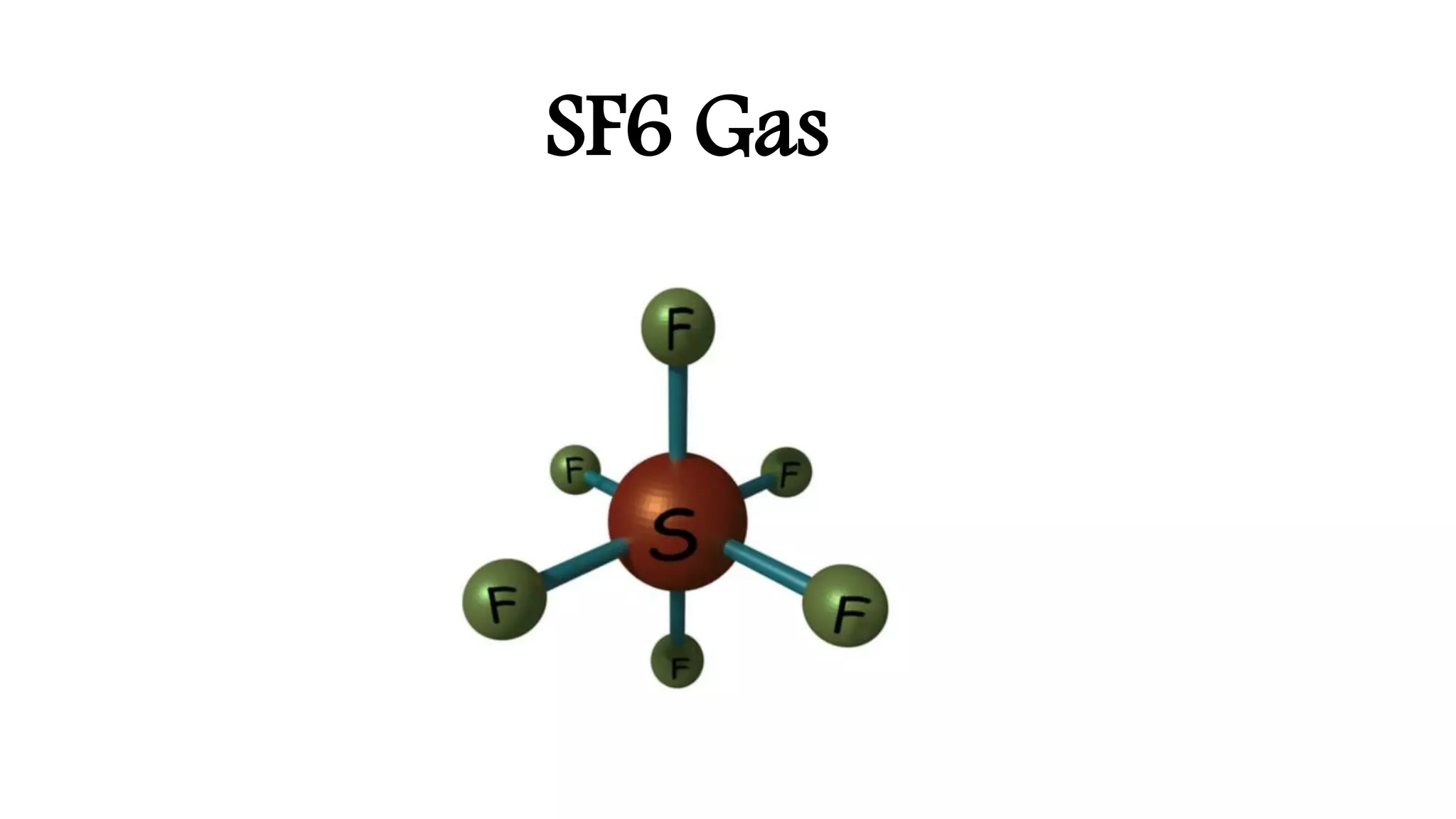
SF6 gas was first synthesized in 1900 and its insulating properties were discovered in the 1930s. It began being used commercially in electrical equipment in the 1940s due to its excellent dielectric properties. SF6 is highly electronegative and stable at high temperatures, allowing it to effectively quench arcs and dissipate heat in equipment like circuit breakers and switchgear. Its non-toxic and non-flammable qualities also make it safer than other insulating gases. SF6 quickly became the preferred gas for insulating high voltage equipment due to these advantages.