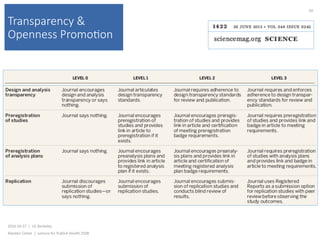
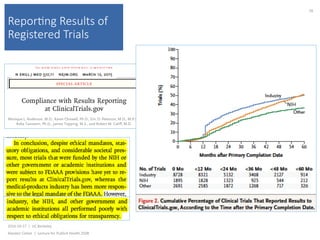
This document summarizes a lecture about transparency and openness in research. It discusses the CONSORT statement, which was developed to standardize the reporting of randomized controlled trials. CONSORT began in the 1990s and has been revised over time, with the 2010 statement providing a checklist of essential items to include in trial reports. The lecture also notes that CONSORT has been expanded to provide guidelines for specifically reporting different types of trials and that hundreds of other reporting guidelines have been developed for other research areas as well.