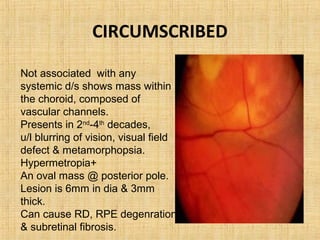
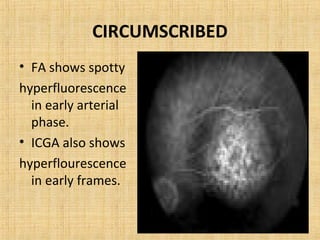
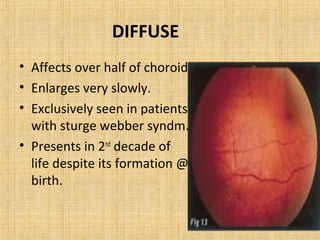
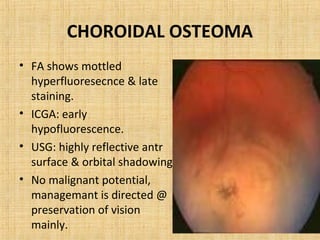
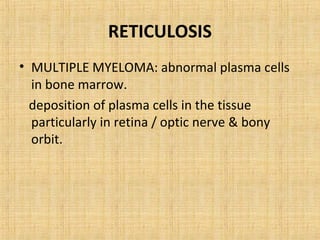
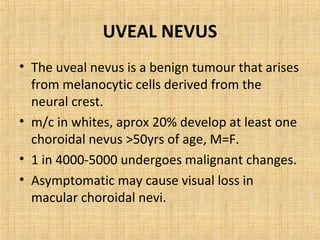
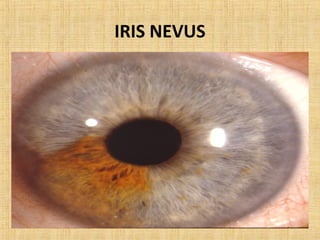
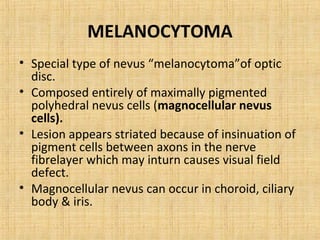
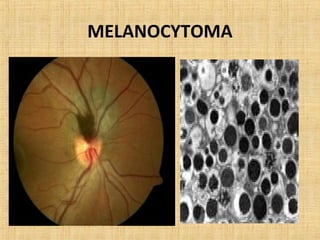
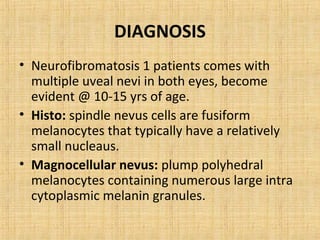
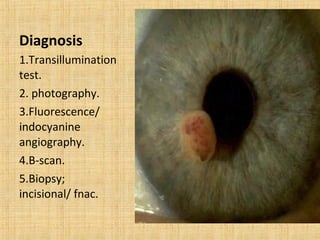
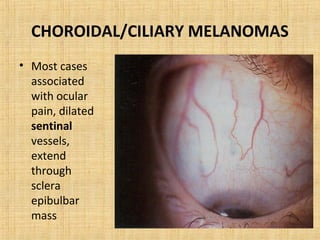
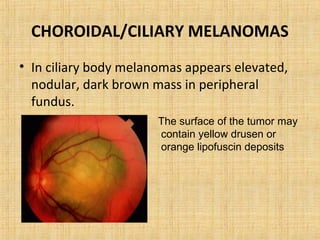
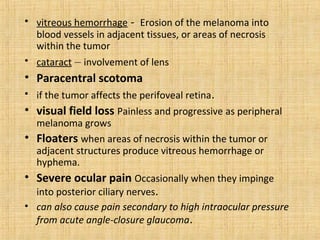
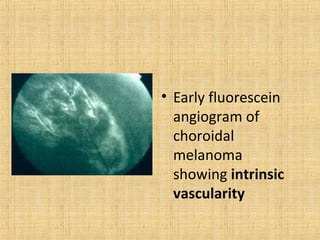
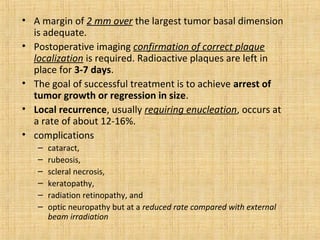
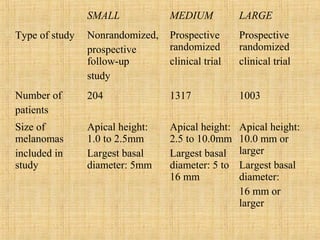
This document discusses various types of uveal tumors including:
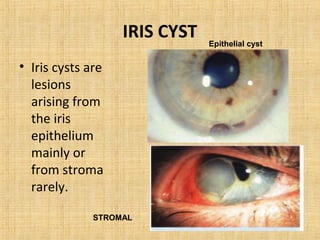
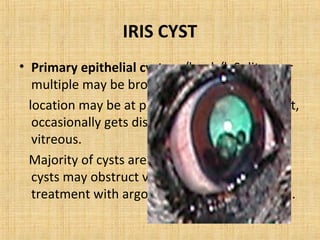
1. Epithelial tumors such as epithelial hyperplasia, benign epithelioma, and malignant epithelioma including dictyoma and malignant ciliary epithelioma.
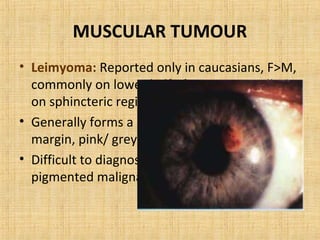
2. Muscular tumors like leiomyoma and leiomyosarcoma.
3. Vascular tumors including hemangioma.
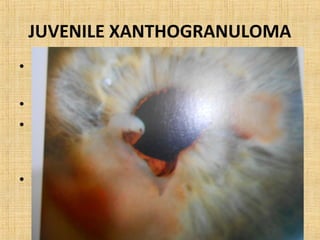
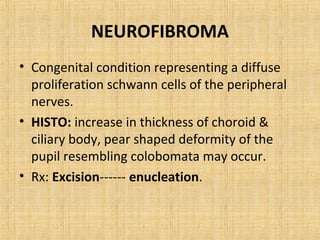
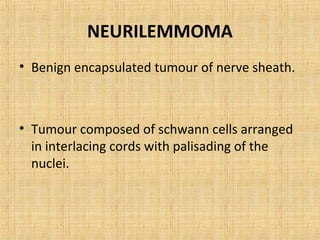
4. Neuroectodermal tumors such as schwannian tumors including neurofibroma and neurilemmoma.
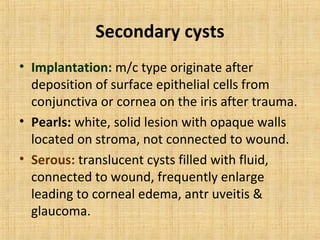
5. Secondary tumors that can spread to the uvea from other sites like the breast, lung, or skin.
It provides details on the classification, presentation, histopathology