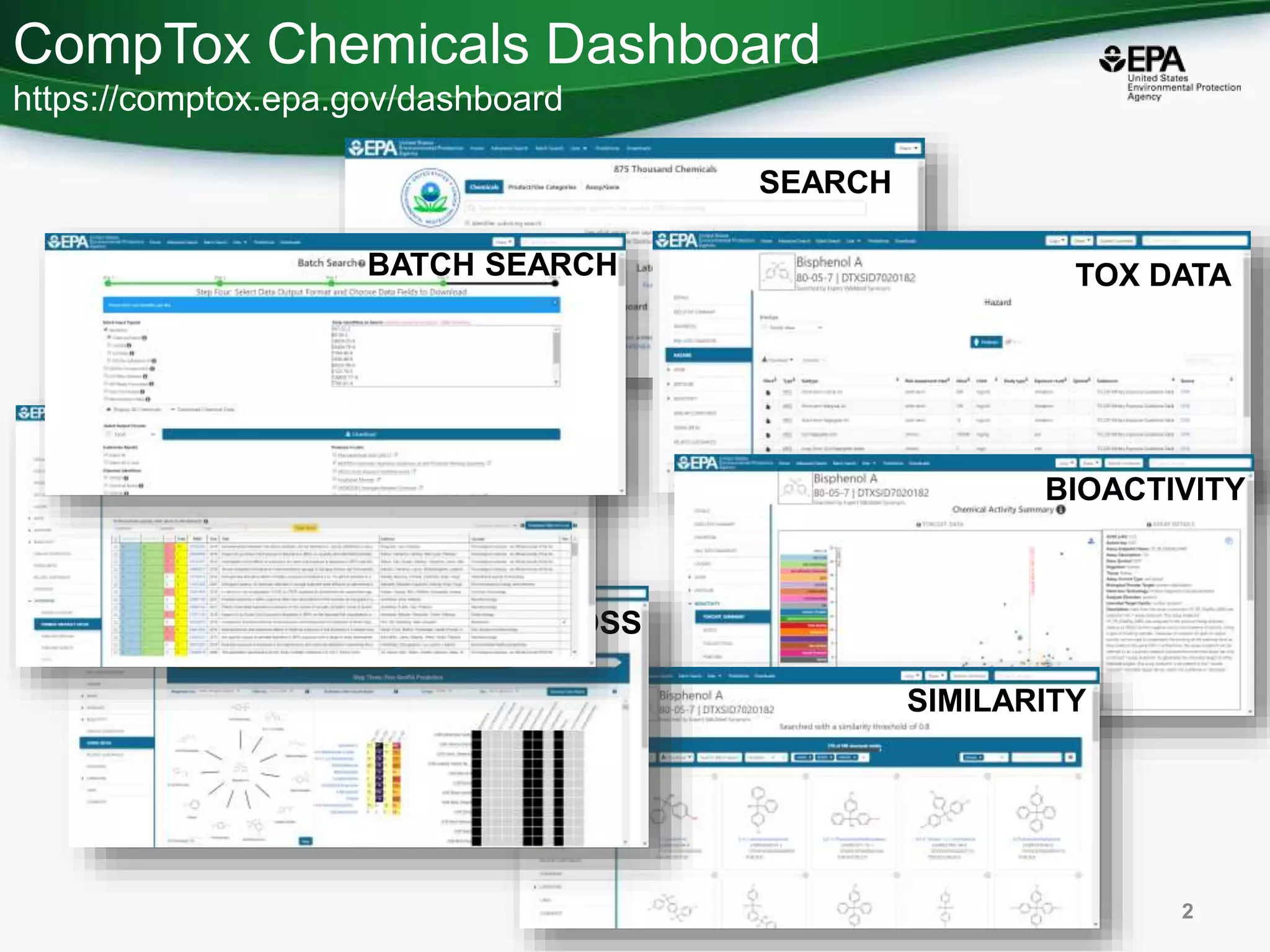
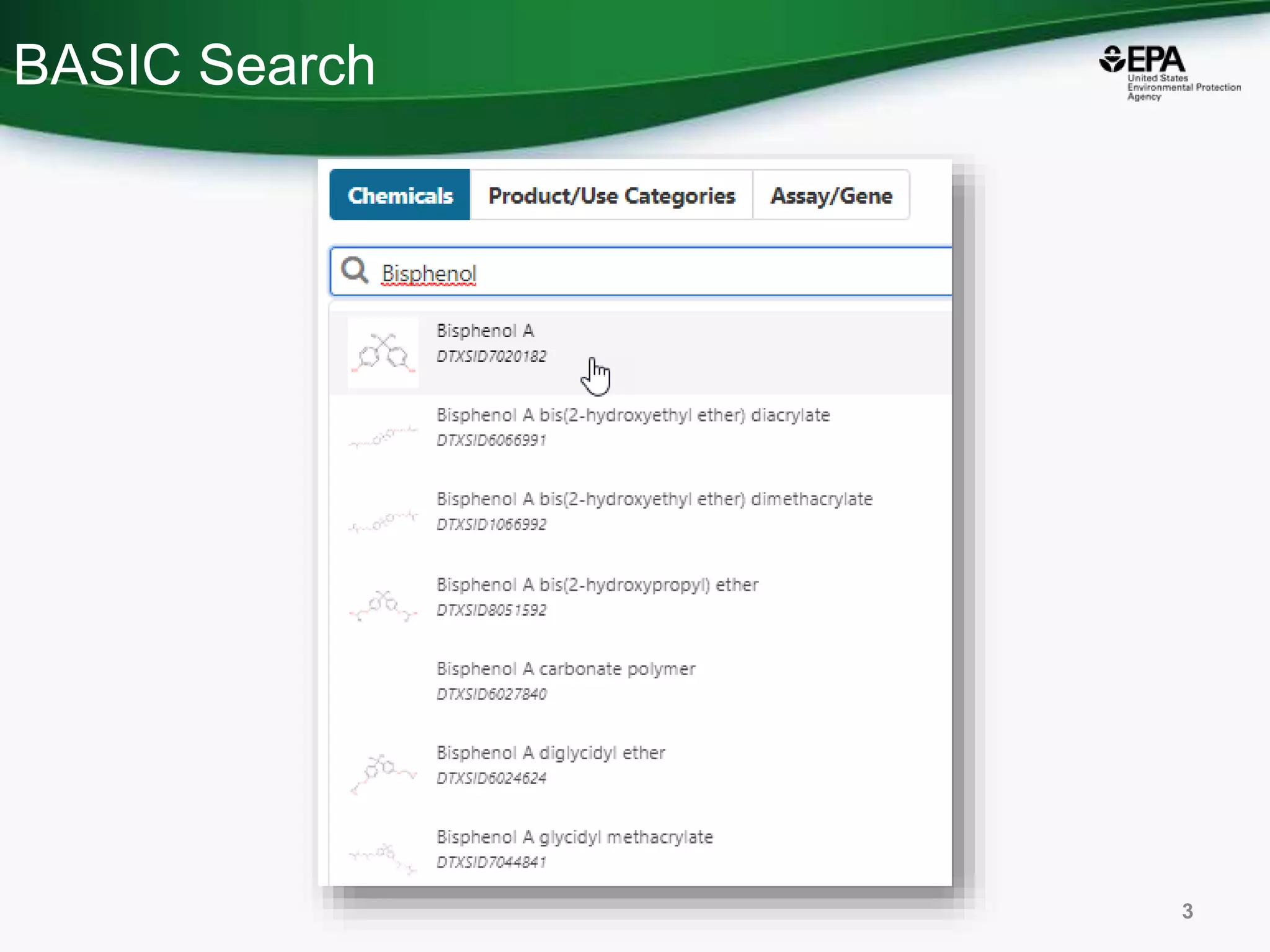
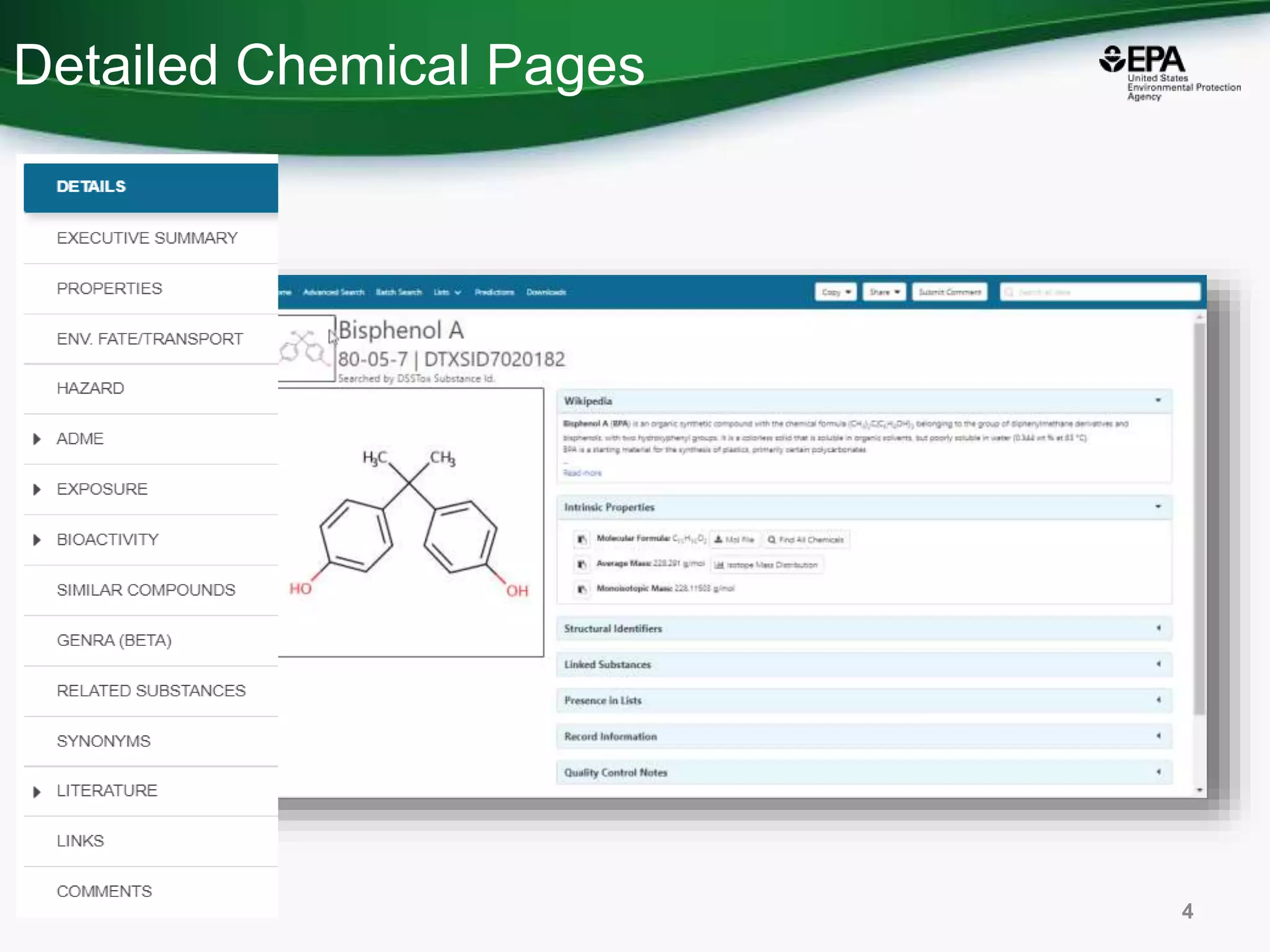
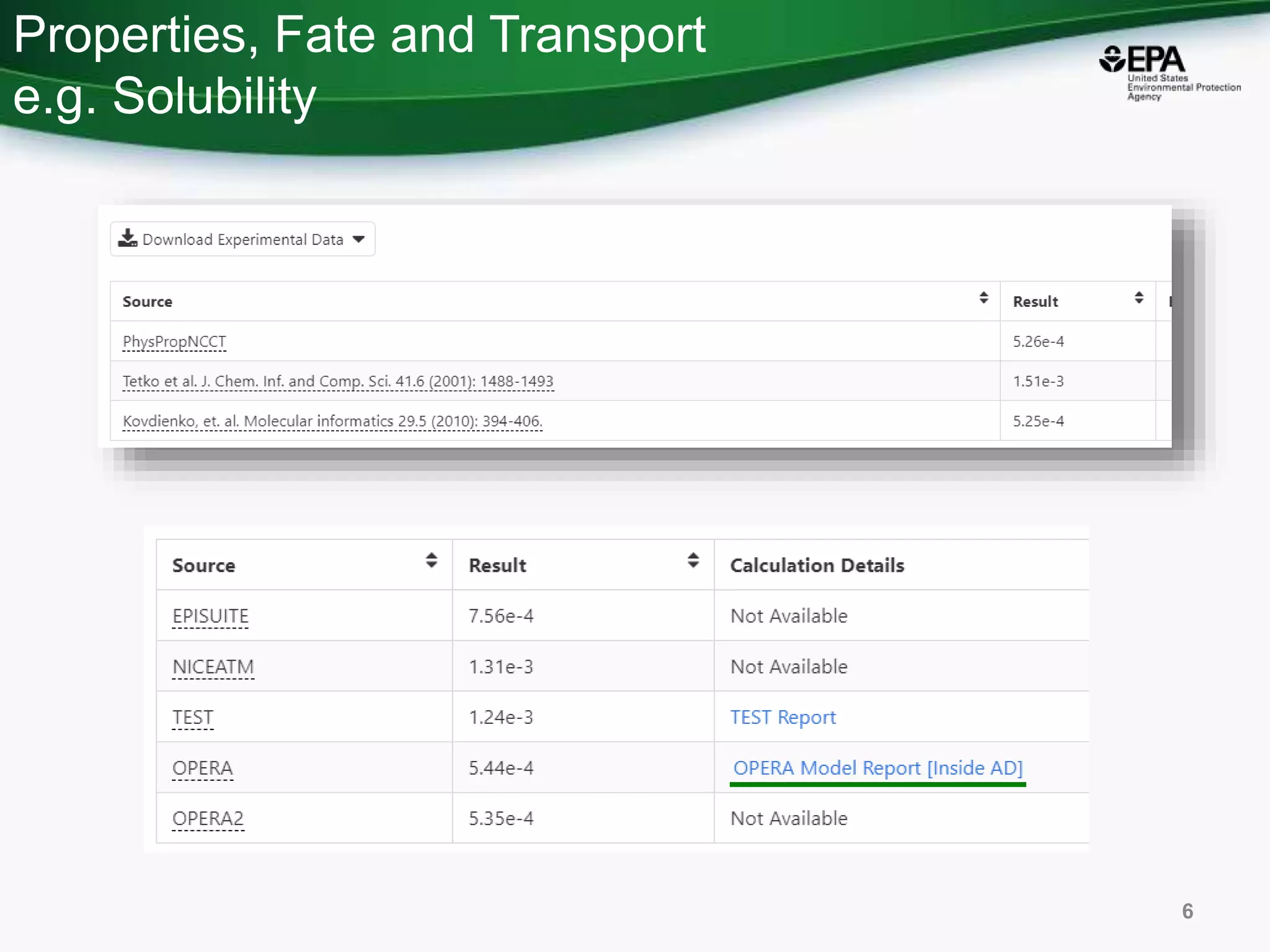
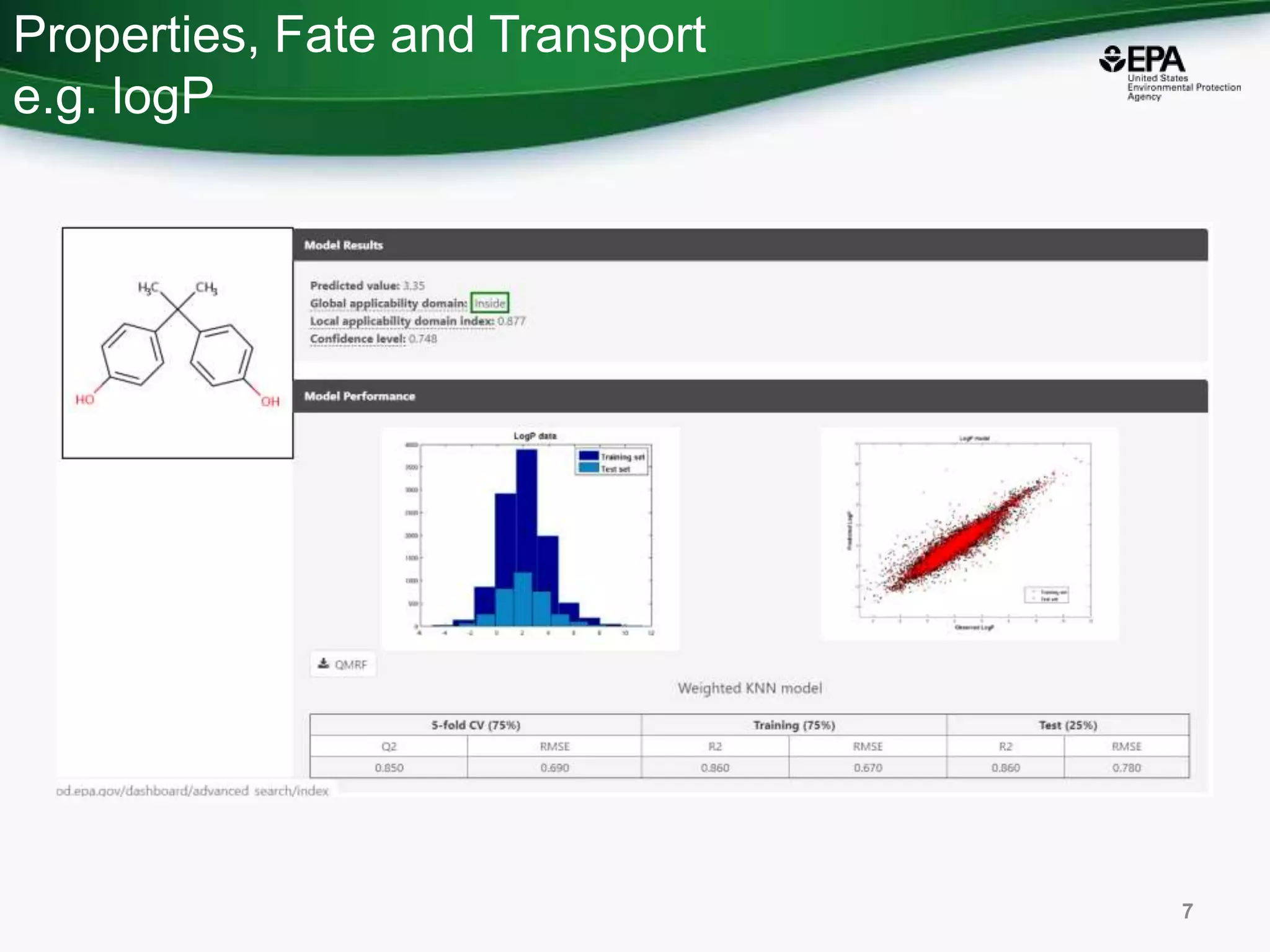
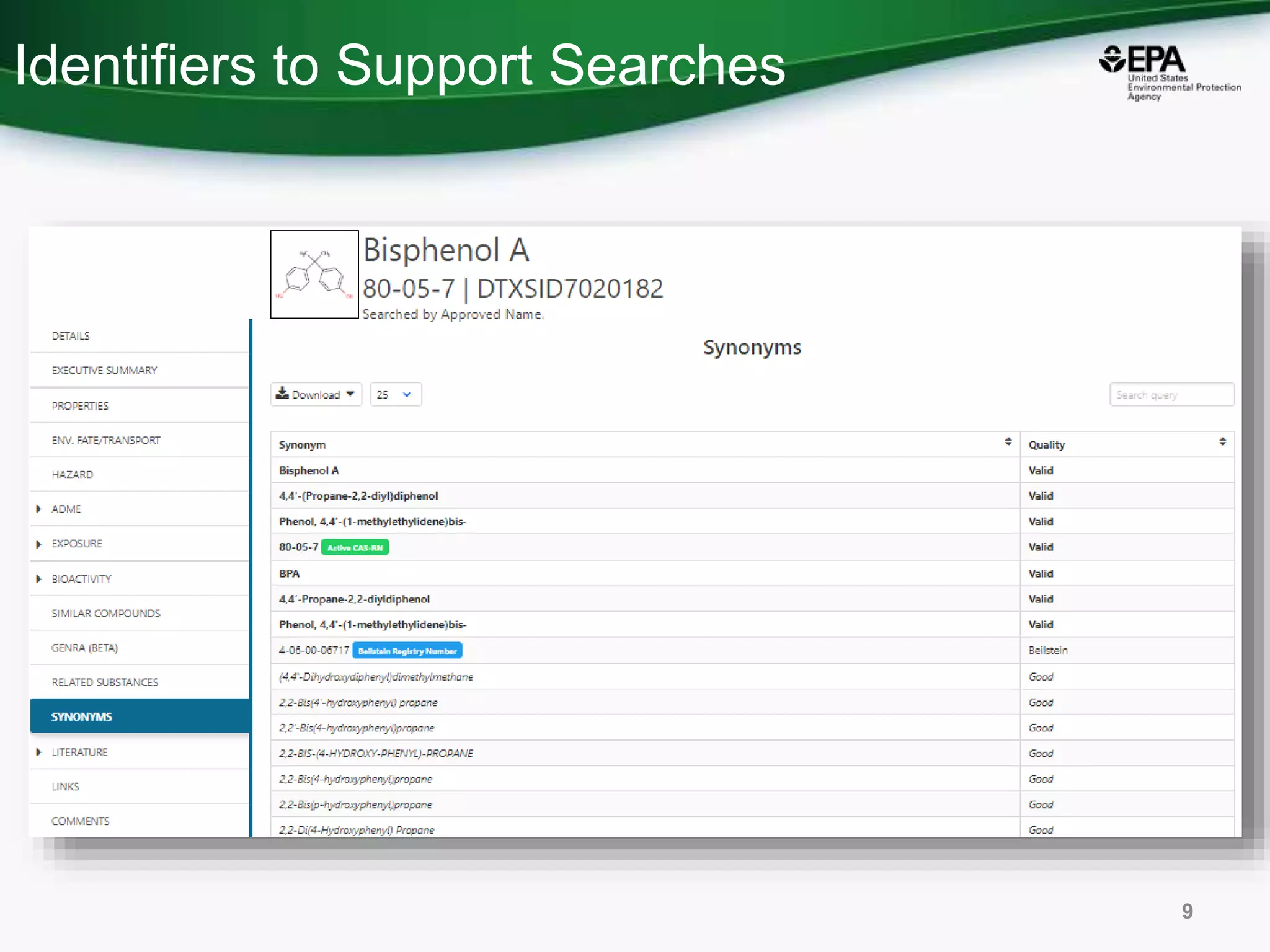
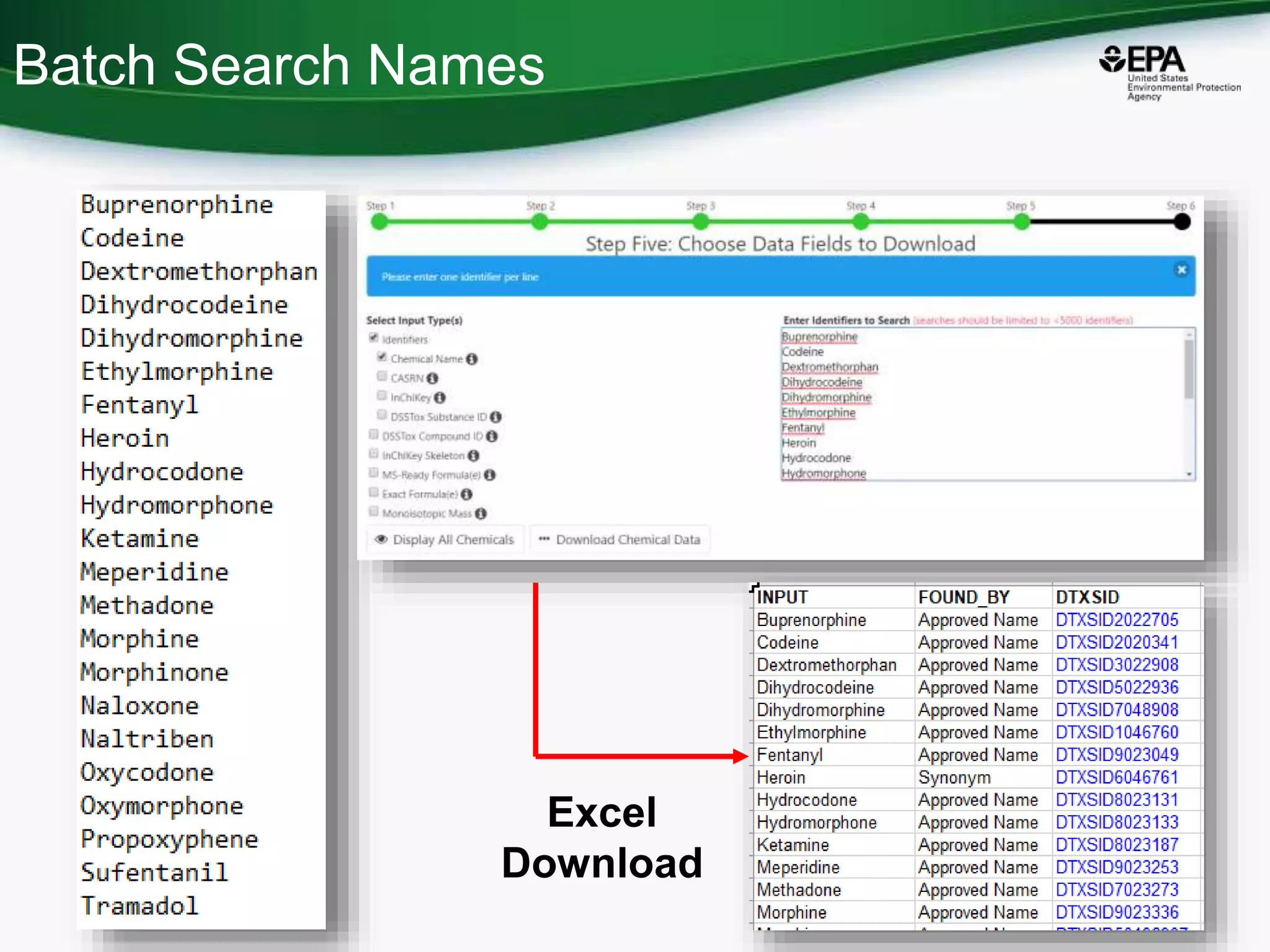
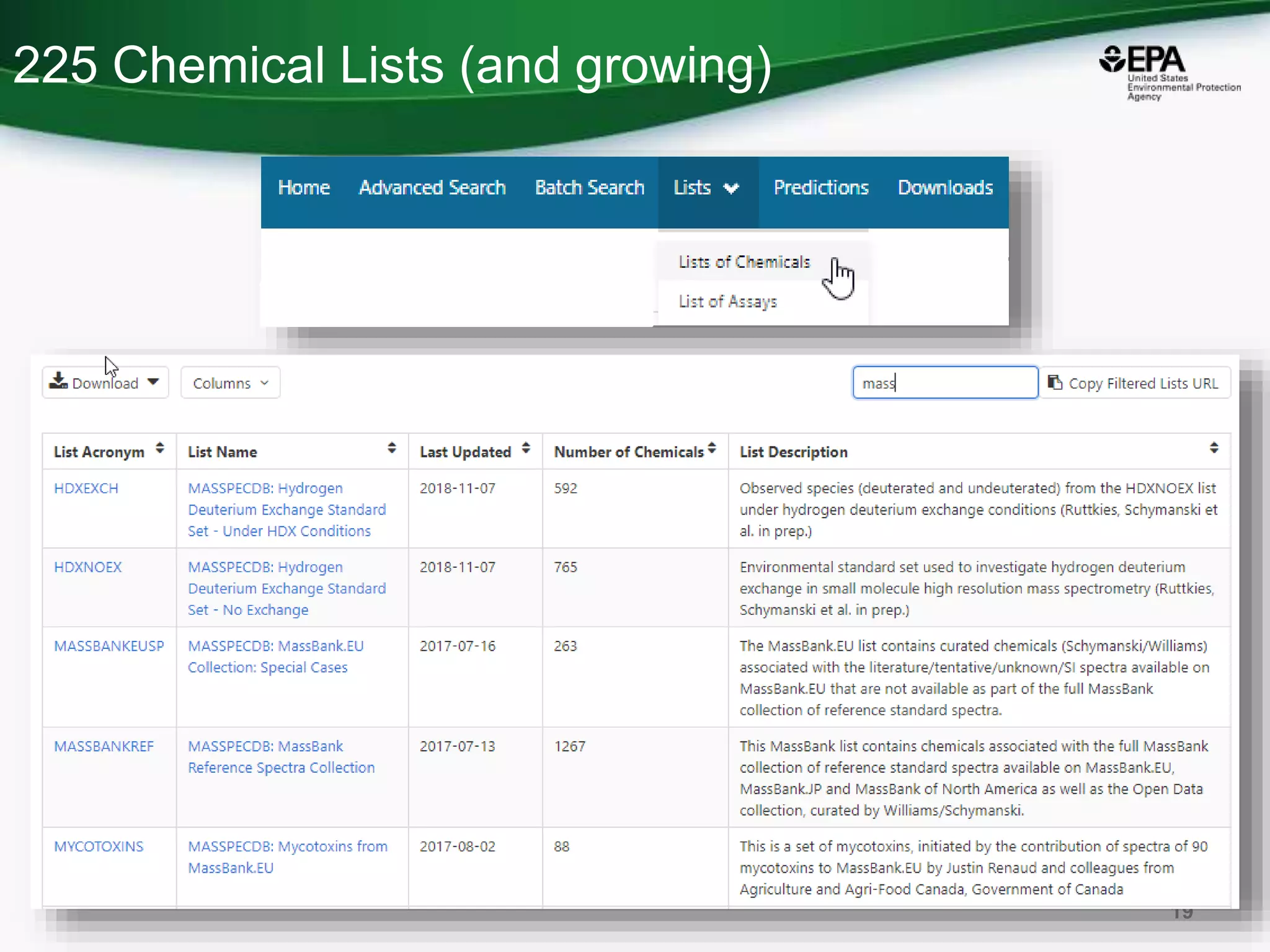
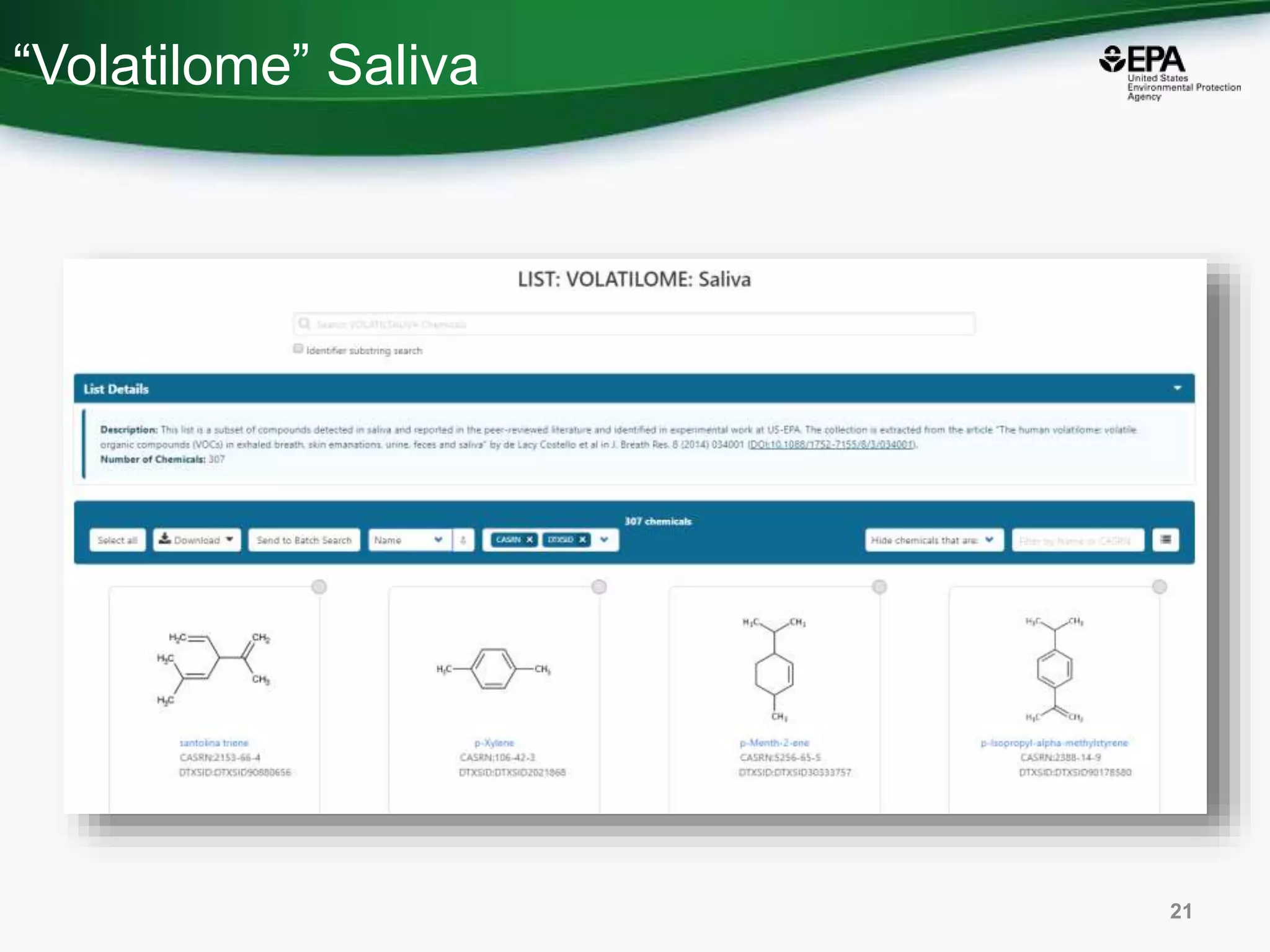
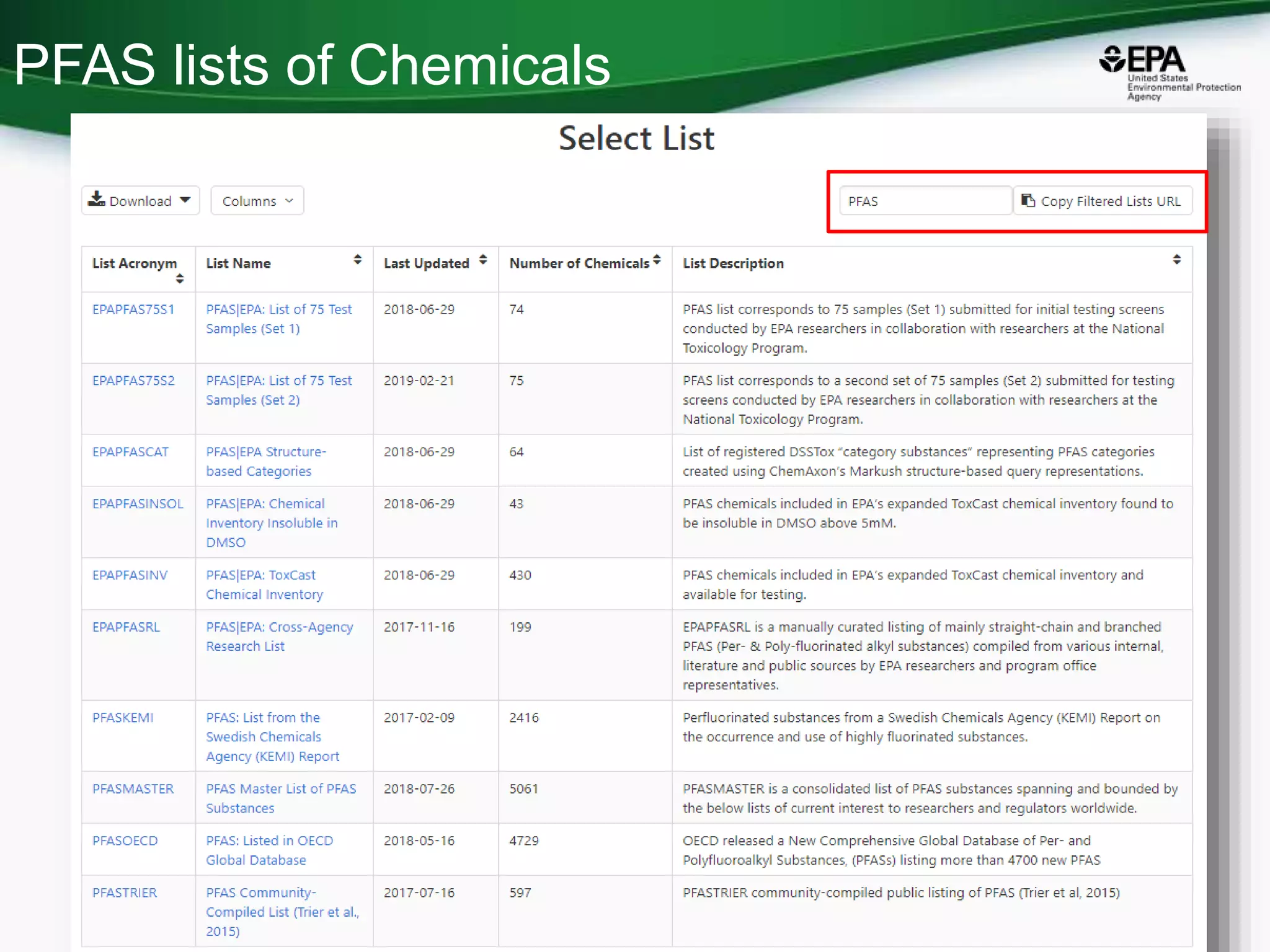
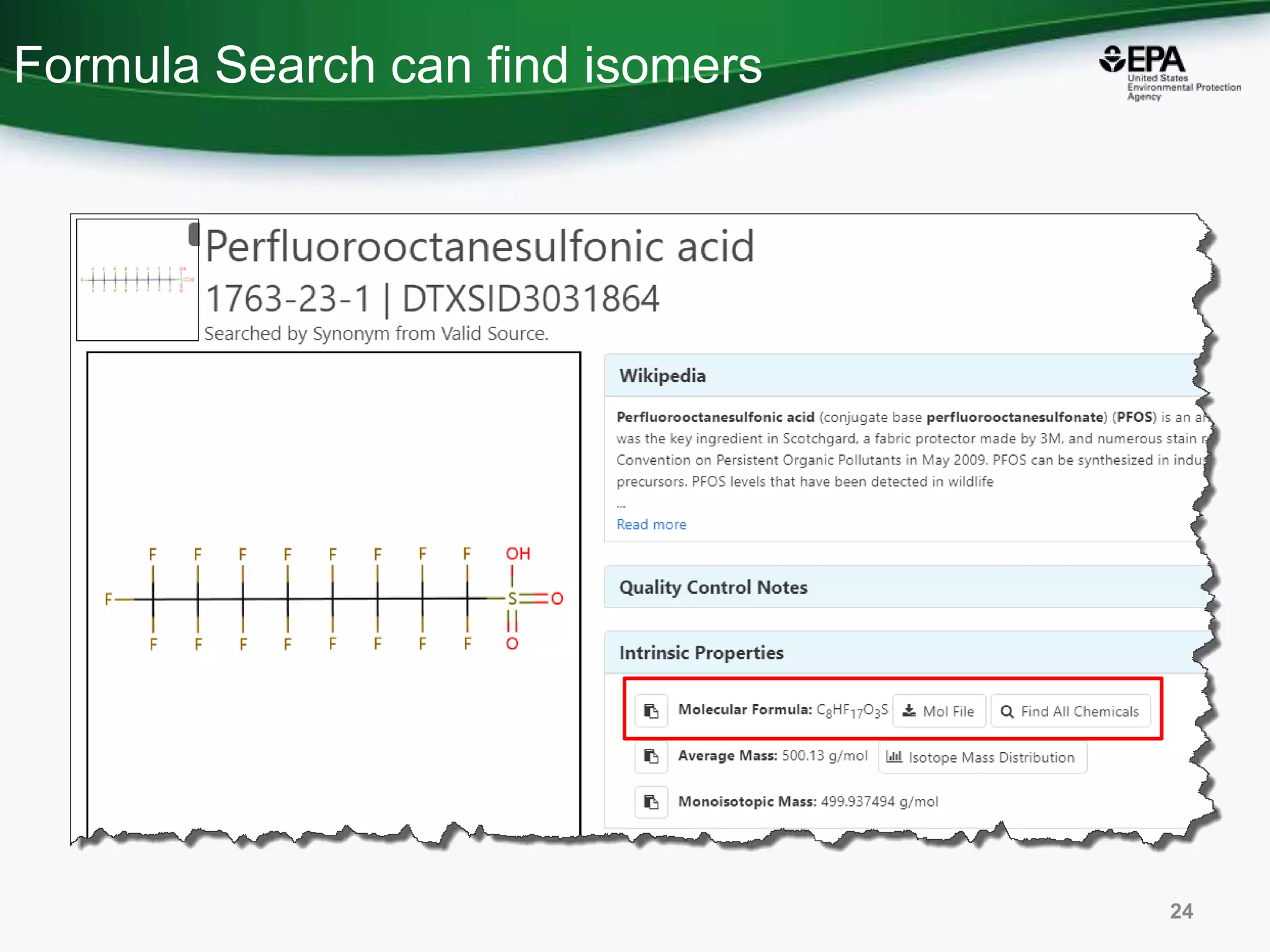
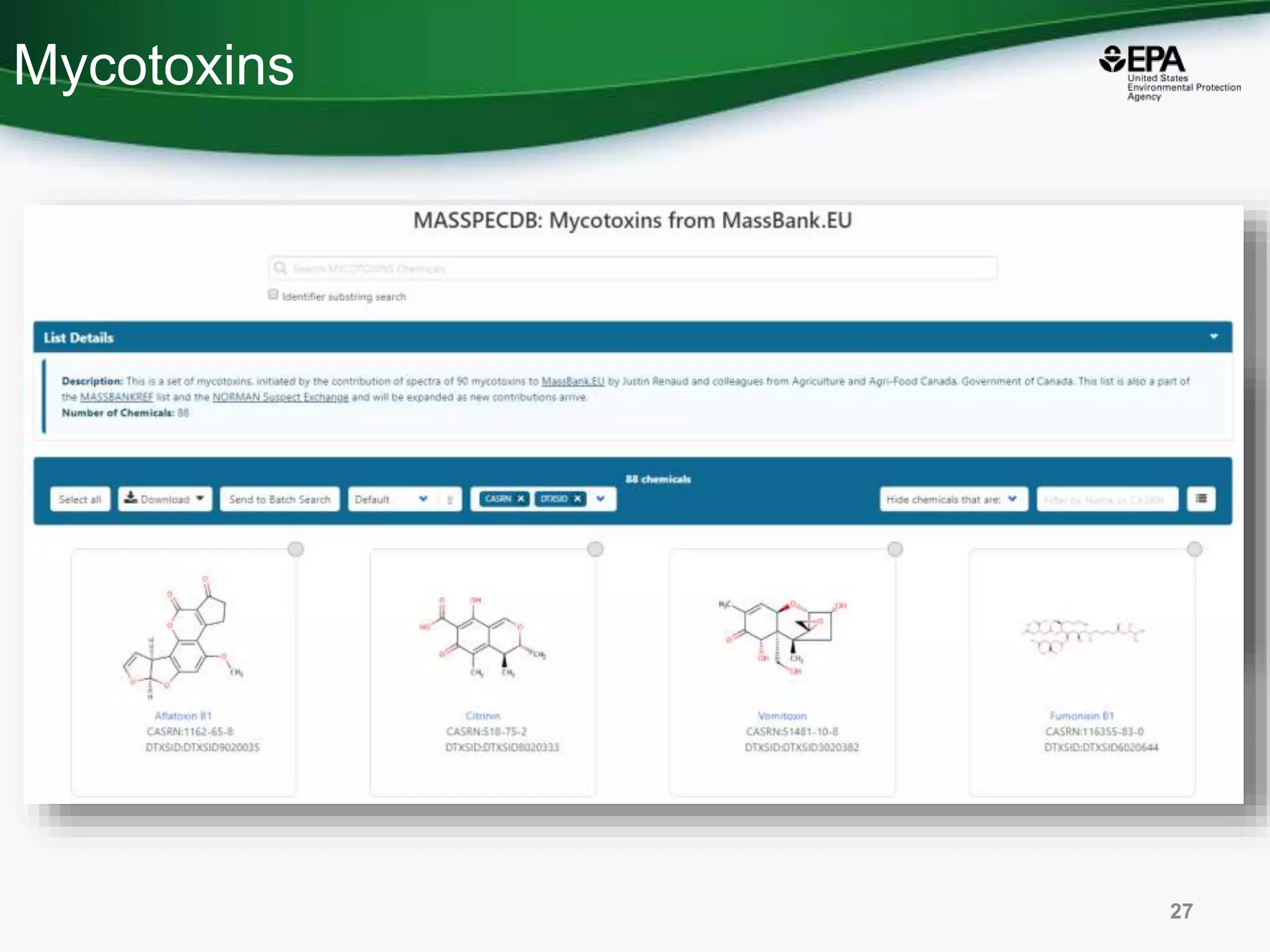
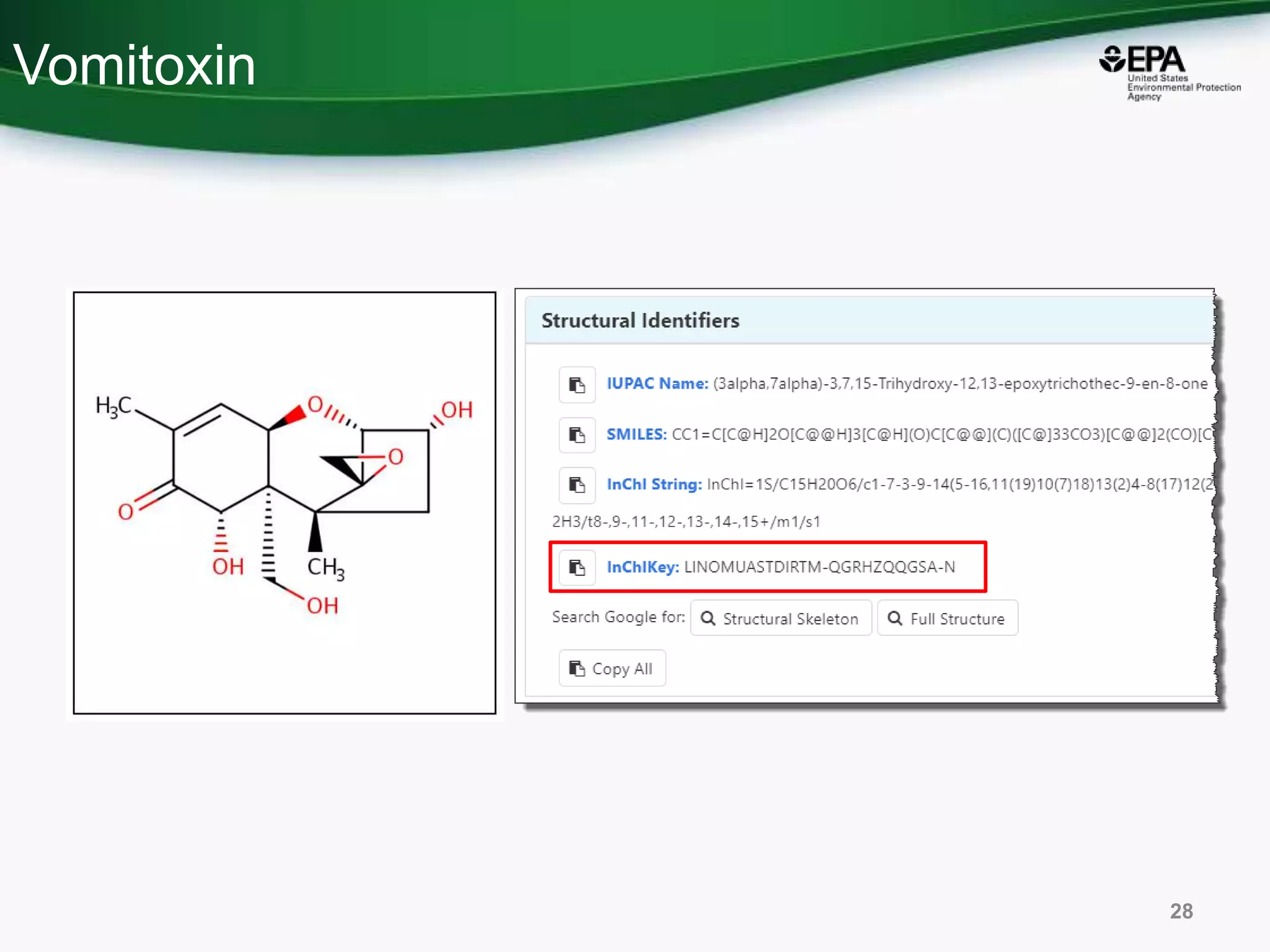
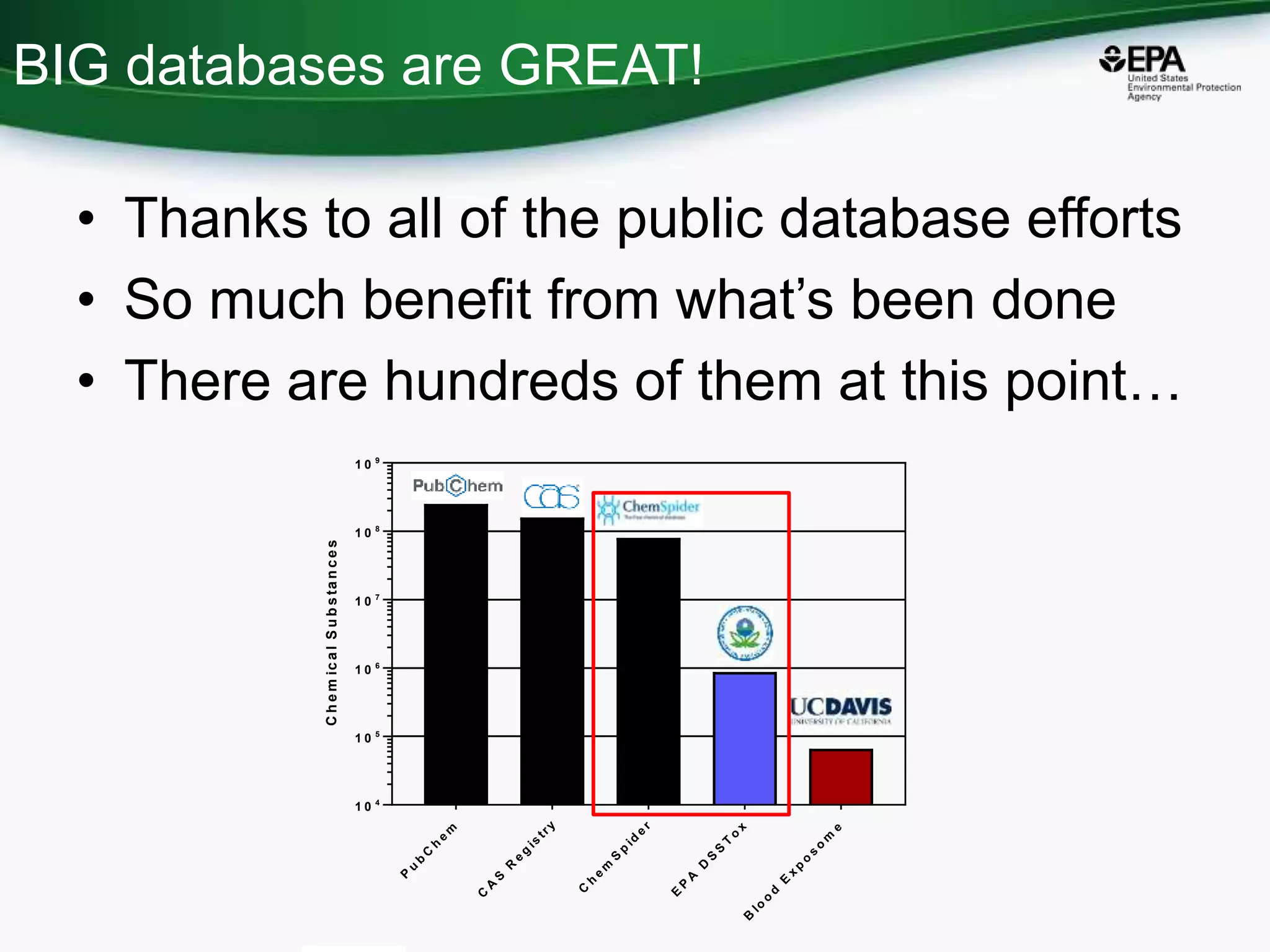
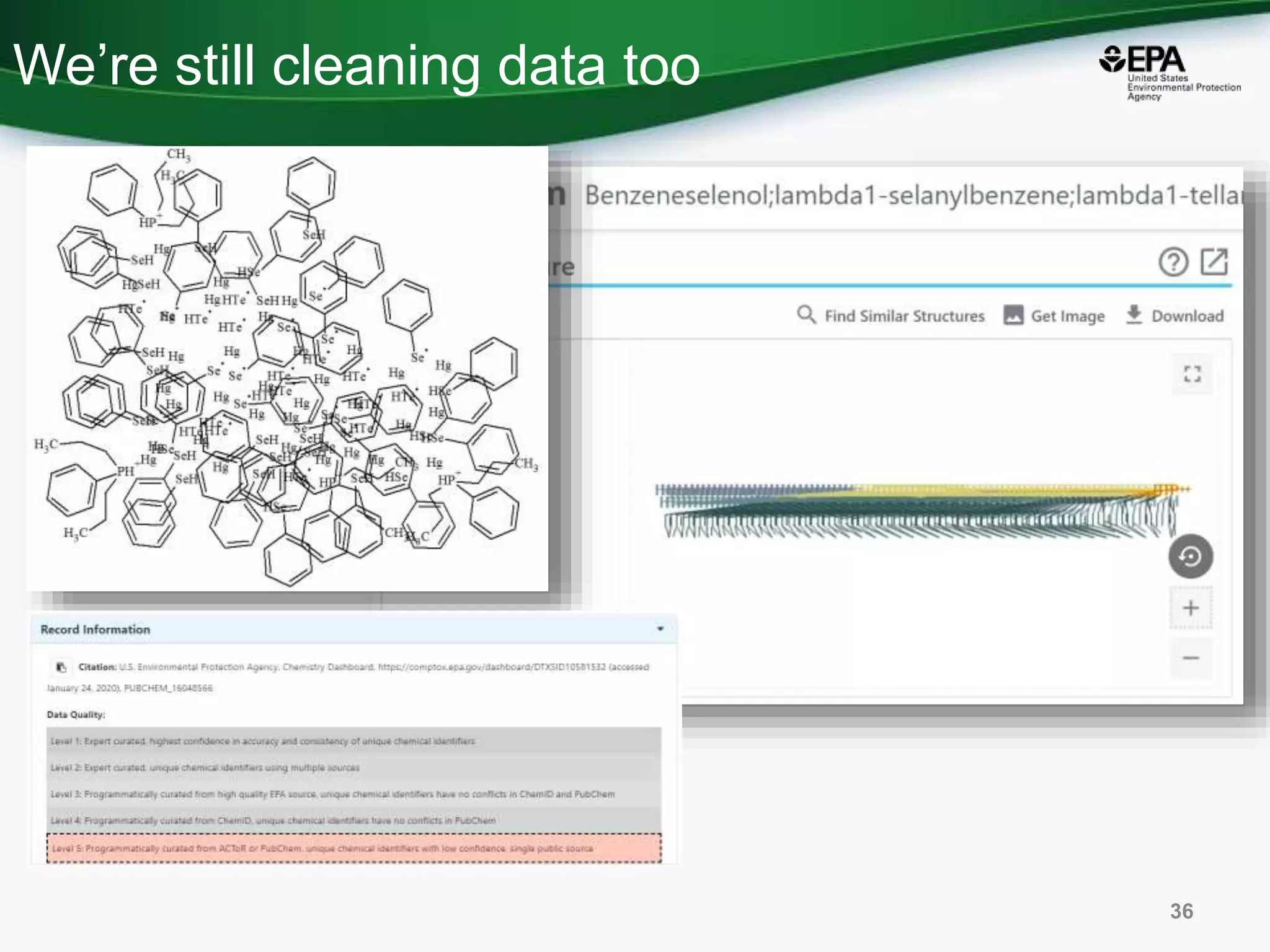
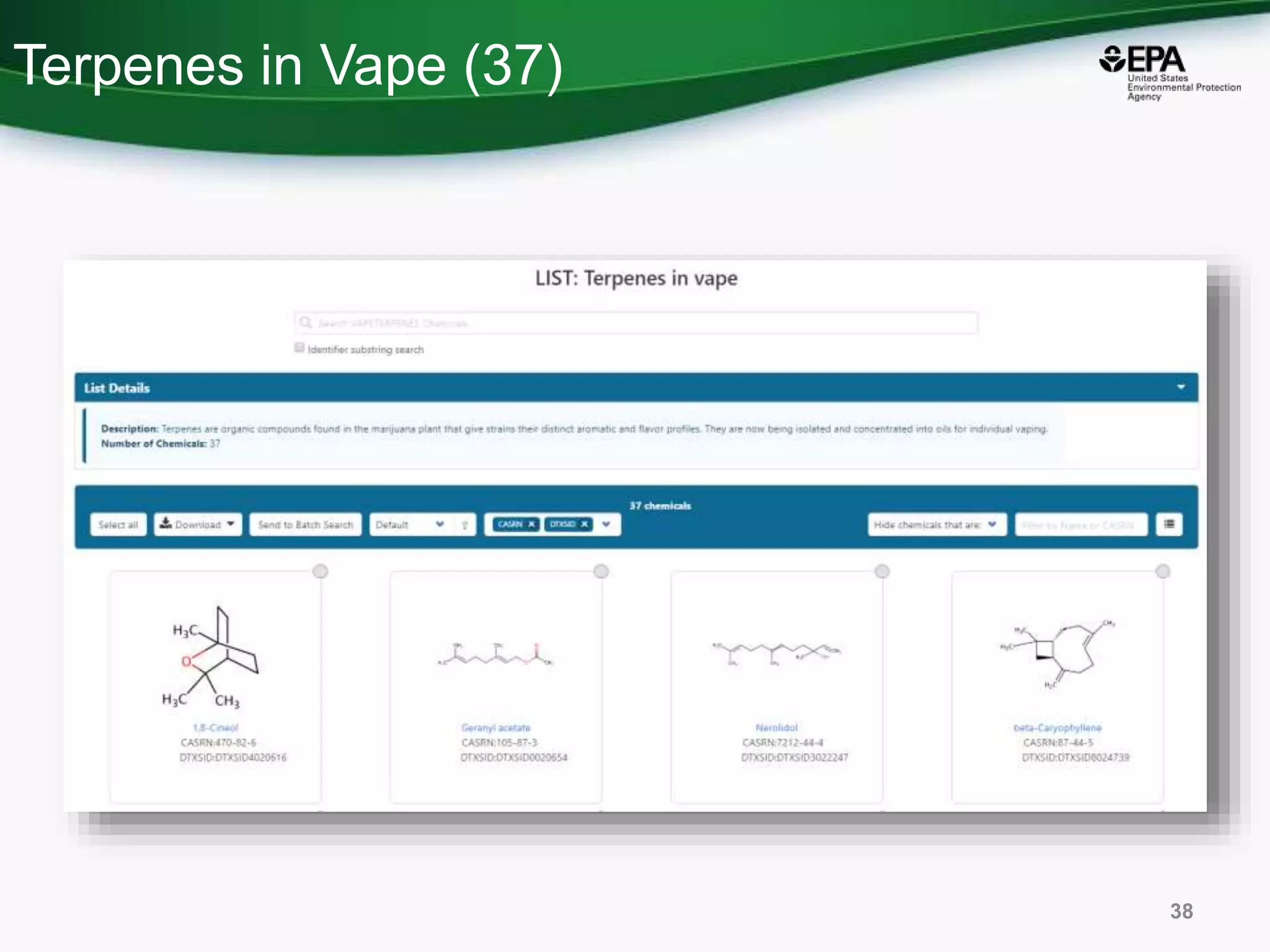
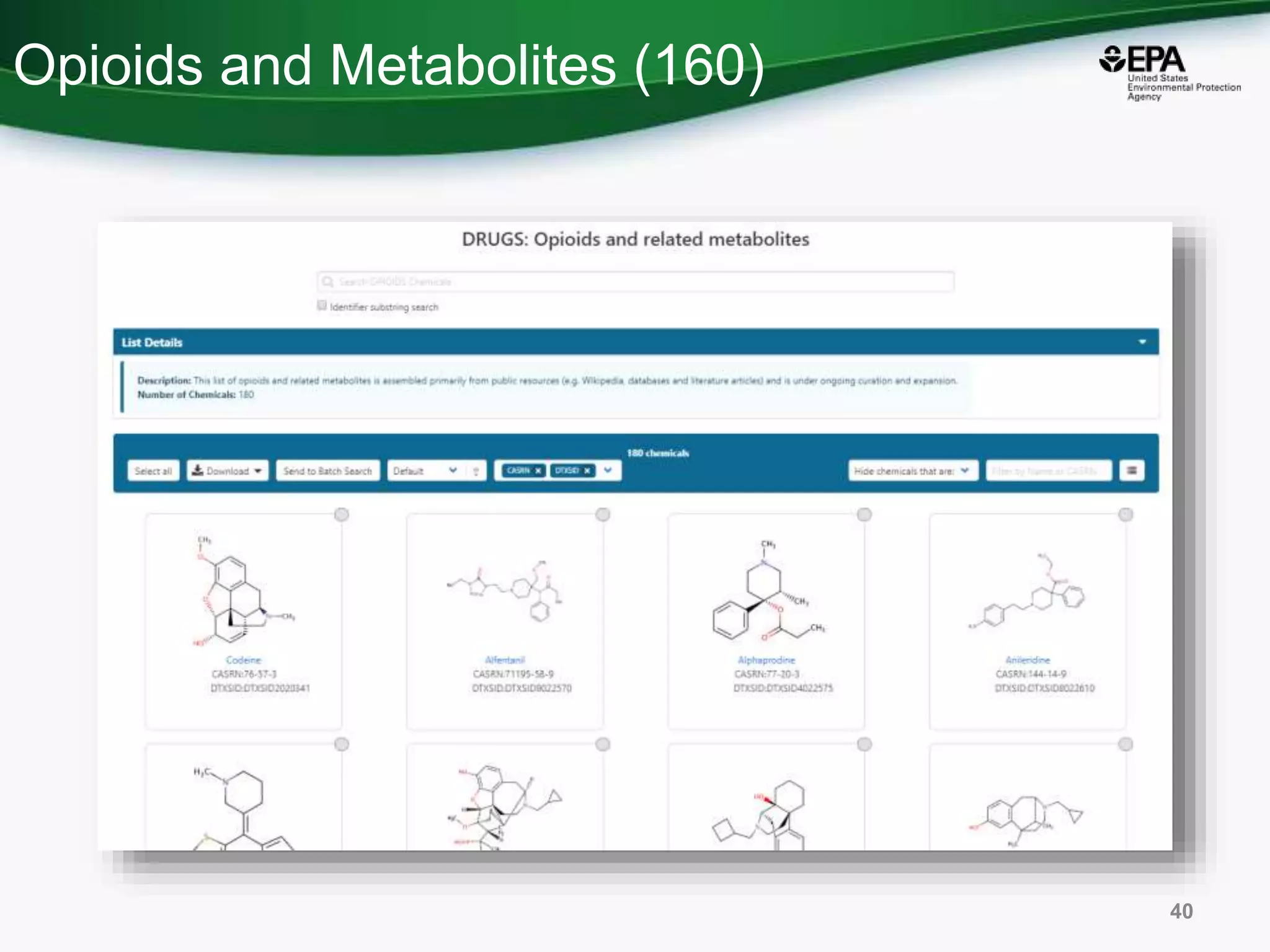
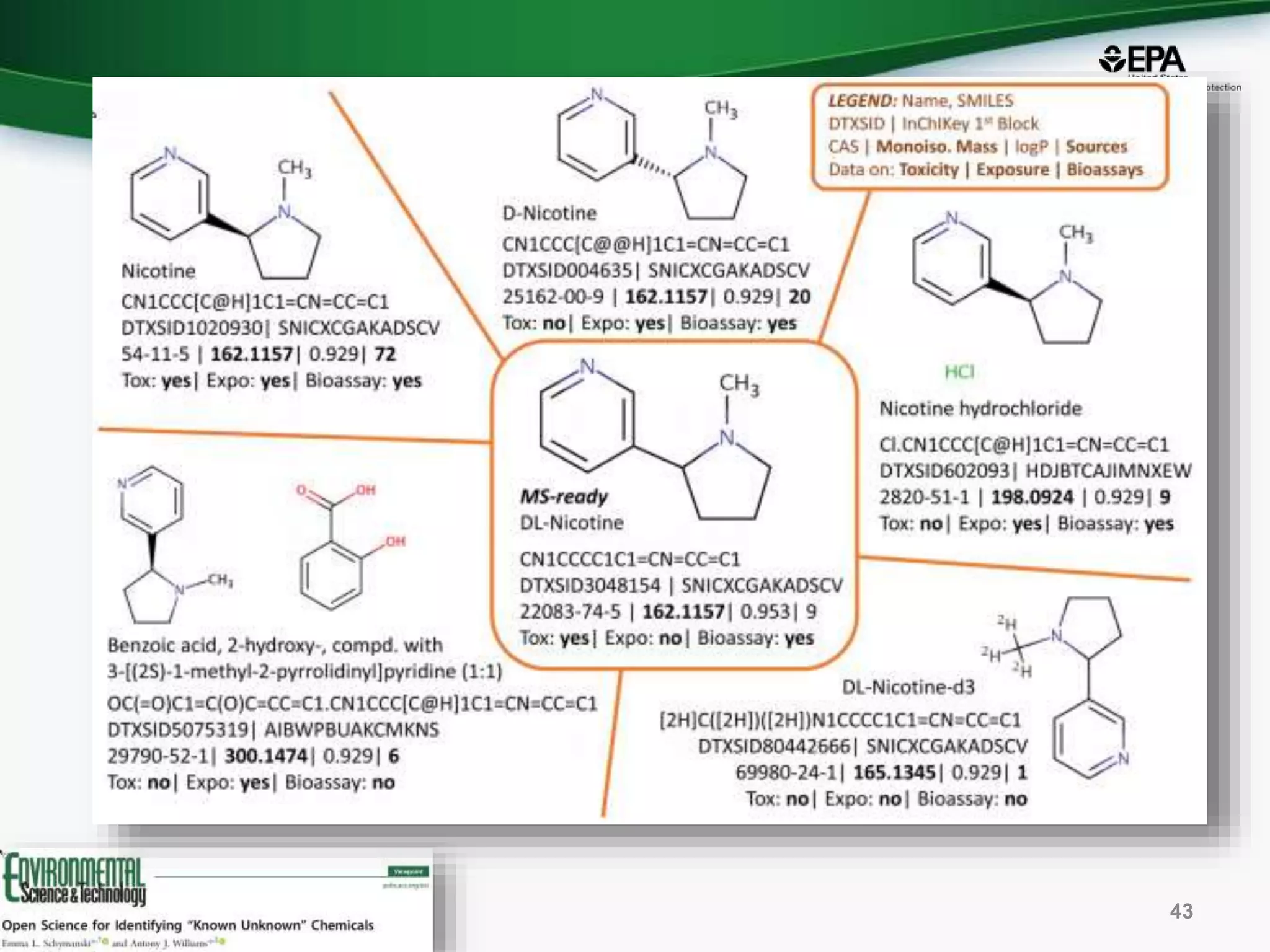
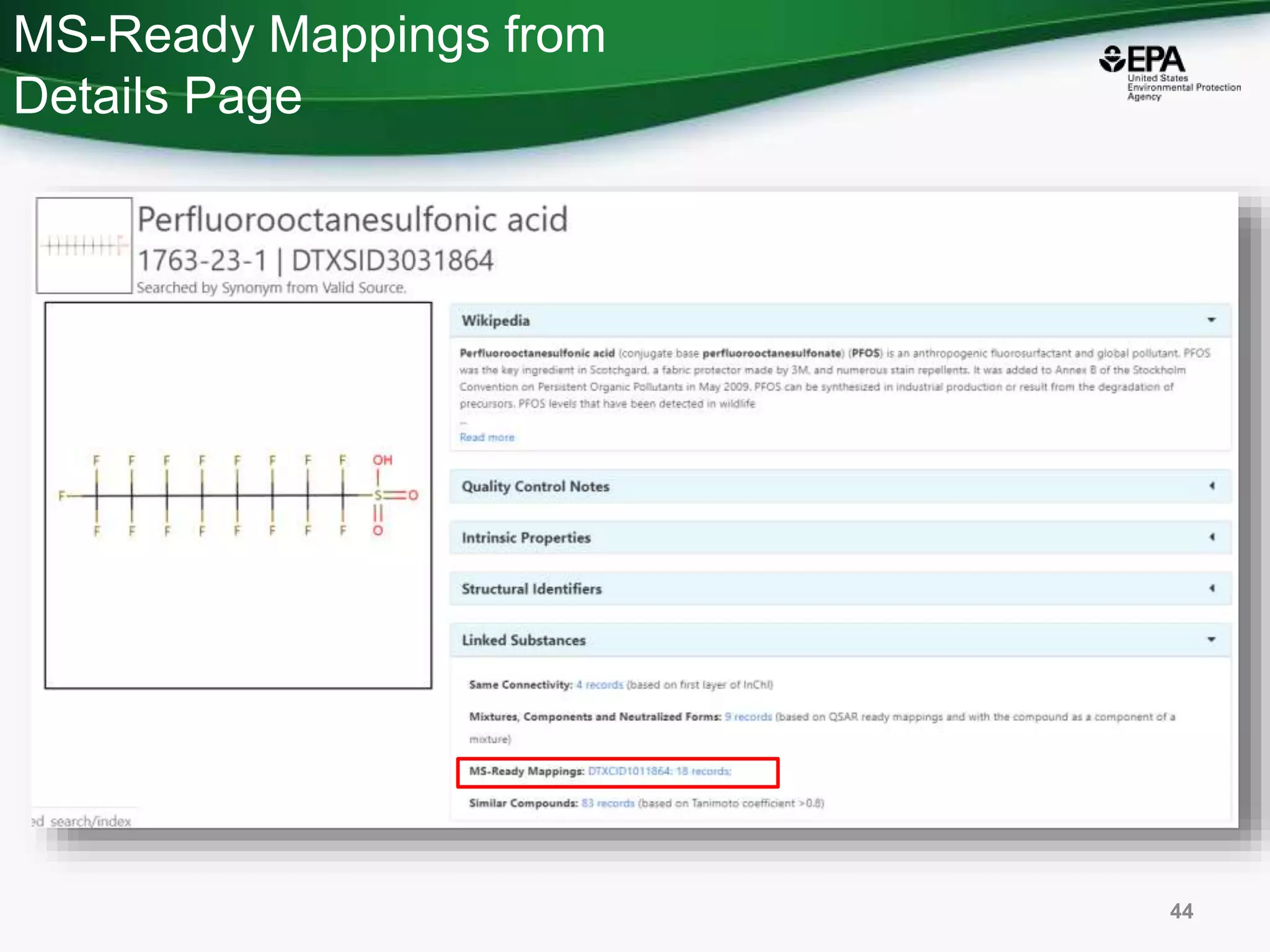
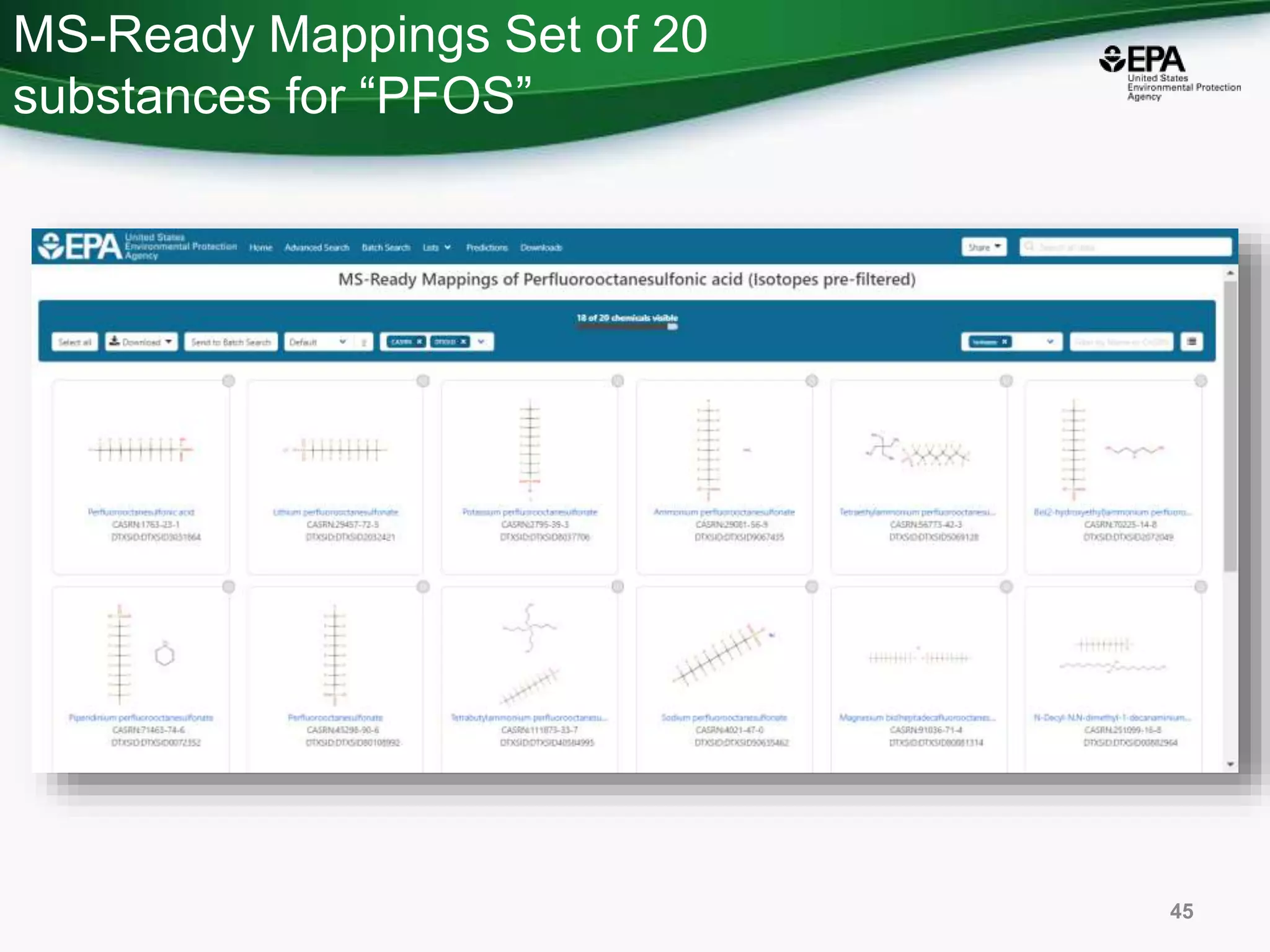
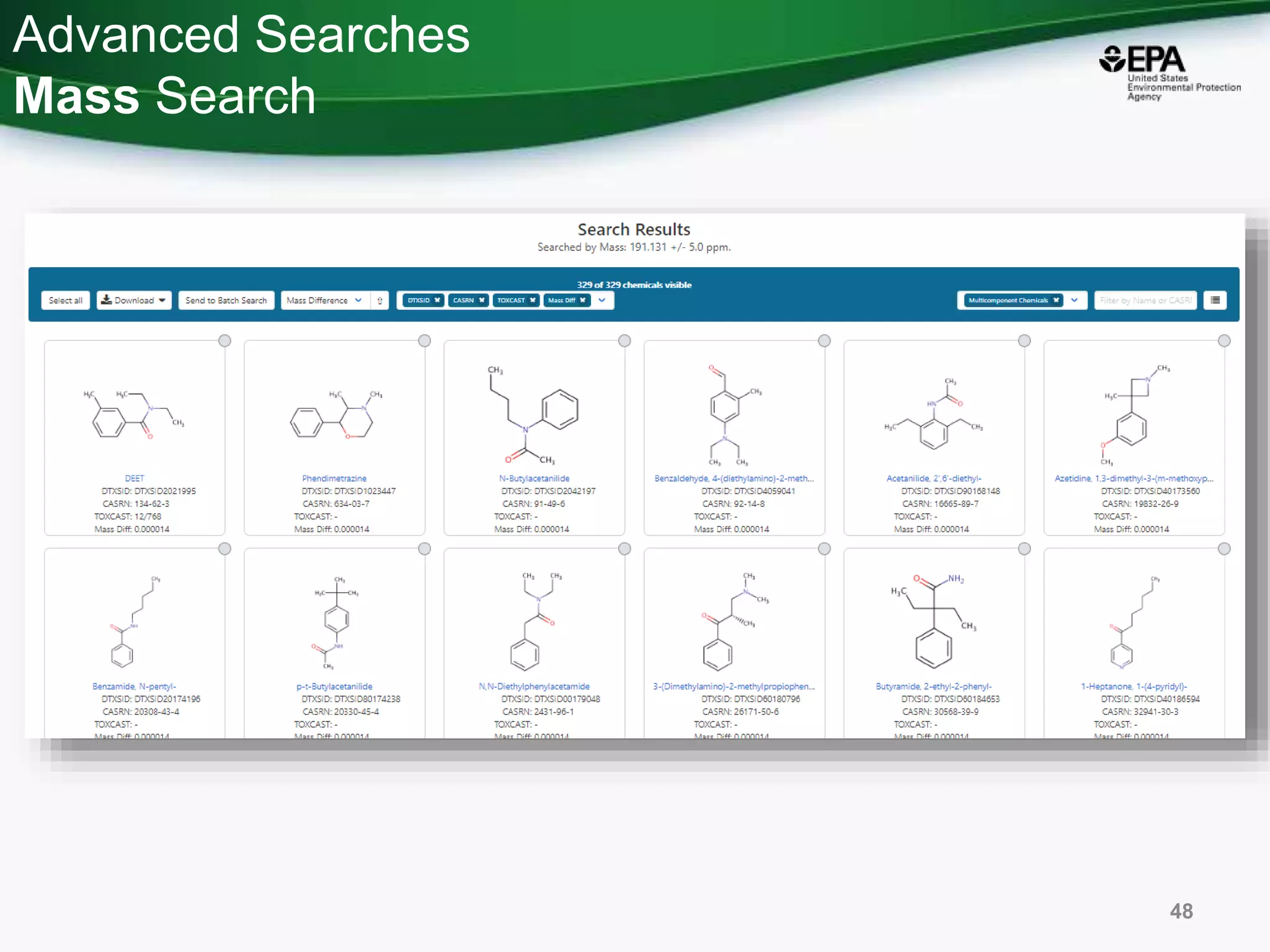
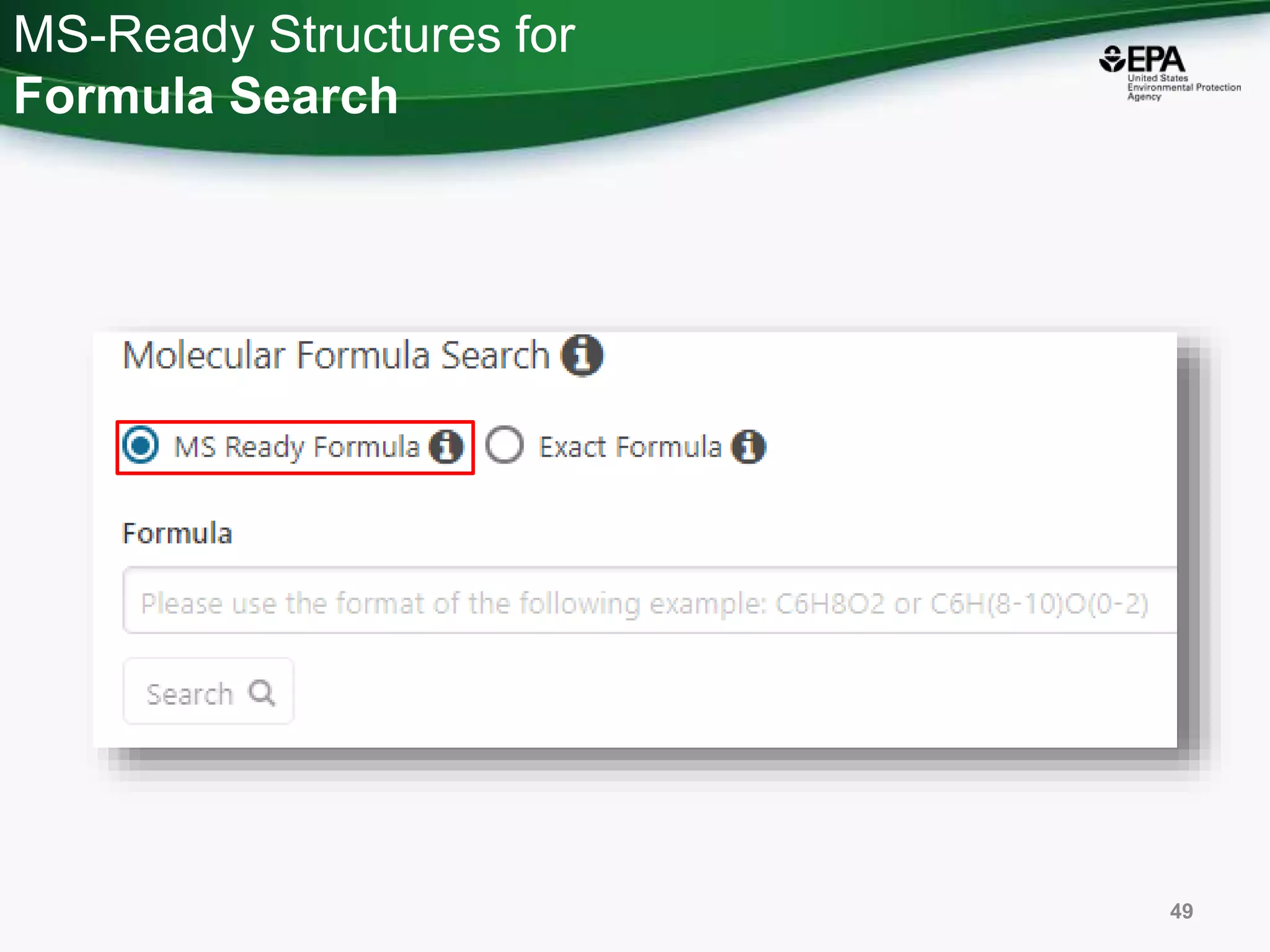
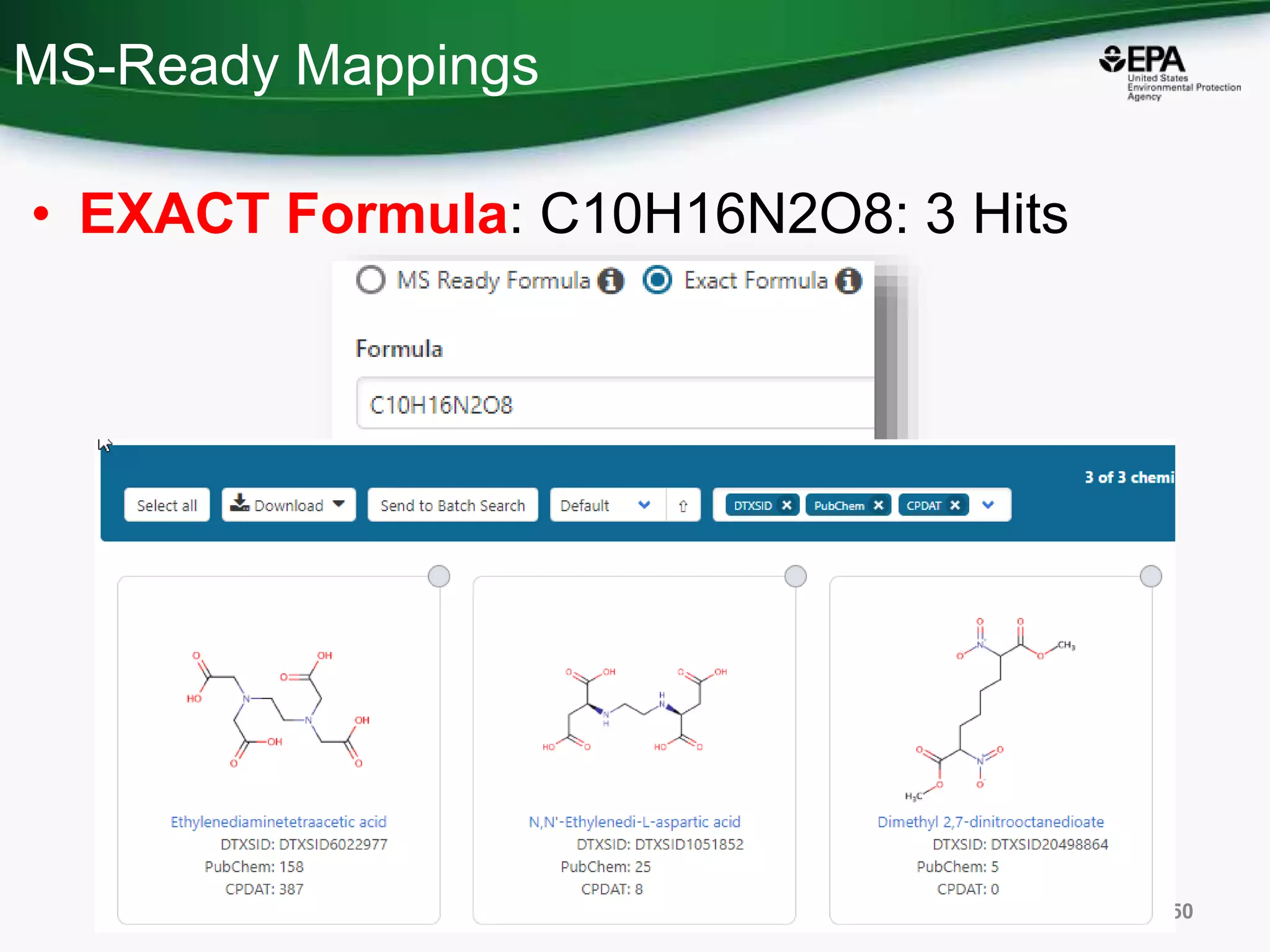
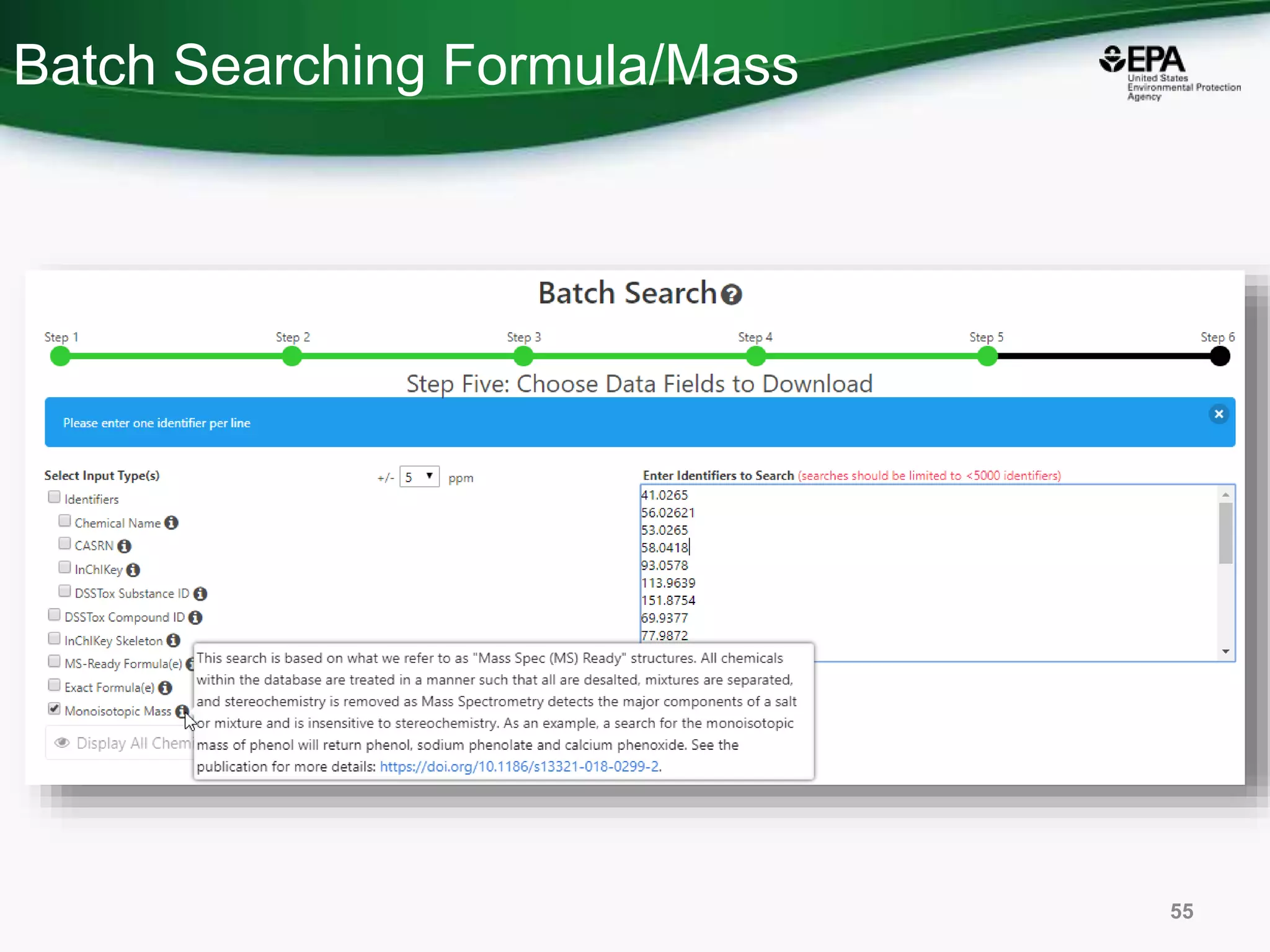
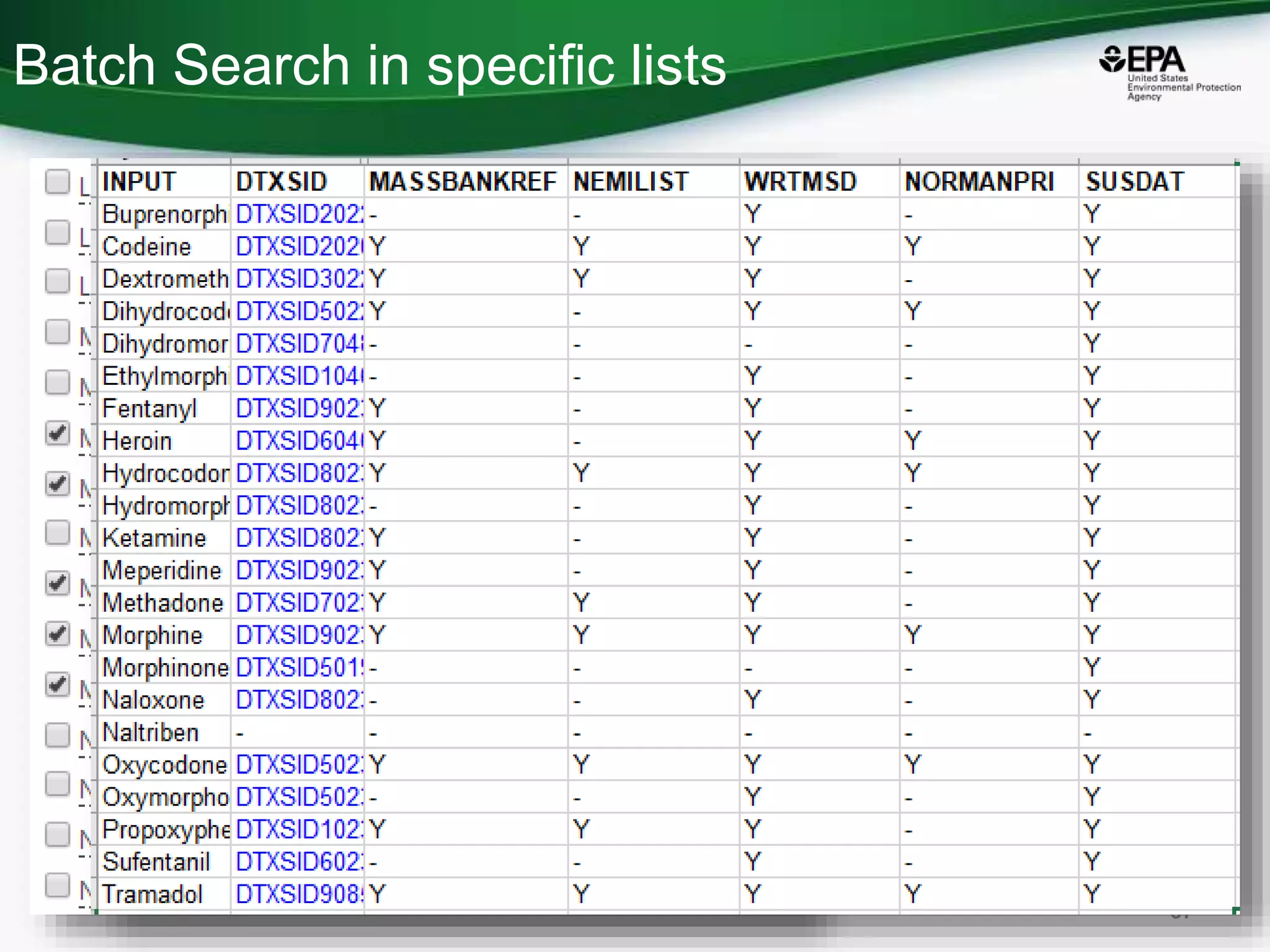
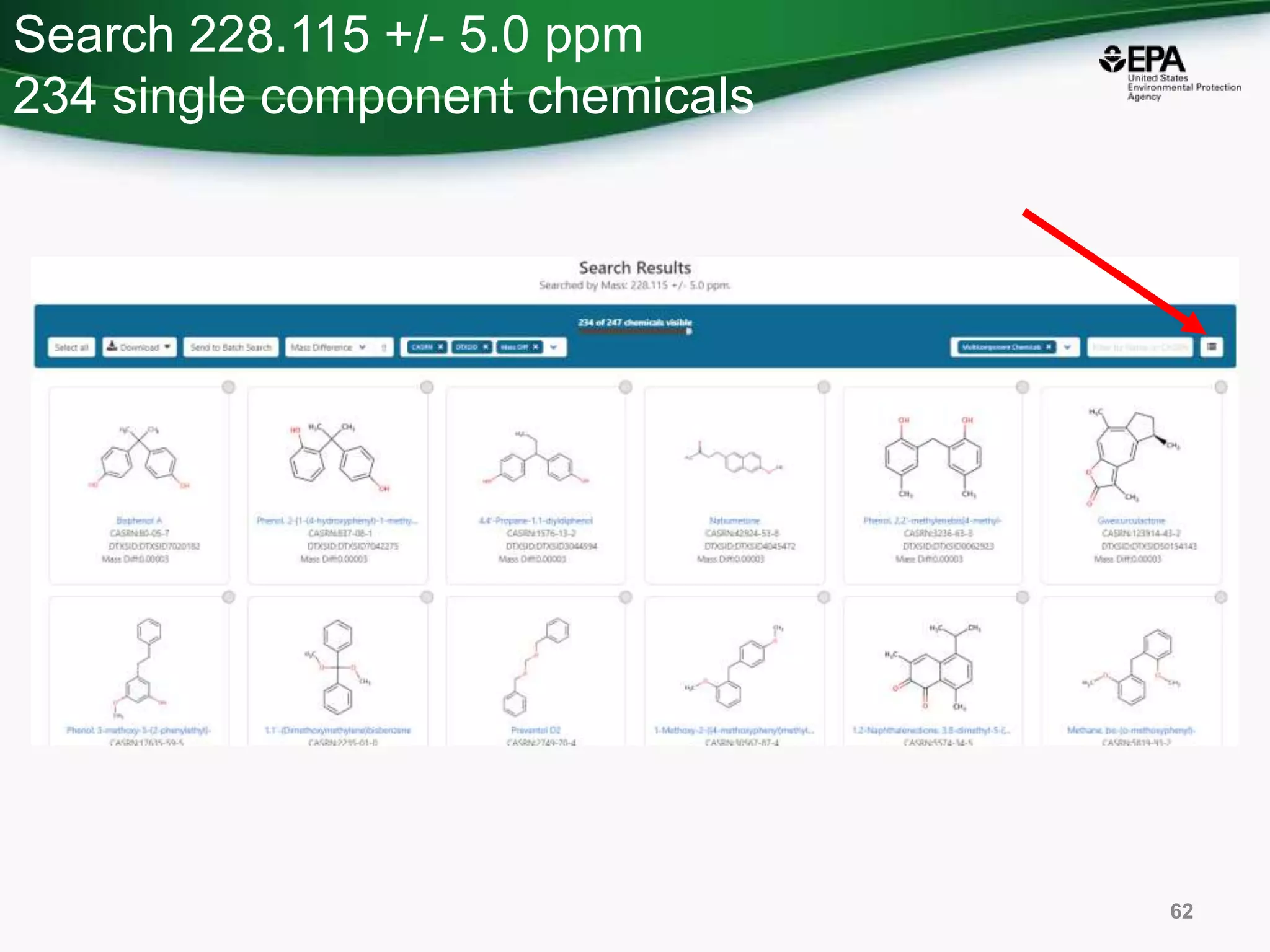
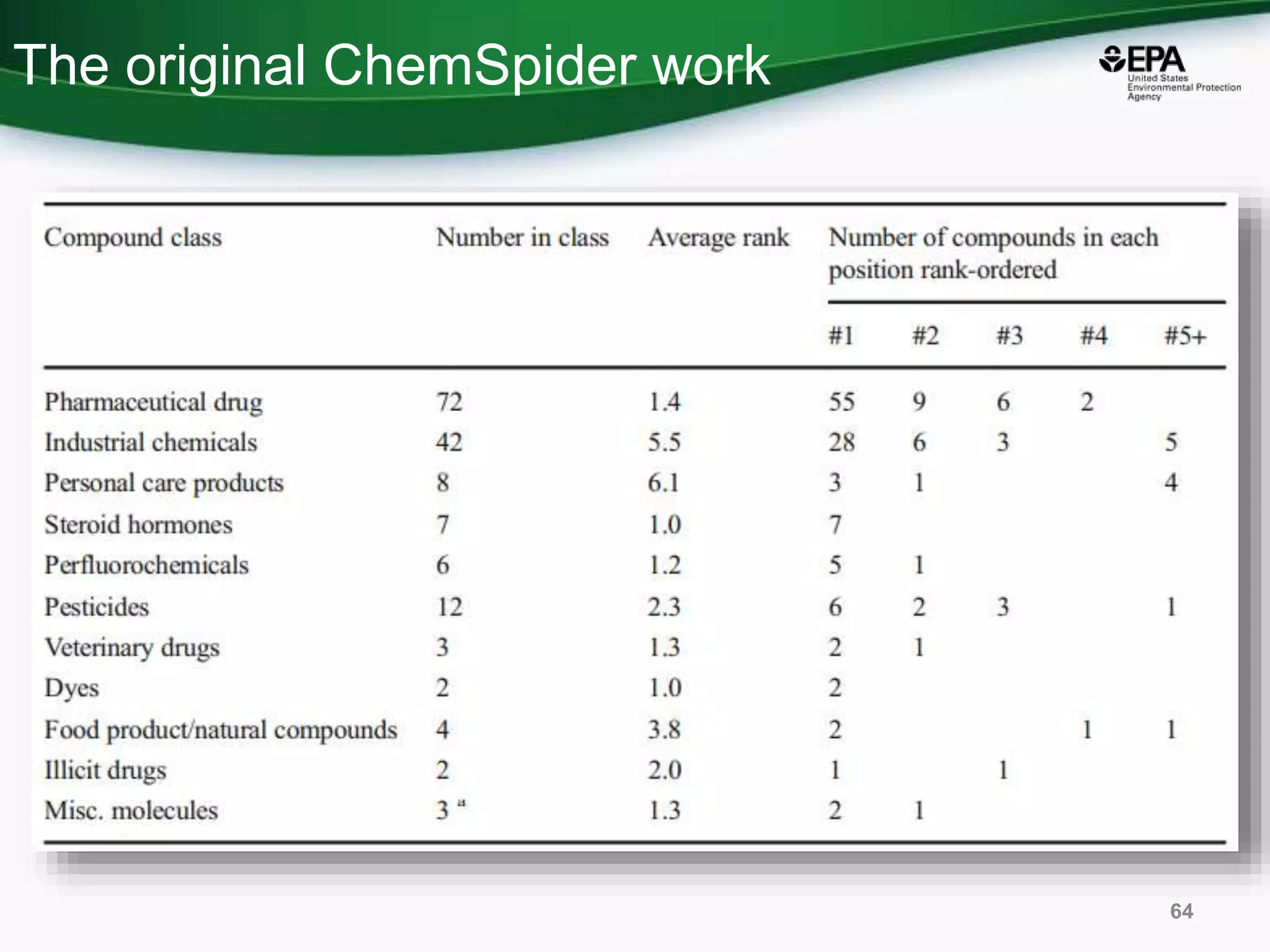
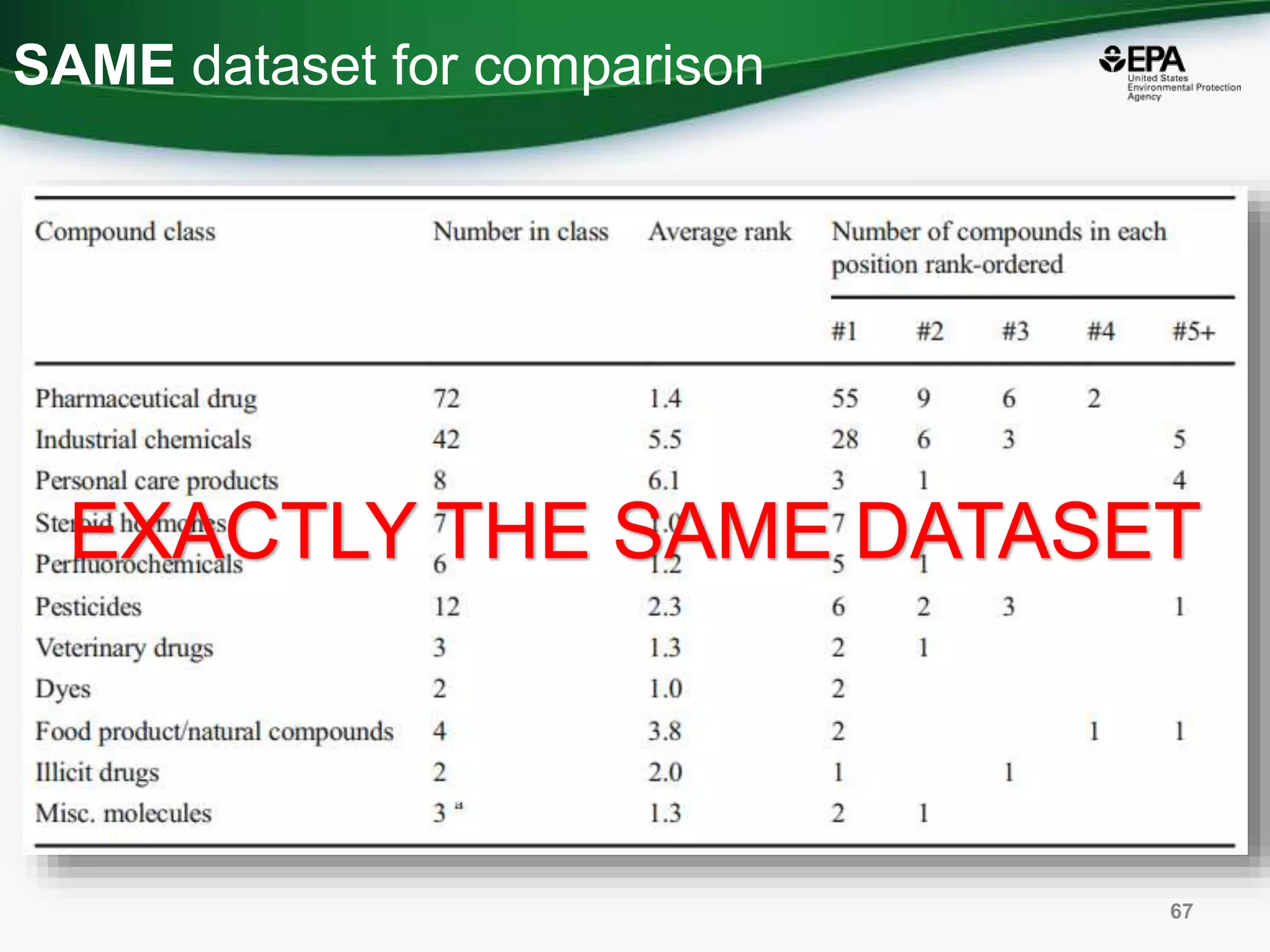
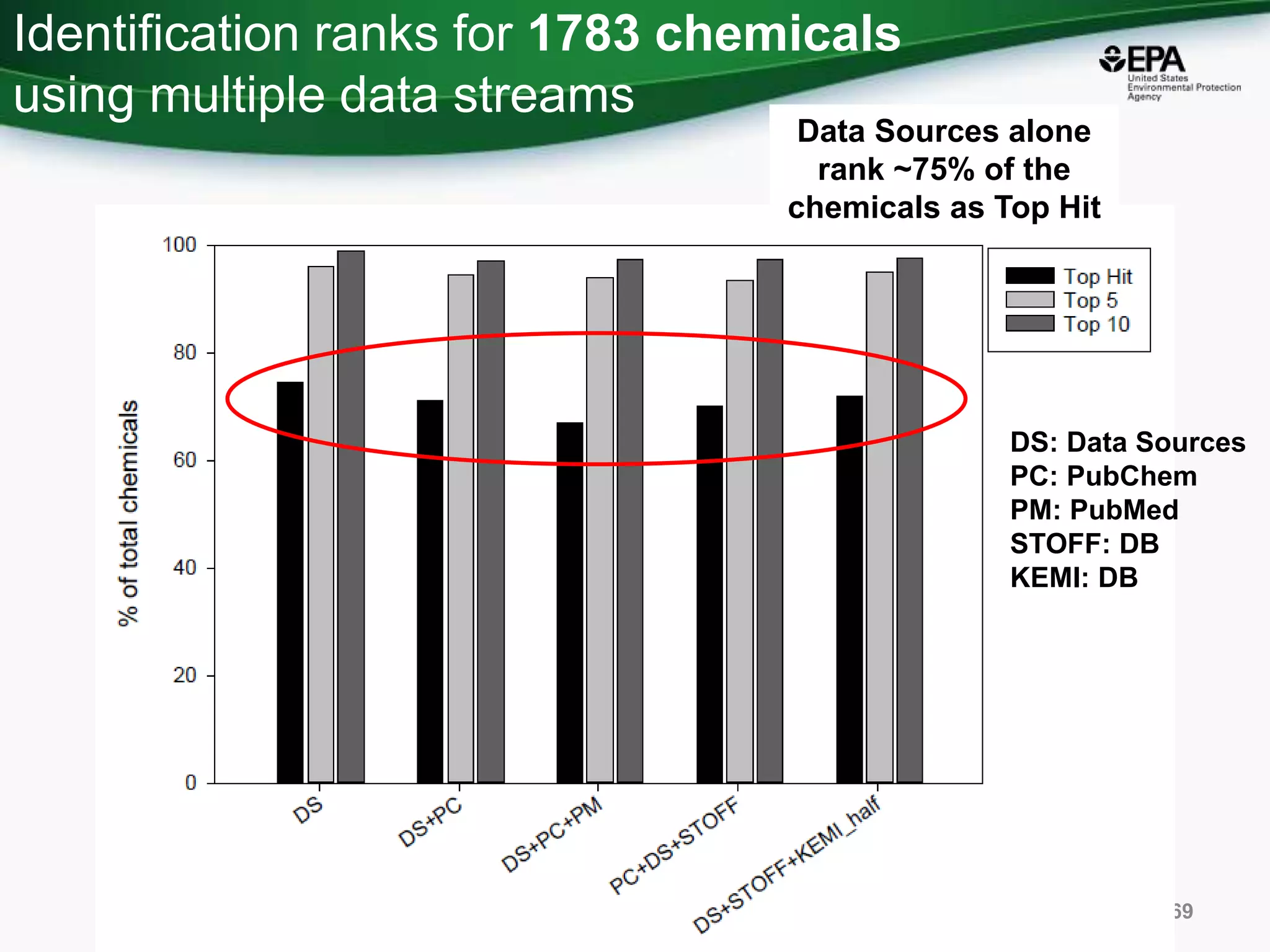
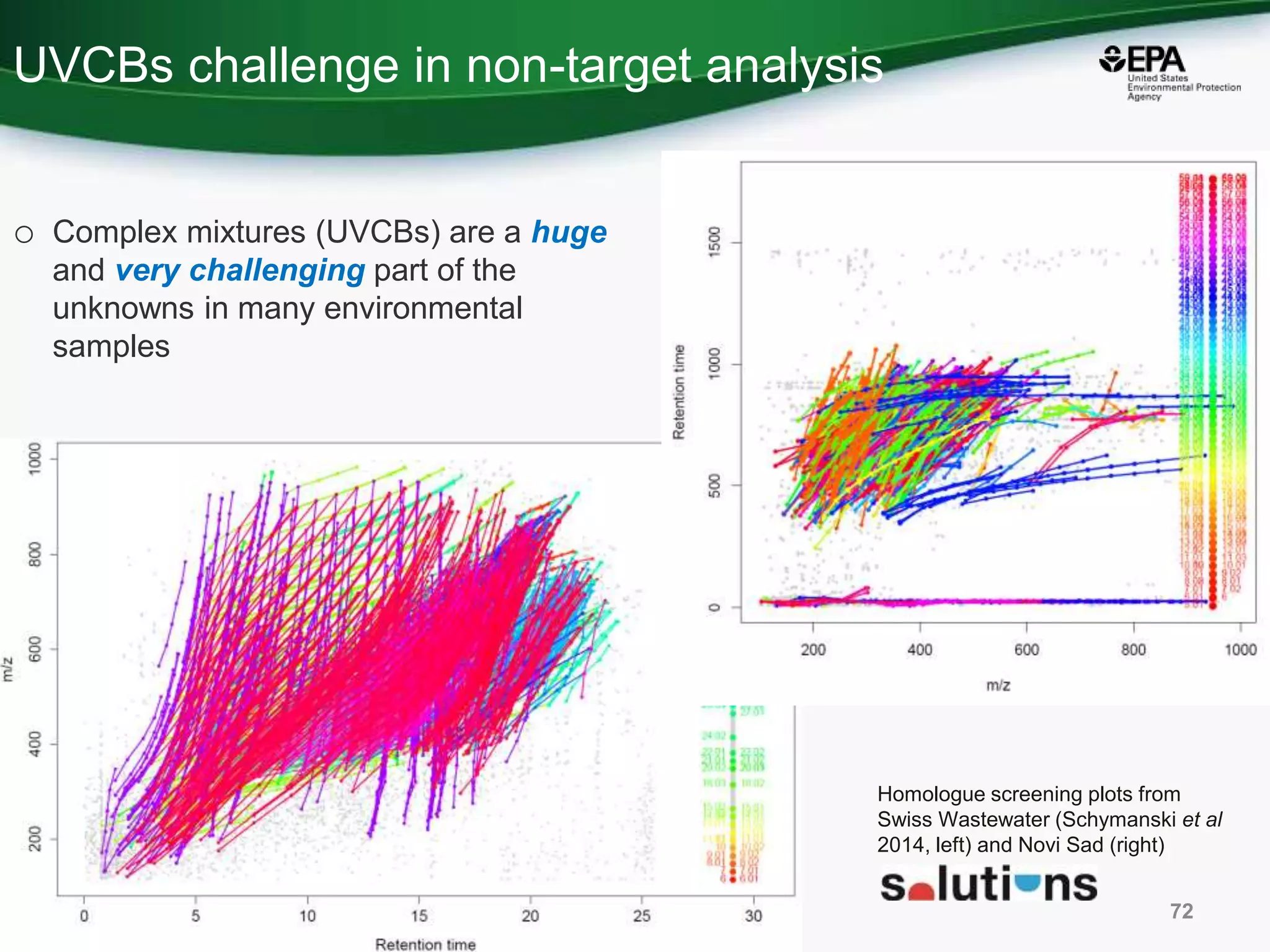
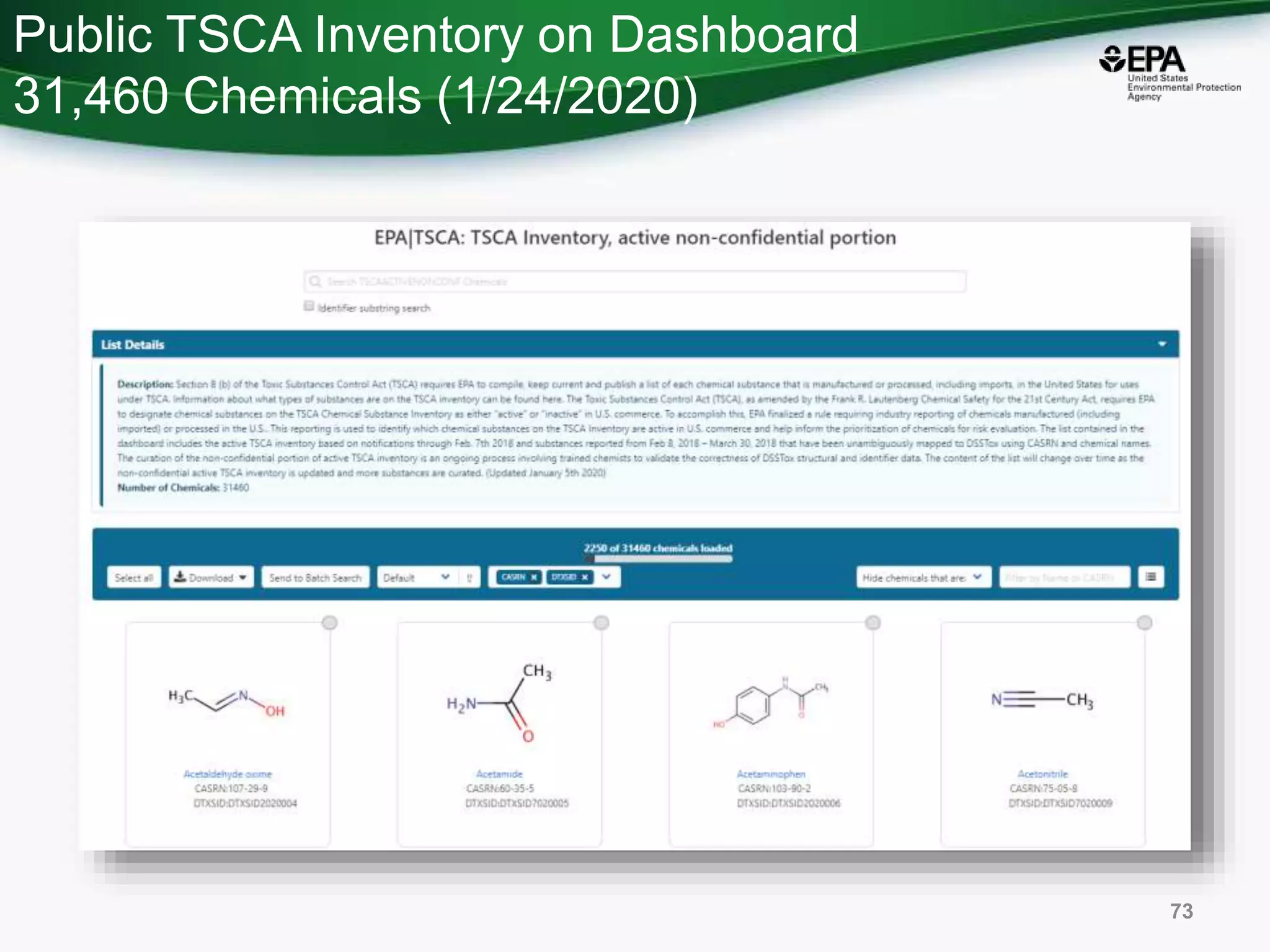
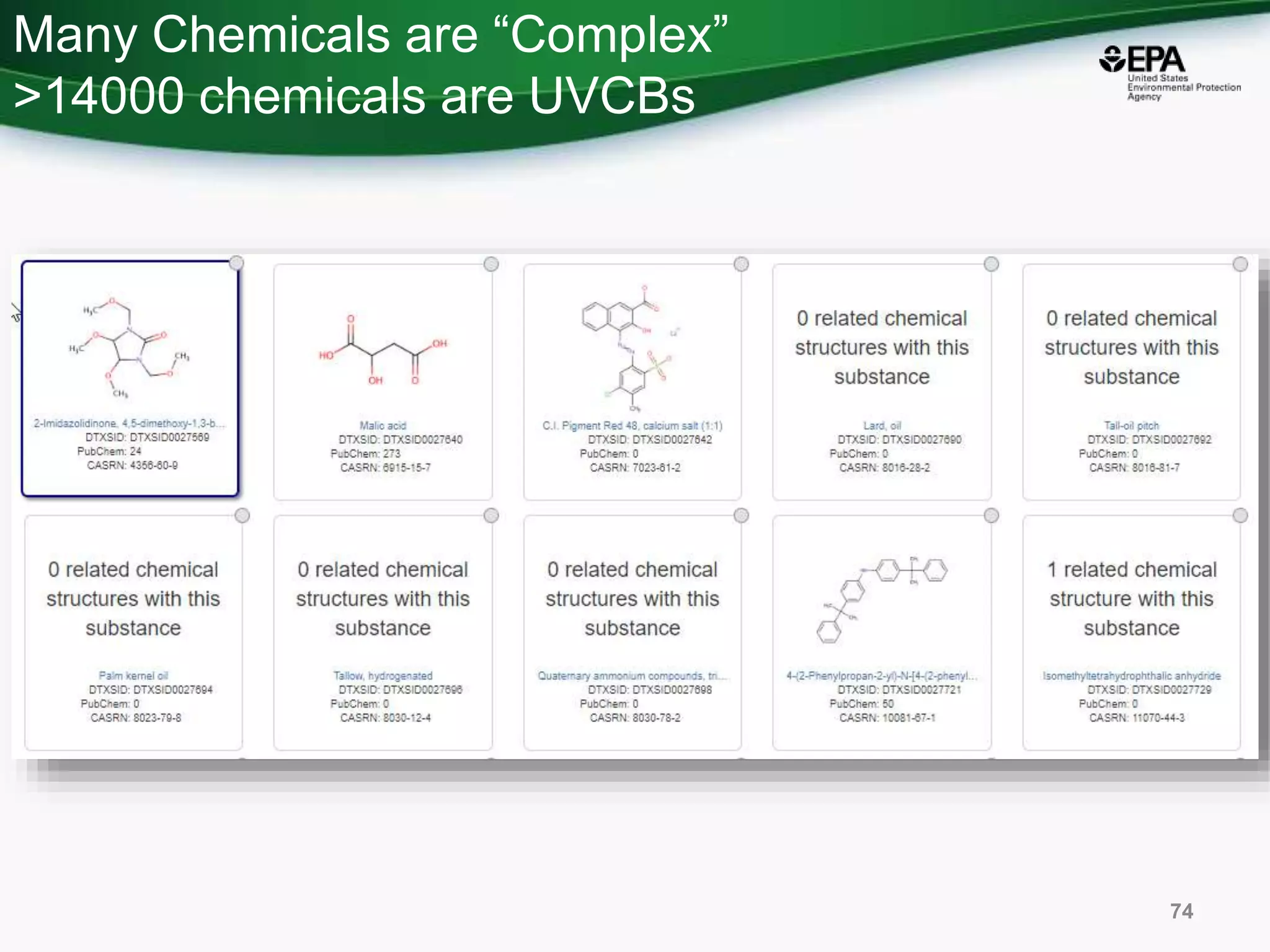
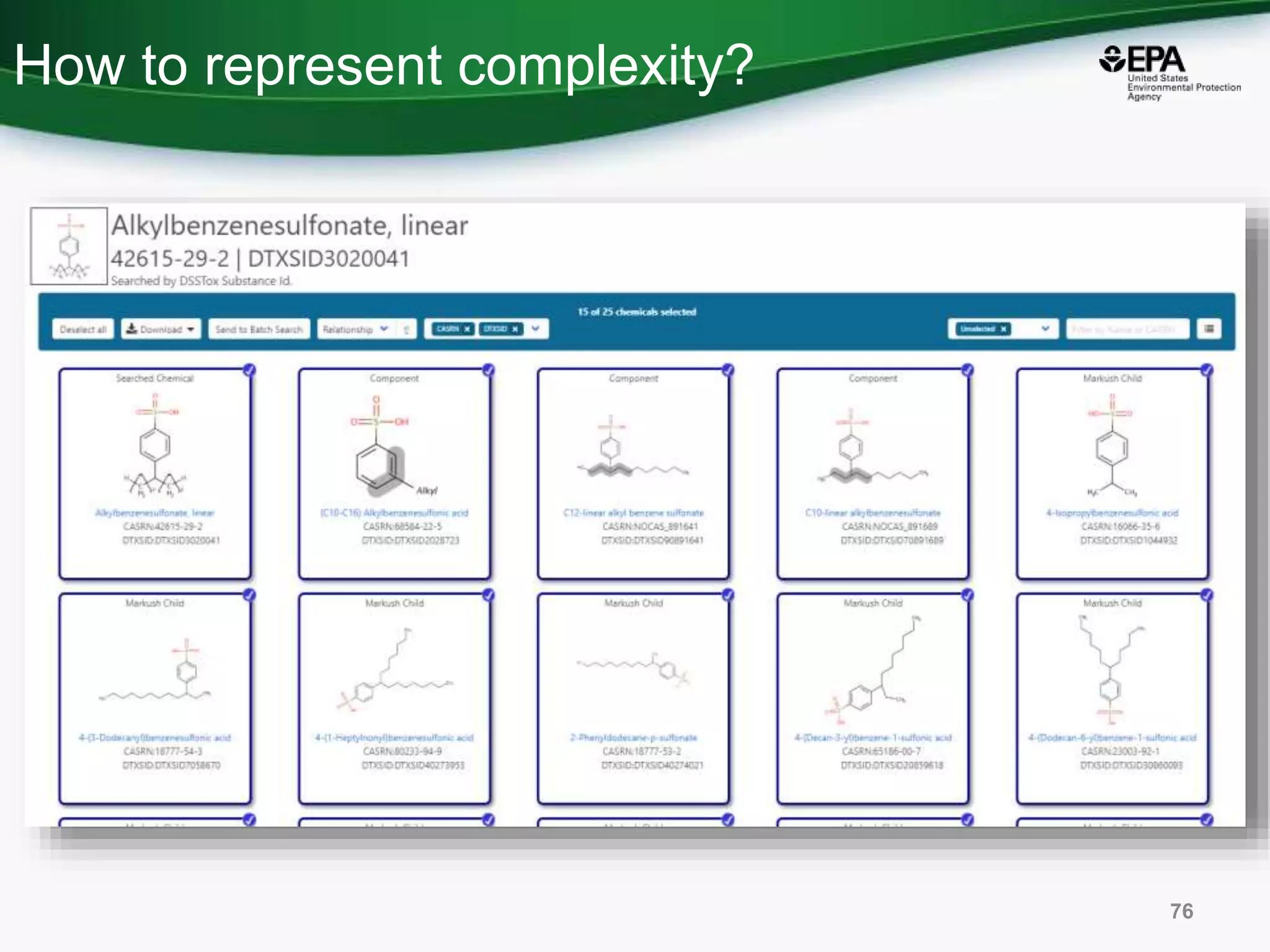
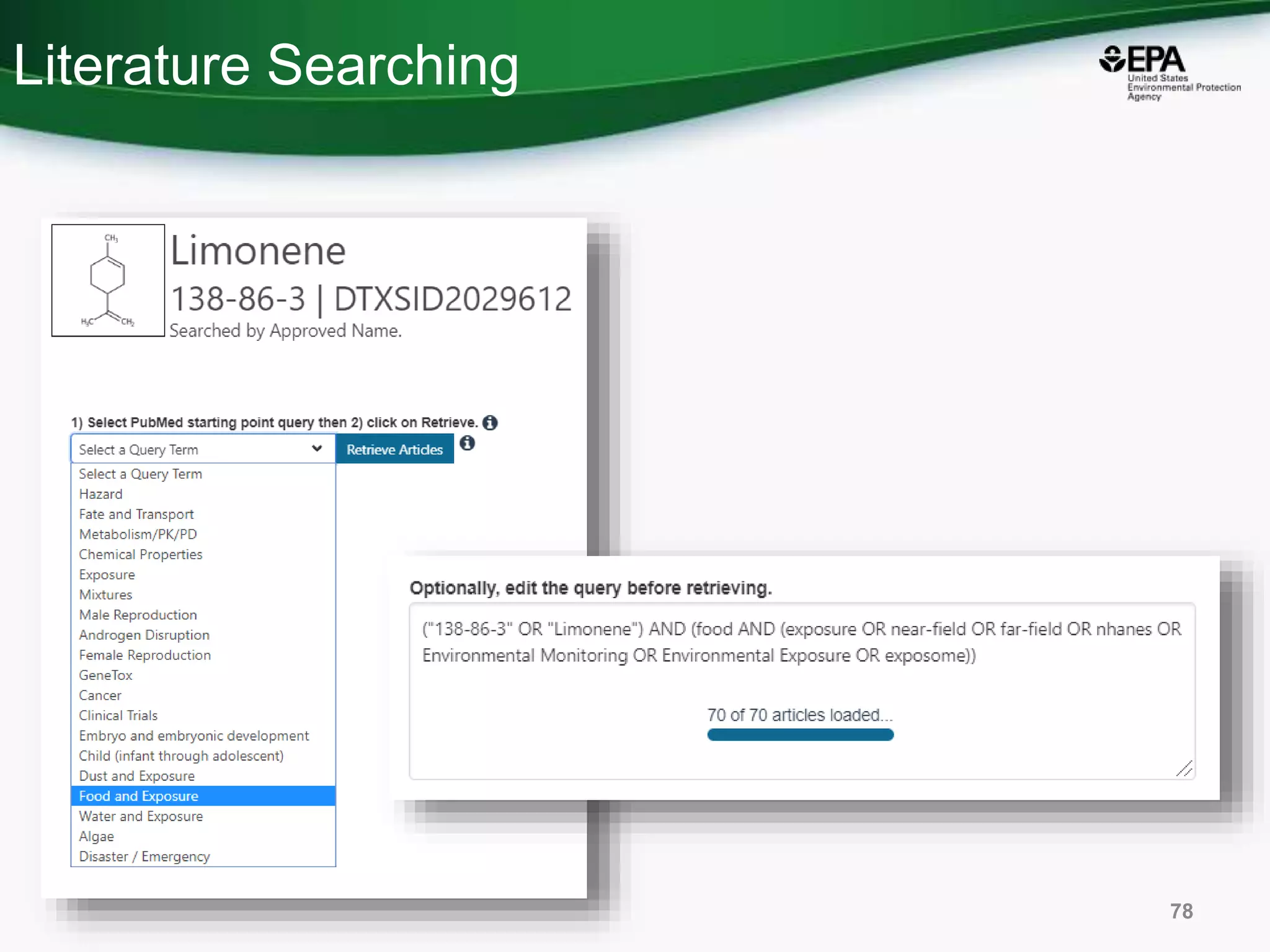
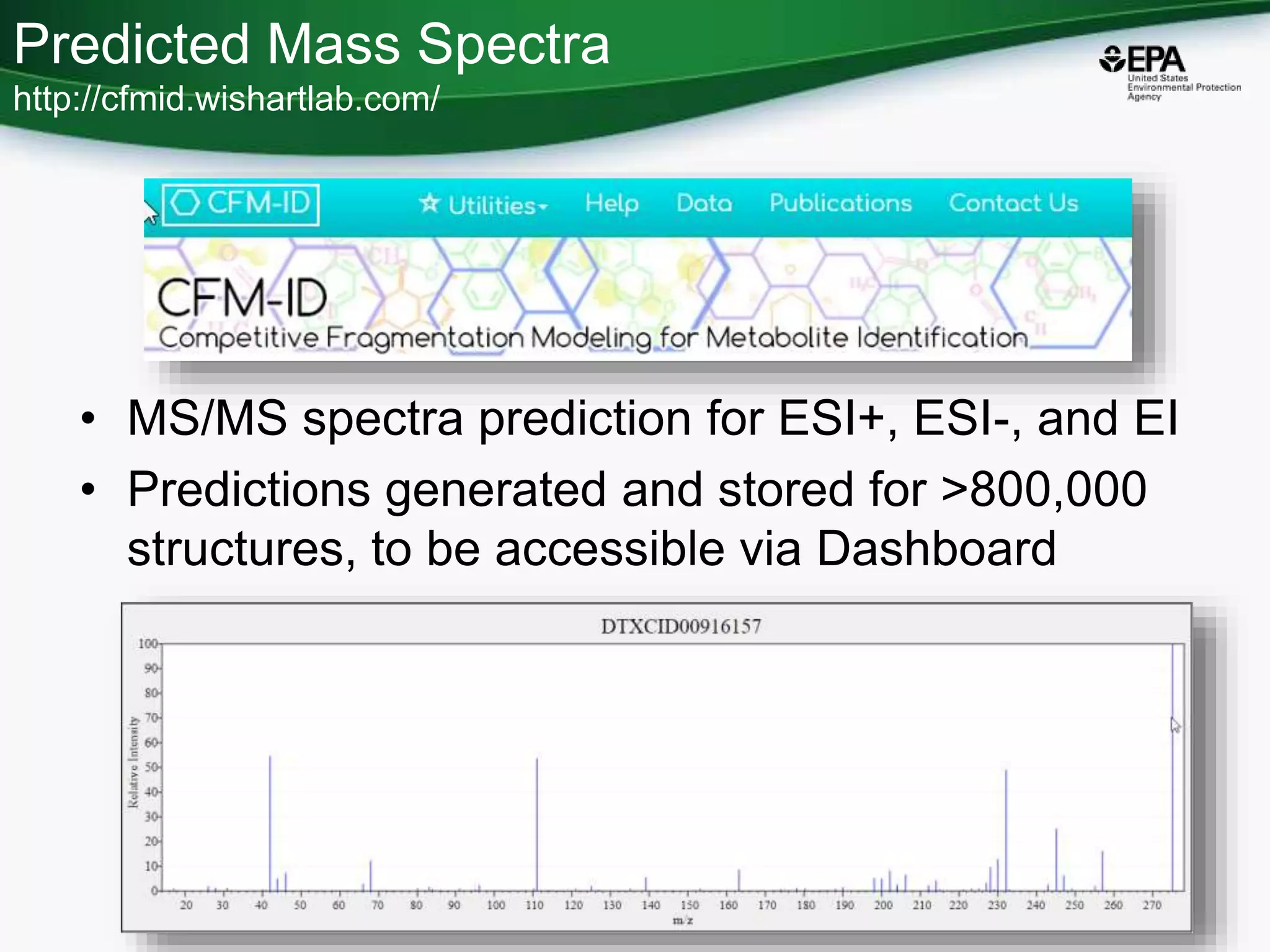
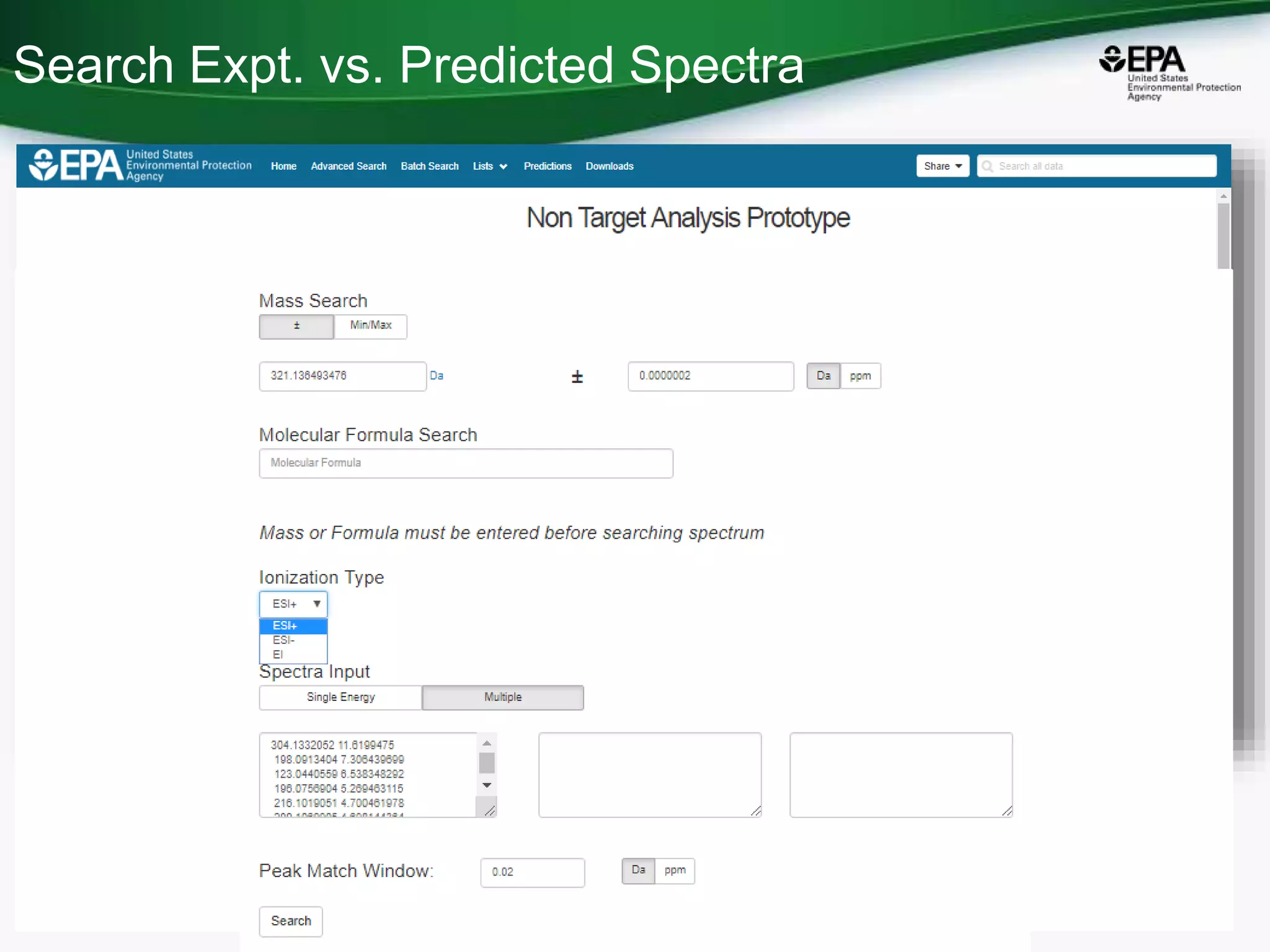
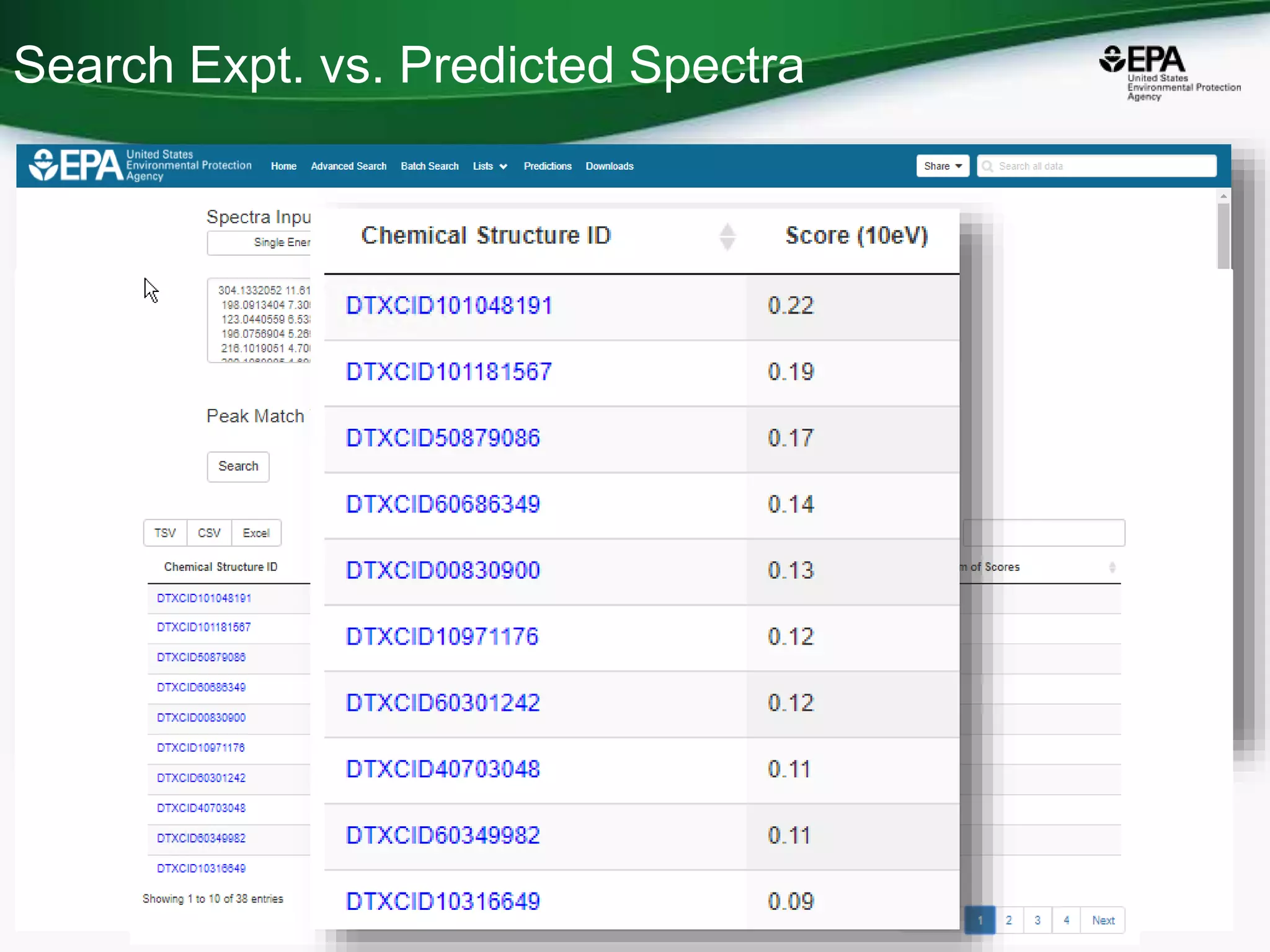
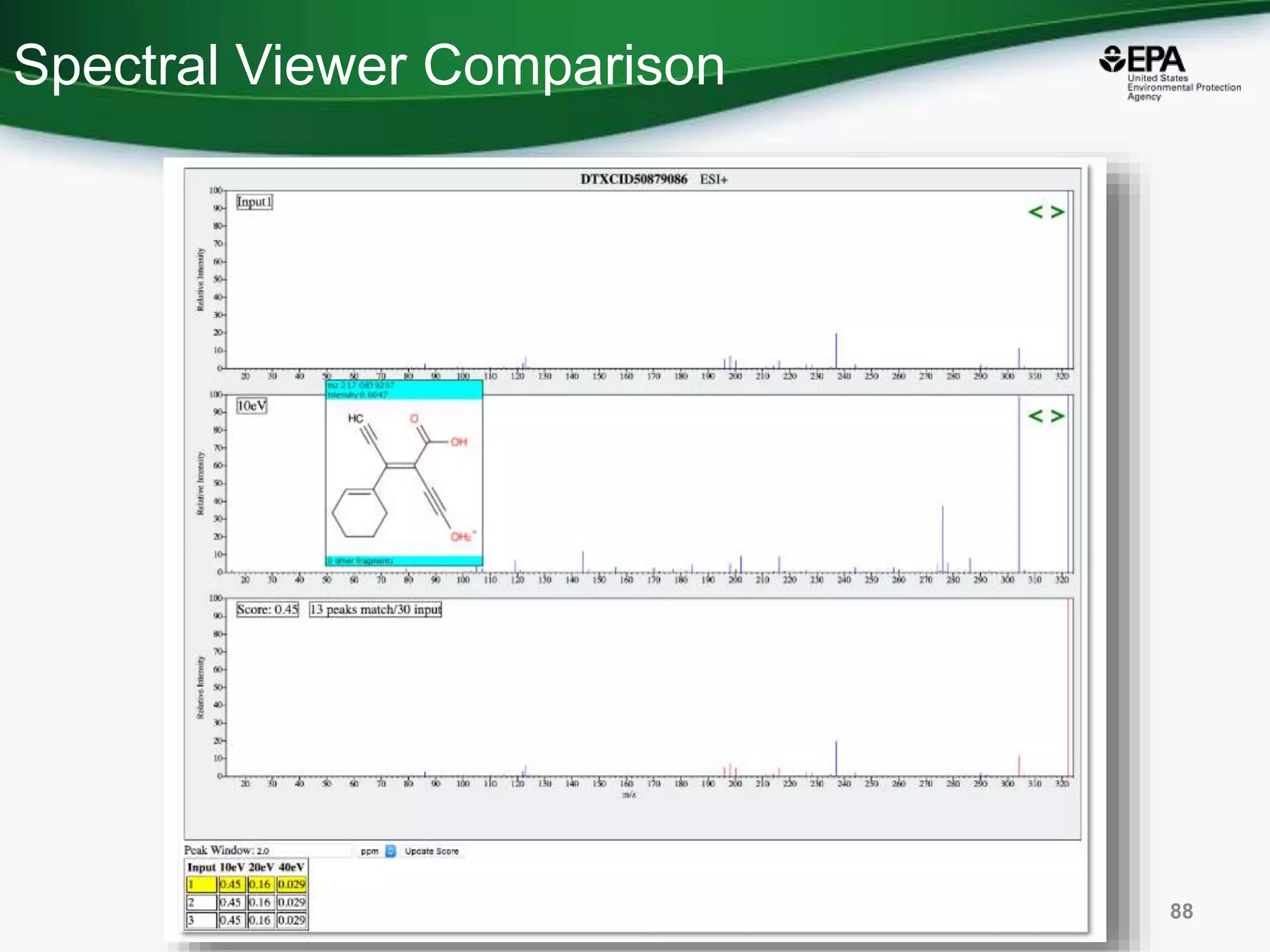
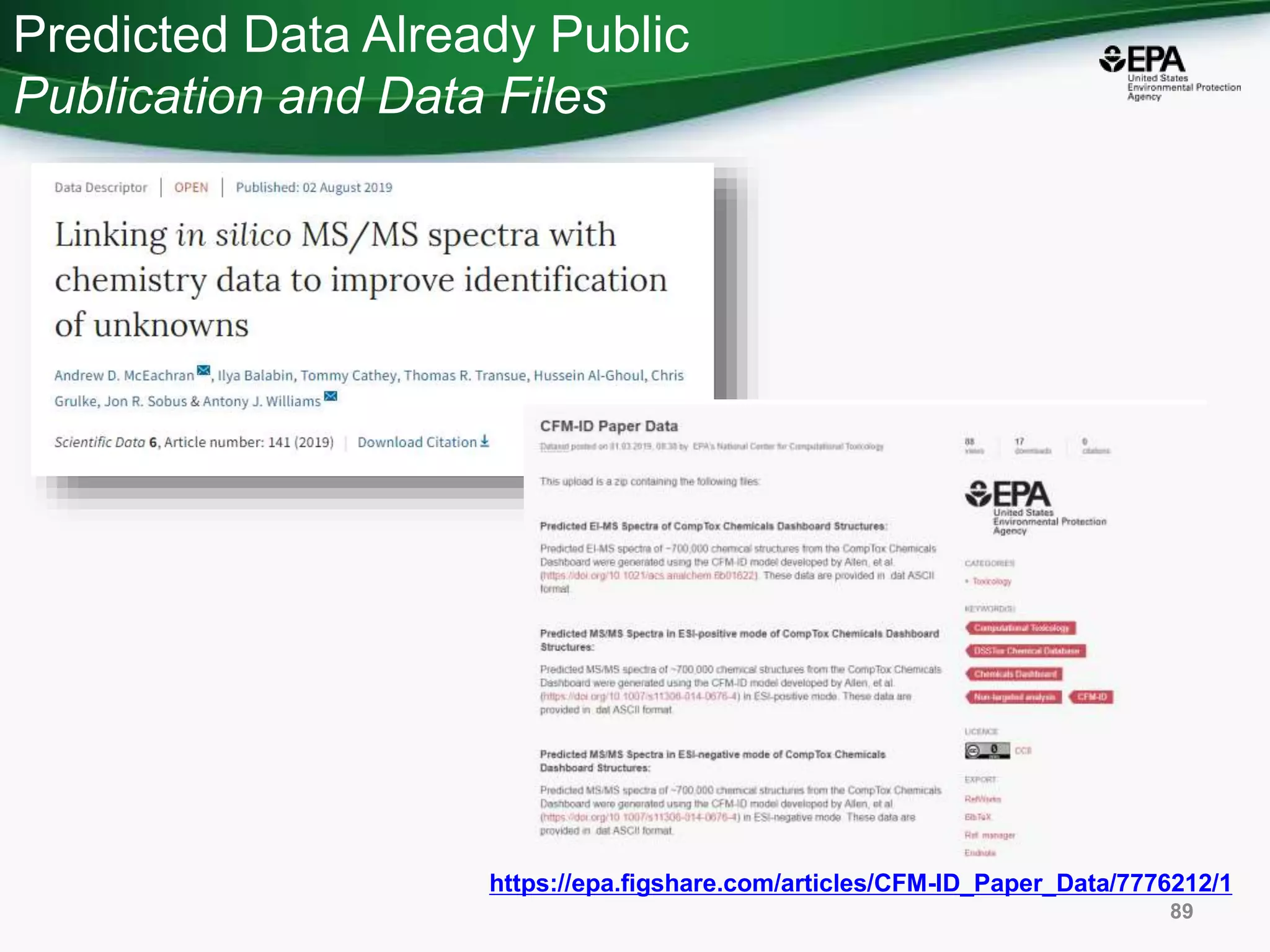
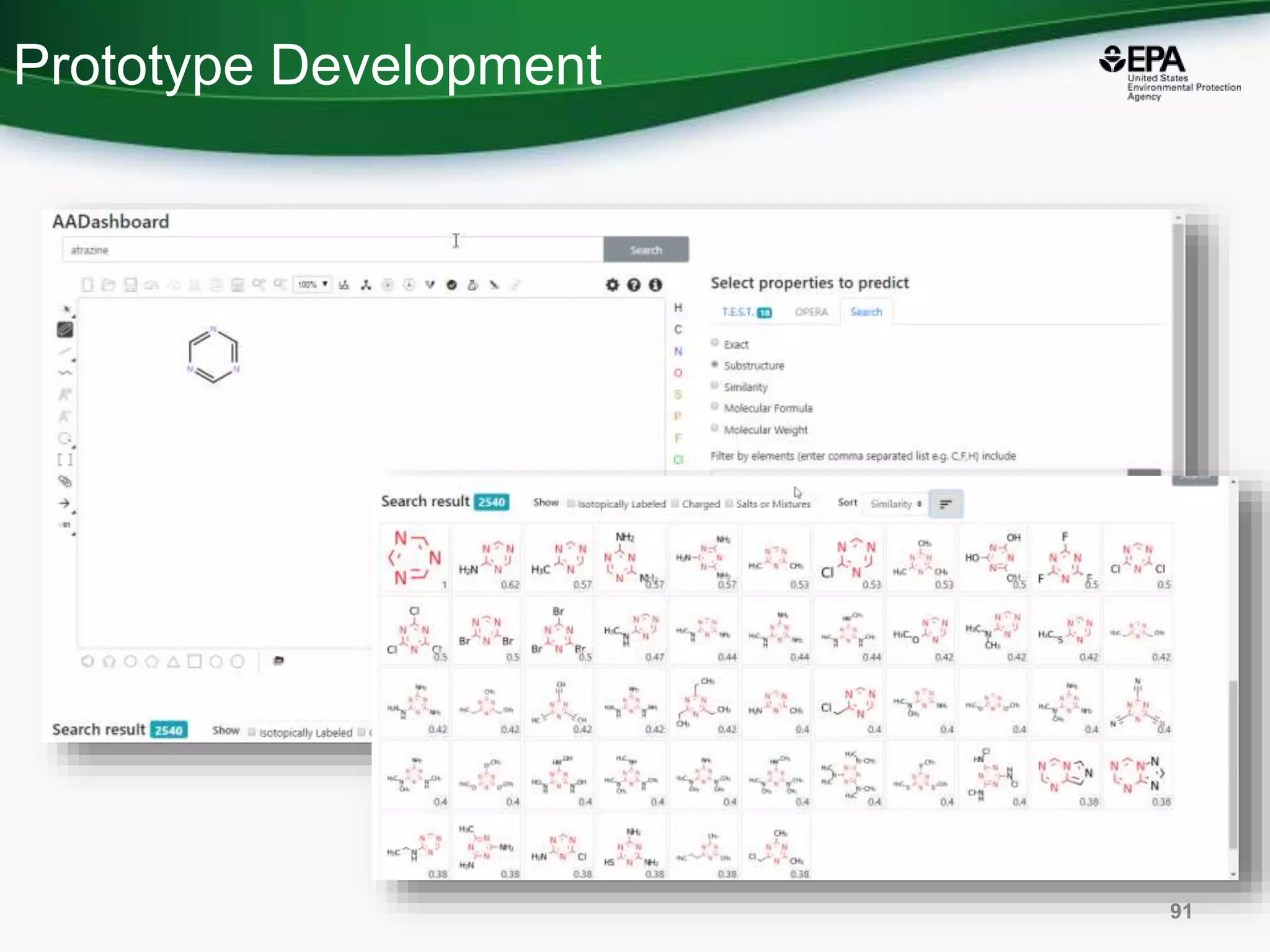
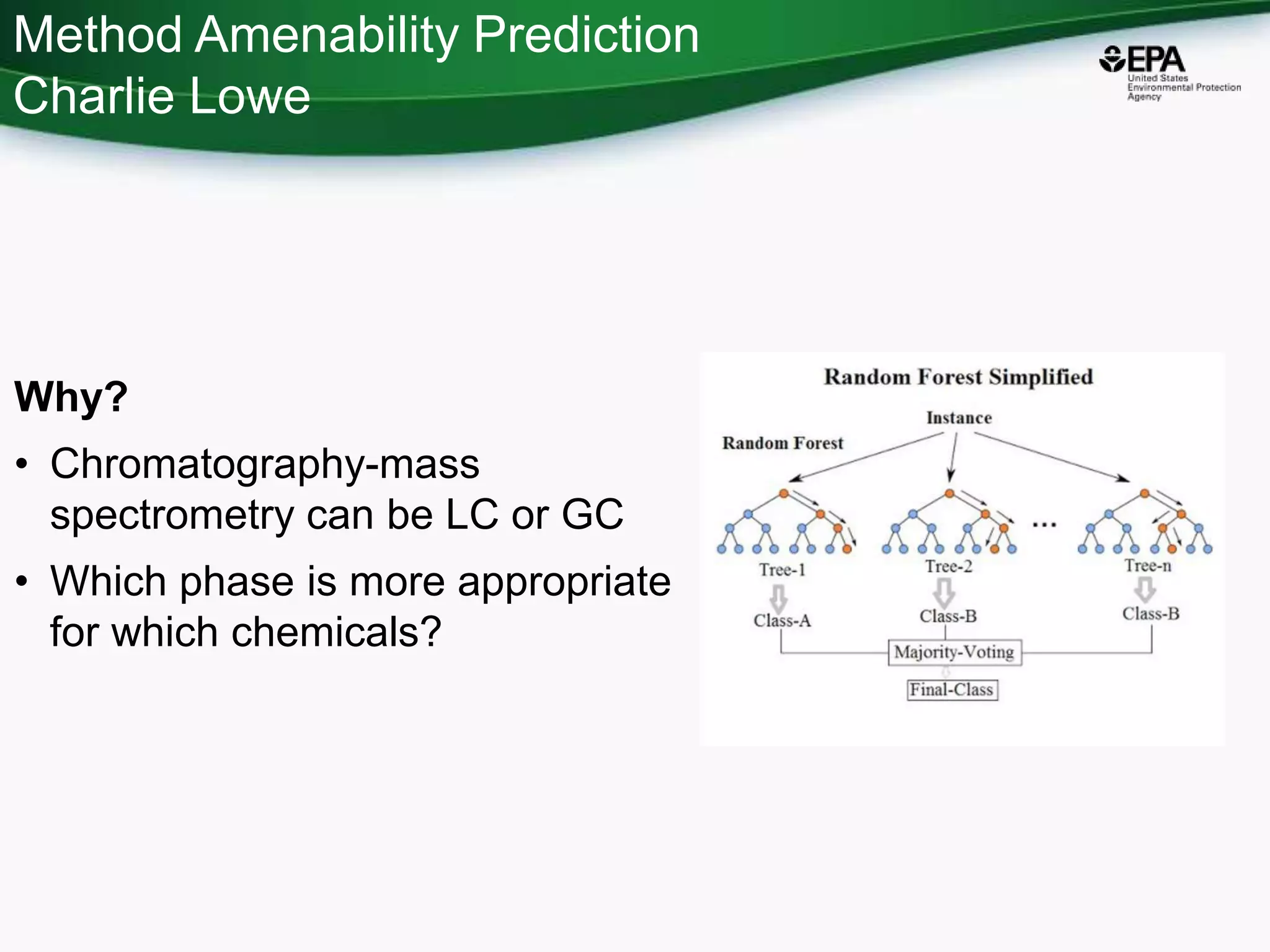
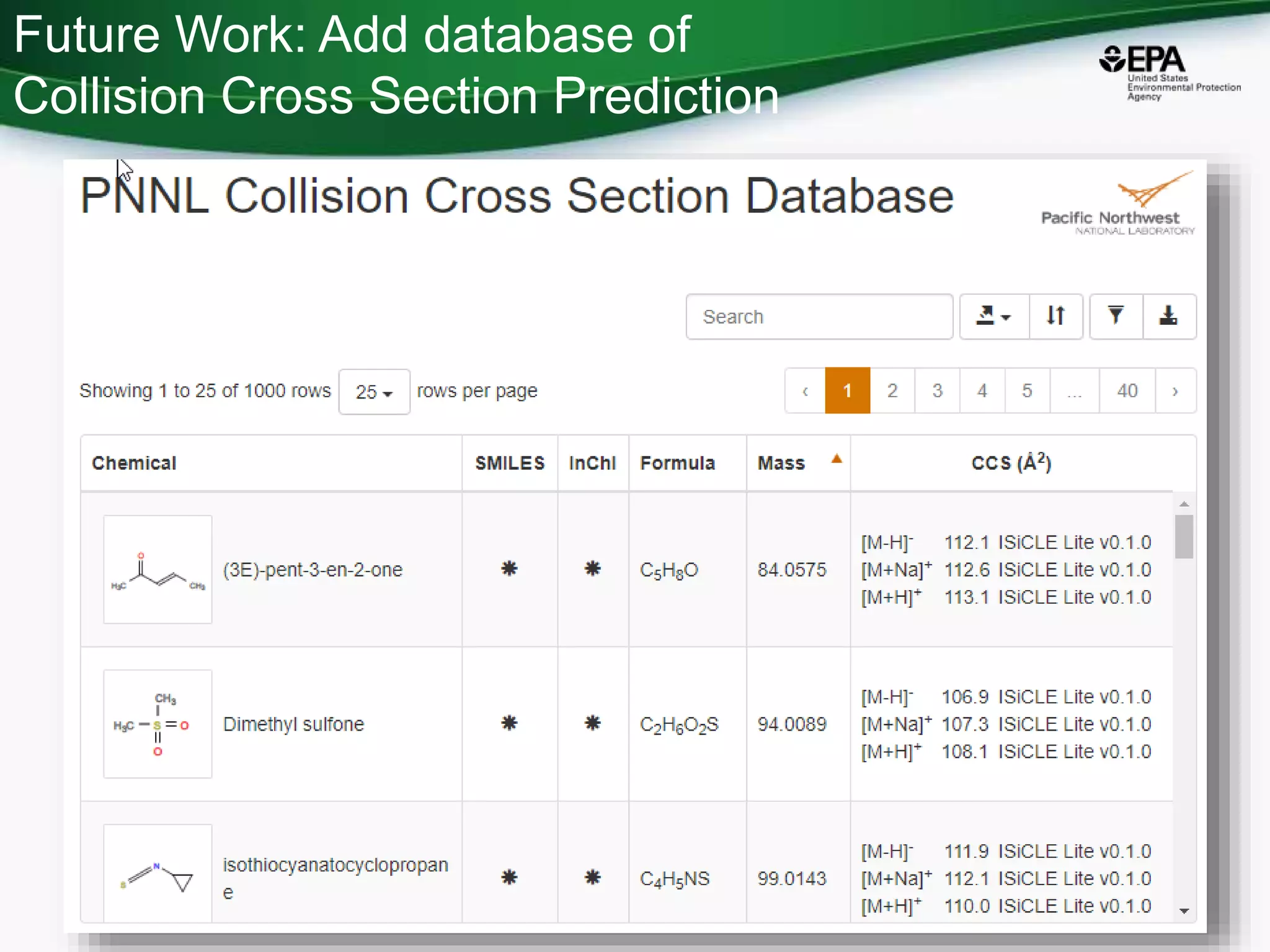
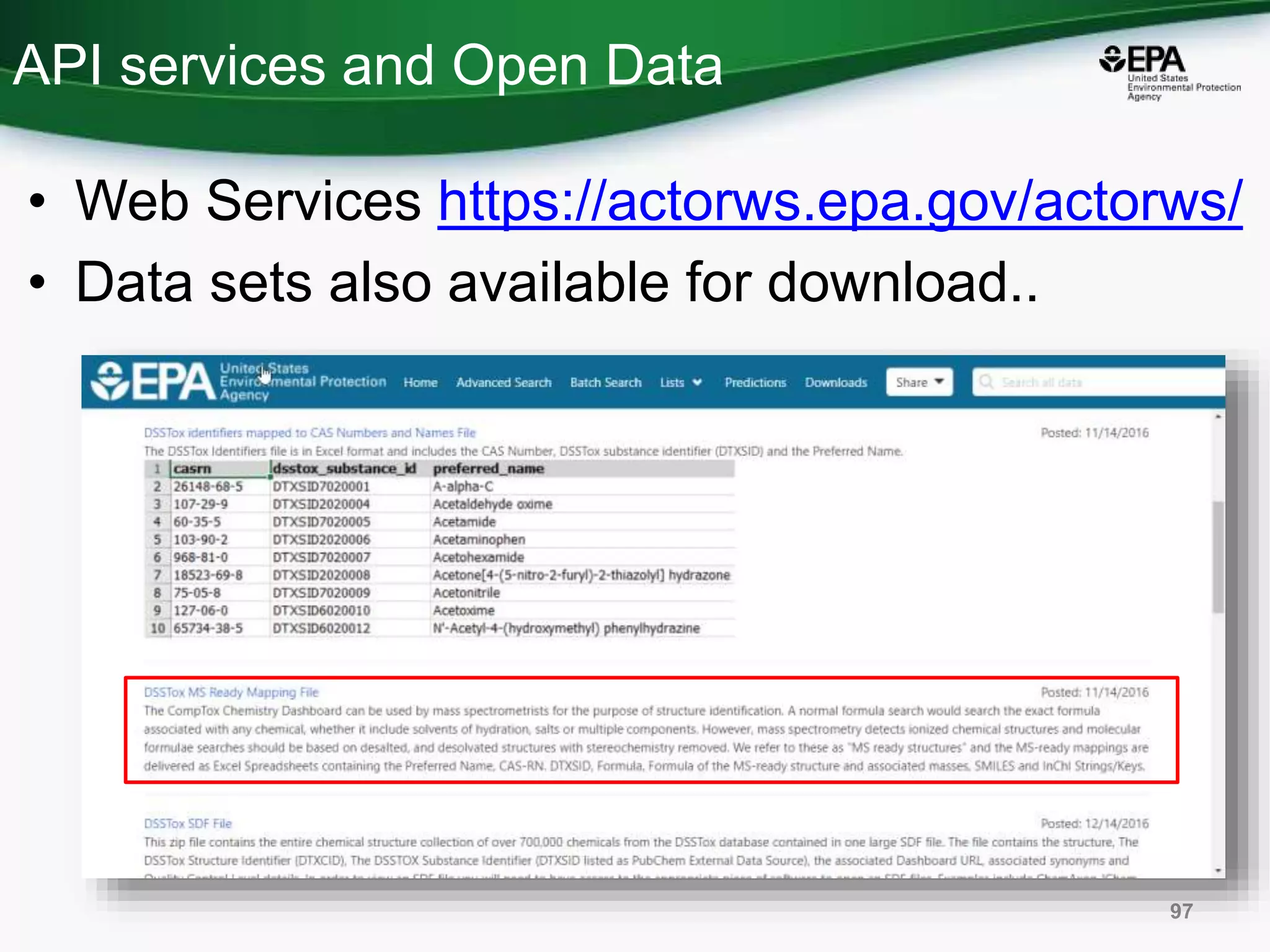
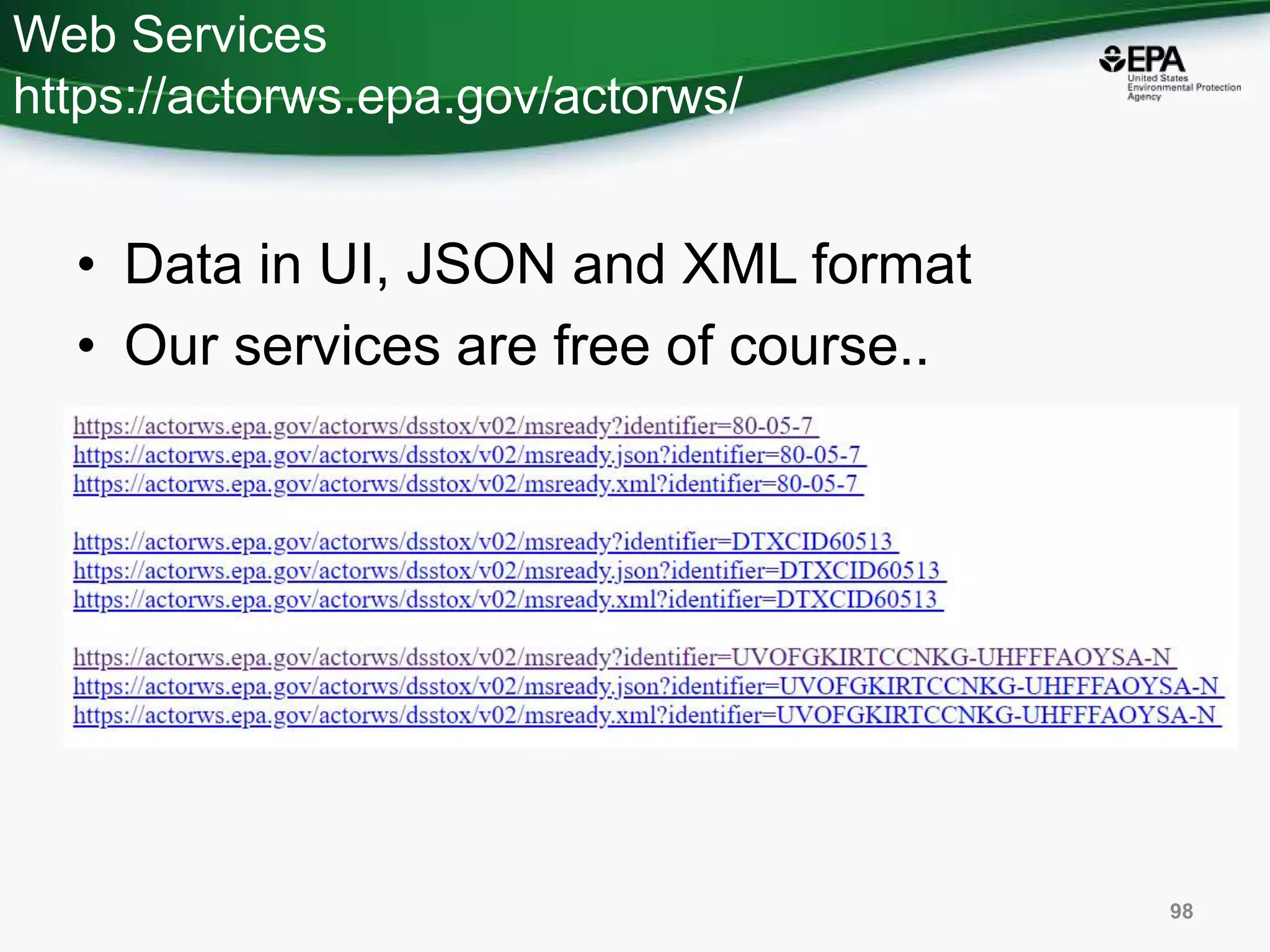
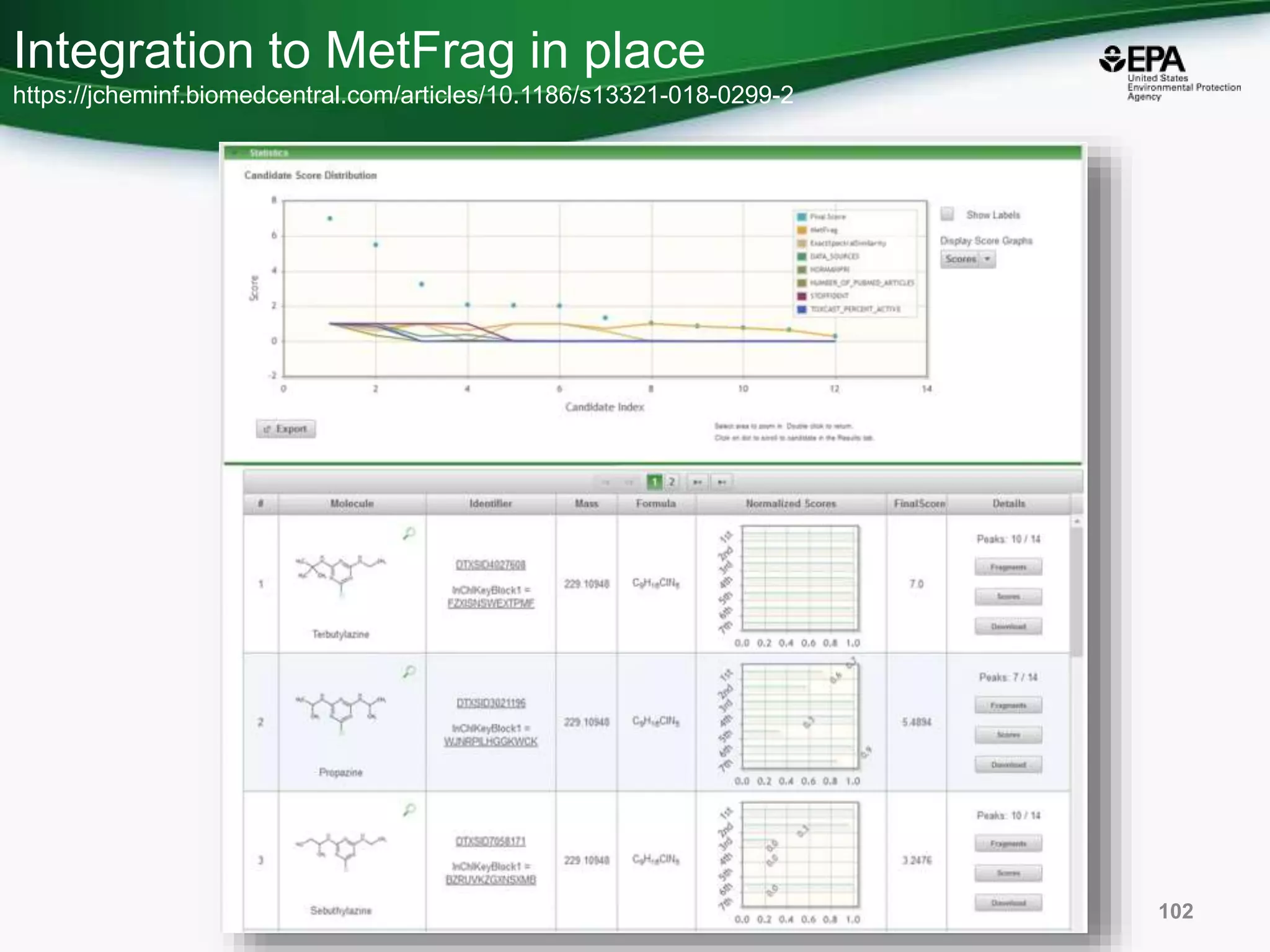
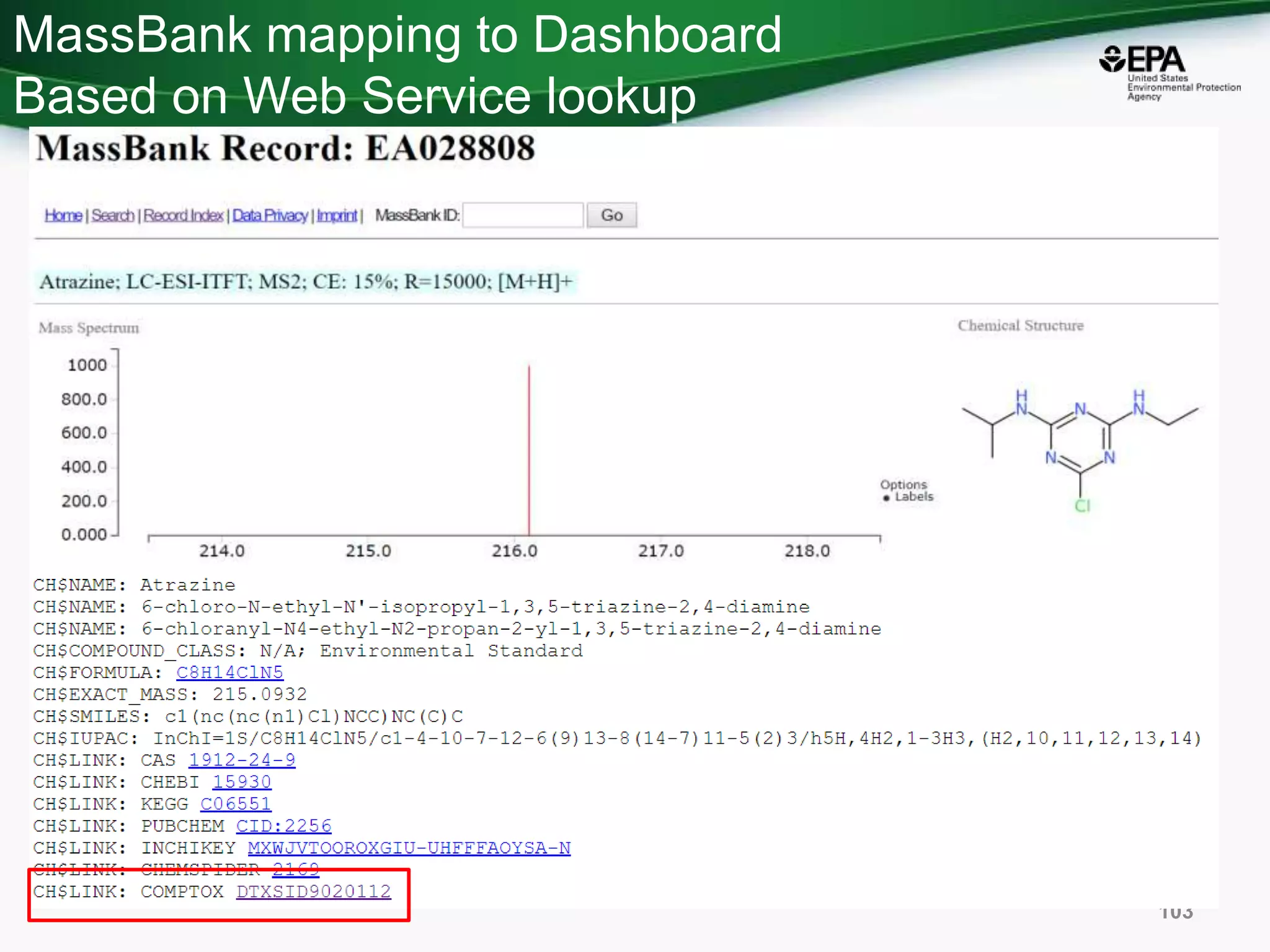
The document presents an overview of the US-EPA CompTox Chemicals Dashboard, focusing on its application in mass spectrometry analysis and the identification of chemical structures. It discusses the dashboard's features, including data quality, batch searching capabilities, and integration of multiple data sources to rank chemicals for toxicity. The presentation also highlights ongoing developments and requests for community collaboration in expanding and curating chemical lists.