only applicable to the Balmer series of the hydrogen spectrum.
Tour plan.pdf

Go for free-cancellation accommodations in a central location between peak seasons to save money. Consider hostels and avoid peak weekends, when prices triple in big cities. Carefully choose accommodations based on your budget, travel style, comfort level, group size, and interests.
Wavelength & frequency relationship of an electromagnetic wave.pdf

1. The wavelength of an electromagnetic wave is the distance between identical points on adjacent waveforms, measured as the distance between crests or troughs for transverse waves and compressions or rarefactions for longitudinal waves.
2. The frequency is the number of waves that pass through a given point in a unit of time, inversely related to wavelength, and velocity equals wavelength multiplied by frequency.
3. Shorter waves have higher frequencies as wavelength and frequency are inversely proportional - when wavelength decreases, frequency increases.
Chemistry exam.pdf

The document provides tips for solving numerical chemistry problems in an upcoming exam. It advises to carefully read the question, identify the key information needed to solve it, recall the appropriate formula, and do the calculations to find the answer. The document also includes a link to a chemistry learning blog for additional help.
More from SaiKalyani11 (20)
Kirchhoff's law formula and its derivation. Numerical problems.pdf

Kirchhoff's law formula and its derivation. Numerical problems.pdf
Comparison between the Rydberg formula and Balmer formula.pdf

Comparison between the Rydberg formula and Balmer formula.pdf
Wavelength & frequency relationship of an electromagnetic wave.pdf

Wavelength & frequency relationship of an electromagnetic wave.pdf
Recently uploaded
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশের অর্থনৈতিক সমীক্ষা ২০২৪ [Bangladesh Economic Review 2024 Bangla.pdf] কম্পিউটার , ট্যাব ও স্মার্ট ফোন ভার্সন সহ সম্পূর্ণ বাংলা ই-বুক বা pdf বই " সুচিপত্র ...বুকমার্ক মেনু 🔖 ও হাইপার লিংক মেনু 📝👆 যুক্ত ..
আমাদের সবার জন্য খুব খুব গুরুত্বপূর্ণ একটি বই ..বিসিএস, ব্যাংক, ইউনিভার্সিটি ভর্তি ও যে কোন প্রতিযোগিতা মূলক পরীক্ষার জন্য এর খুব ইম্পরট্যান্ট একটি বিষয় ...তাছাড়া বাংলাদেশের সাম্প্রতিক যে কোন ডাটা বা তথ্য এই বইতে পাবেন ...
তাই একজন নাগরিক হিসাবে এই তথ্য গুলো আপনার জানা প্রয়োজন ...।
বিসিএস ও ব্যাংক এর লিখিত পরীক্ষা ...+এছাড়া মাধ্যমিক ও উচ্চমাধ্যমিকের স্টুডেন্টদের জন্য অনেক কাজে আসবে ...
How to Build a Module in Odoo 17 Using the Scaffold Method

Odoo provides an option for creating a module by using a single line command. By using this command the user can make a whole structure of a module. It is very easy for a beginner to make a module. There is no need to make each file manually. This slide will show how to create a module using the scaffold method.
Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...

Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...Dr. Vinod Kumar Kanvaria
Exploiting Artificial Intelligence for Empowering Researchers and Faculty,
International FDP on Fundamentals of Research in Social Sciences
at Integral University, Lucknow, 06.06.2024
By Dr. Vinod Kumar KanvariaSouth African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)Academy of Science of South Africa
A workshop hosted by the South African Journal of Science aimed at postgraduate students and early career researchers with little or no experience in writing and publishing journal articles.Top five deadliest dog breeds in America

Thinking of getting a dog? Be aware that breeds like Pit Bulls, Rottweilers, and German Shepherds can be loyal and dangerous. Proper training and socialization are crucial to preventing aggressive behaviors. Ensure safety by understanding their needs and always supervising interactions. Stay safe, and enjoy your furry friends!
How to Manage Your Lost Opportunities in Odoo 17 CRM

Odoo 17 CRM allows us to track why we lose sales opportunities with "Lost Reasons." This helps analyze our sales process and identify areas for improvement. Here's how to configure lost reasons in Odoo 17 CRM
How to Add Chatter in the odoo 17 ERP Module

In Odoo, the chatter is like a chat tool that helps you work together on records. You can leave notes and track things, making it easier to talk with your team and partners. Inside chatter, all communication history, activity, and changes will be displayed.
PCOS corelations and management through Ayurveda.

This presentation includes basic of PCOS their pathology and treatment and also Ayurveda correlation of PCOS and Ayurvedic line of treatment mentioned in classics.
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
clinical examination of hip joint (1).pdf

described clinical examination all orthopeadic conditions .
Main Java[All of the Base Concepts}.docx

This is part 1 of my Java Learning Journey. This Contains Custom methods, classes, constructors, packages, multithreading , try- catch block, finally block and more.
A Strategic Approach: GenAI in Education

Artificial Intelligence (AI) technologies such as Generative AI, Image Generators and Large Language Models have had a dramatic impact on teaching, learning and assessment over the past 18 months. The most immediate threat AI posed was to Academic Integrity with Higher Education Institutes (HEIs) focusing their efforts on combating the use of GenAI in assessment. Guidelines were developed for staff and students, policies put in place too. Innovative educators have forged paths in the use of Generative AI for teaching, learning and assessments leading to pockets of transformation springing up across HEIs, often with little or no top-down guidance, support or direction.
This Gasta posits a strategic approach to integrating AI into HEIs to prepare staff, students and the curriculum for an evolving world and workplace. We will highlight the advantages of working with these technologies beyond the realm of teaching, learning and assessment by considering prompt engineering skills, industry impact, curriculum changes, and the need for staff upskilling. In contrast, not engaging strategically with Generative AI poses risks, including falling behind peers, missed opportunities and failing to ensure our graduates remain employable. The rapid evolution of AI technologies necessitates a proactive and strategic approach if we are to remain relevant.
The Diamonds of 2023-2024 in the IGRA collection

A review of the growth of the Israel Genealogy Research Association Database Collection for the last 12 months. Our collection is now passed the 3 million mark and still growing. See which archives have contributed the most. See the different types of records we have, and which years have had records added. You can also see what we have for the future.
Advanced Java[Extra Concepts, Not Difficult].docx![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Advanced Java[Extra Concepts, Not Difficult].docx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
This is part 2 of my Java Learning Journey. This contains Hashing, ArrayList, LinkedList, Date and Time Classes, Calendar Class and more.
Recently uploaded (20)
Digital Artefact 1 - Tiny Home Environmental Design

Digital Artefact 1 - Tiny Home Environmental Design
বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf

বাংলাদেশ অর্থনৈতিক সমীক্ষা (Economic Review) ২০২৪ UJS App.pdf
How to Build a Module in Odoo 17 Using the Scaffold Method

How to Build a Module in Odoo 17 Using the Scaffold Method
Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...

Exploiting Artificial Intelligence for Empowering Researchers and Faculty, In...
South African Journal of Science: Writing with integrity workshop (2024)

South African Journal of Science: Writing with integrity workshop (2024)
How to Manage Your Lost Opportunities in Odoo 17 CRM

How to Manage Your Lost Opportunities in Odoo 17 CRM
Azure Interview Questions and Answers PDF By ScholarHat

Azure Interview Questions and Answers PDF By ScholarHat
Liberal Approach to the Study of Indian Politics.pdf

Liberal Approach to the Study of Indian Politics.pdf
Film vocab for eal 3 students: Australia the movie

Film vocab for eal 3 students: Australia the movie
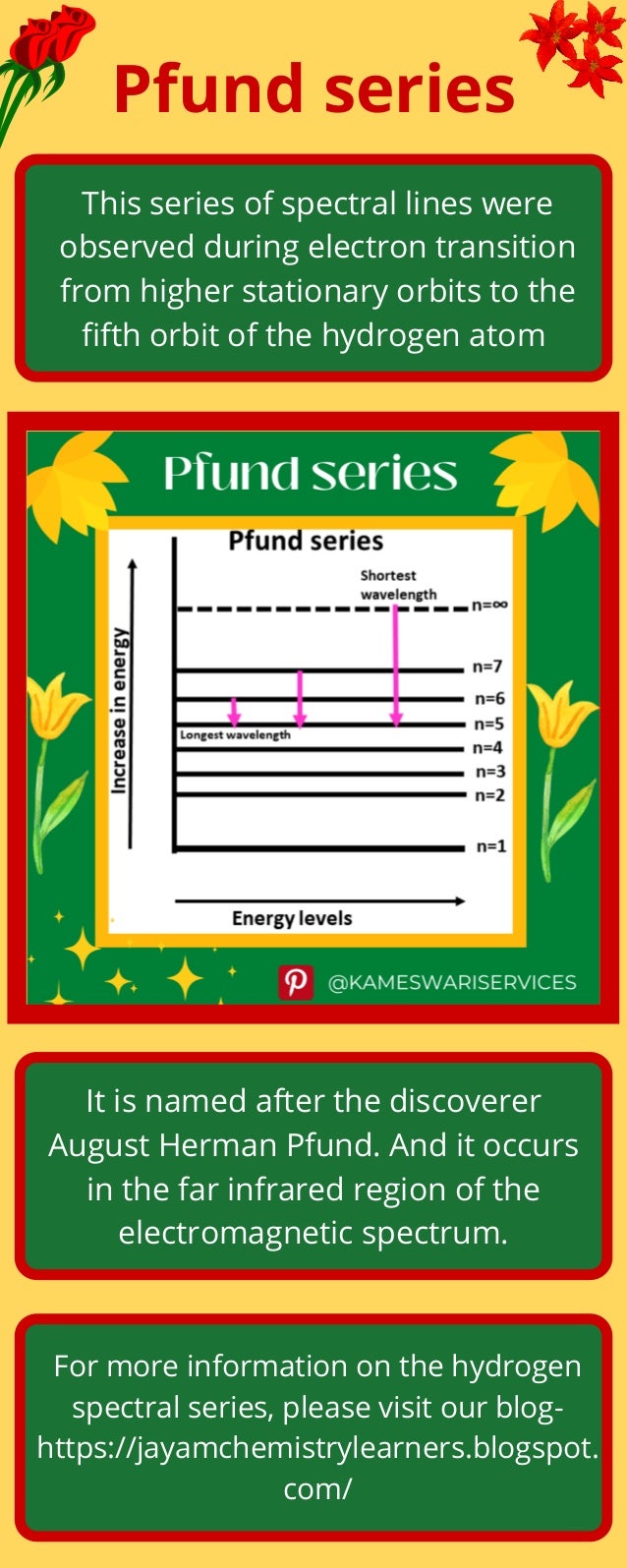
What is Pfund series of the hydrogen spectrum?
- 1. Pfund series This series of spectral lines were observed during electron transition from higher stationary orbits to the fifth orbit of the hydrogen atom It is named after the discoverer August Herman Pfund. And it occurs in the far infrared region of the electromagnetic spectrum. For more information on the hydrogen spectral series, please visit our blog- https://jayamchemistrylearners.blogspot. com/