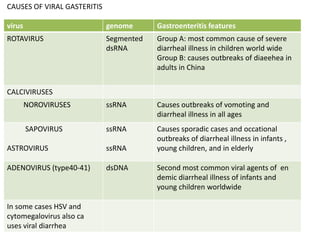
This document provides information on viral gastroenteritis. It discusses several viruses that can cause acute gastroenteritis in humans, with rotavirus and norovirus being the most common. Rotavirus is the leading cause of severe diarrhea in infants and children worldwide. It outlines the symptoms of viral gastroenteritis as vomiting, watery diarrhea, nausea, fever, abdominal pain, and headache. Dehydration is a potential complication if left untreated. The document also discusses transmission, pathogenesis, diagnosis and treatment of rotavirus and norovirus infections.













![Rotavirus Vaccines
• RV5 (RotaTeq) contains five reassortant
rotaviruses developed from human and bovine
parent rotavirus strains
• vaccine viruses suspended in a buffer solution
• contains no preservatives or thimerosal
• RV1 (Rotarix) contains one strain of live
attenuated human rotavirus (type G1PA[8])
• provided as a lyophilized powder that is
reconstituted before administration
• contains no preservatives or thimerosal](https://image.slidesharecdn.com/viralgastroenteritis-240229041030-775d01bd/85/VIRAL-GASTROENTERITIS-pptx-for-educational-16-320.jpg)














