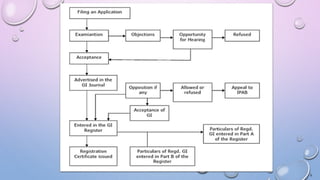
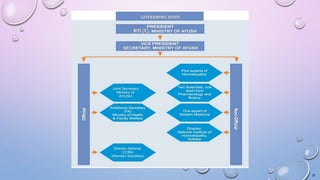
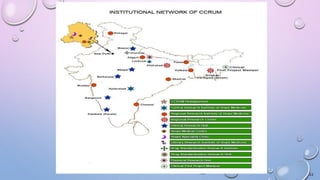
This document provides information on Traditional Knowledge Digital Library (TKDL), Geographical Indications, Central Council for Research in Ayurvedic Sciences (CCRAS), Central Council for Research in Siddha (CCRS), Central Council for Research in Homoeopathy (CCRH), and Central Council for Research in Unani Medicine (CCRUM). It defines each topic, describes their objectives and functions, and lists their research institutes. The document serves as a reference for these traditional medicine organizations and initiatives in India.