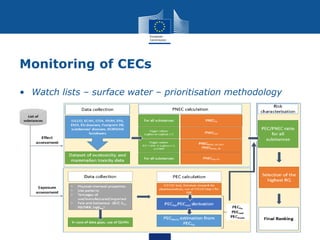
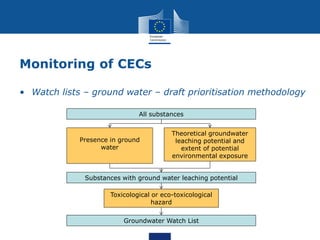
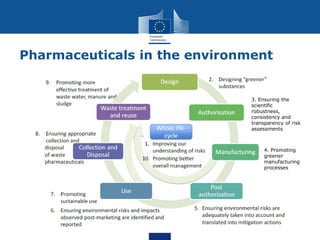
This document summarizes the state of policy development in the EU to manage contaminants of emerging concern (CECs). It discusses the EU's chemicals legislation and monitoring efforts for CECs in surface water and groundwater. The EU is developing watch lists of substances of concern for surface and groundwater. It is also exploring effect-based monitoring methods and using grouping approaches to assess chemical risks. The document notes efforts to increase data access and sharing, as well as increasing focus on persistent and mobile substances with potential for groundwater contamination. It concludes by mentioning the upcoming EU strategic approach on pharmaceuticals in the environment.