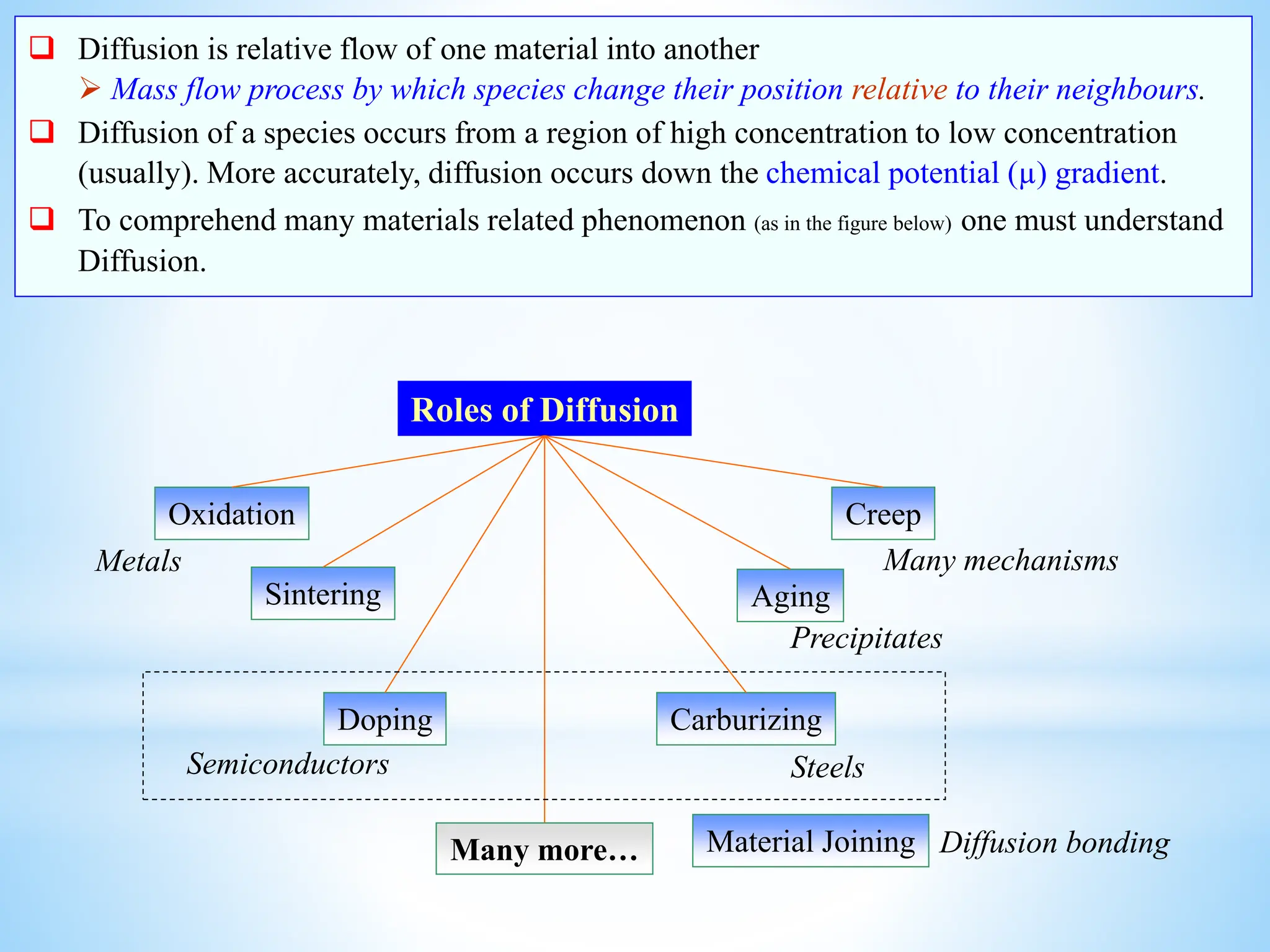
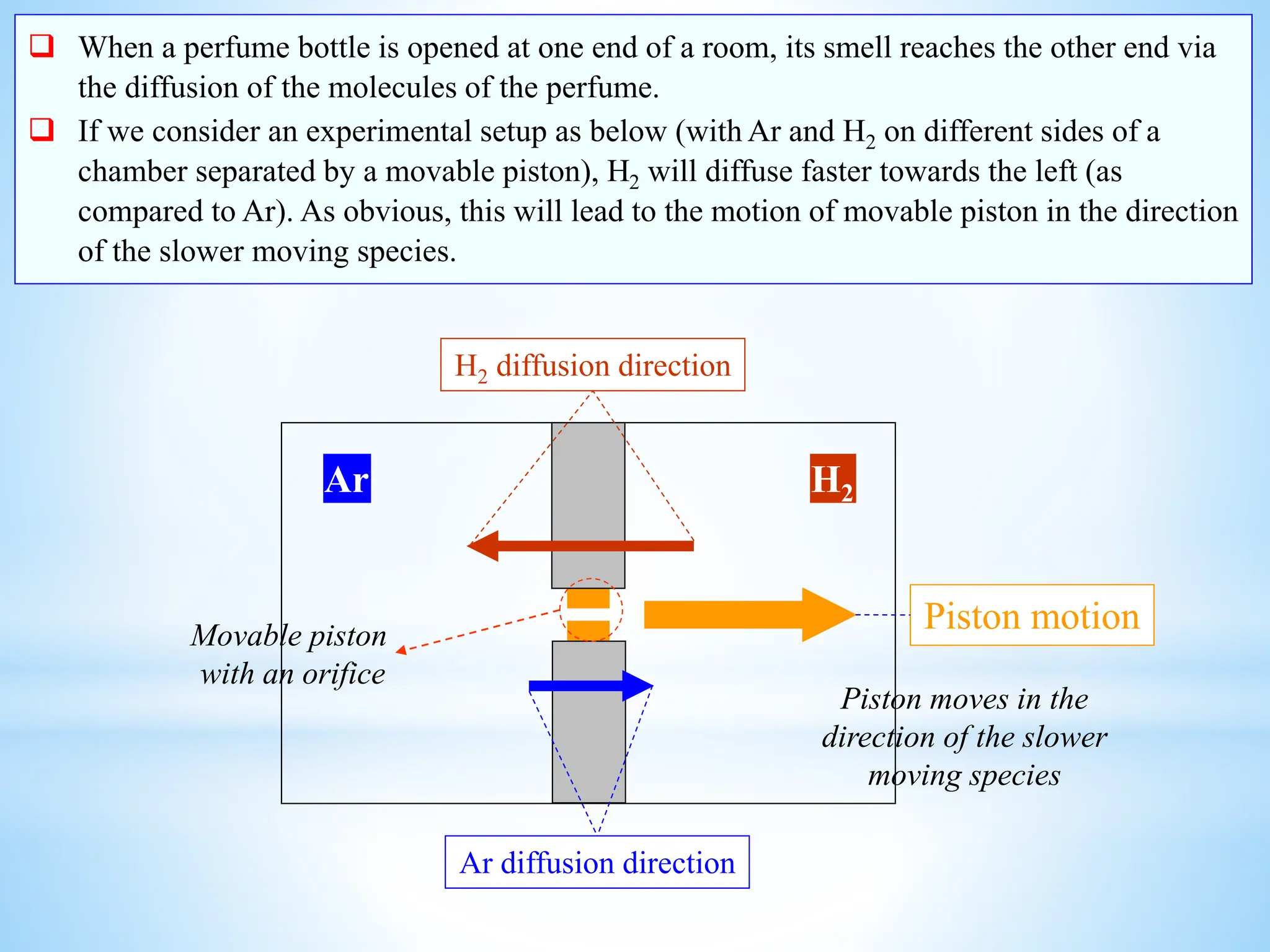
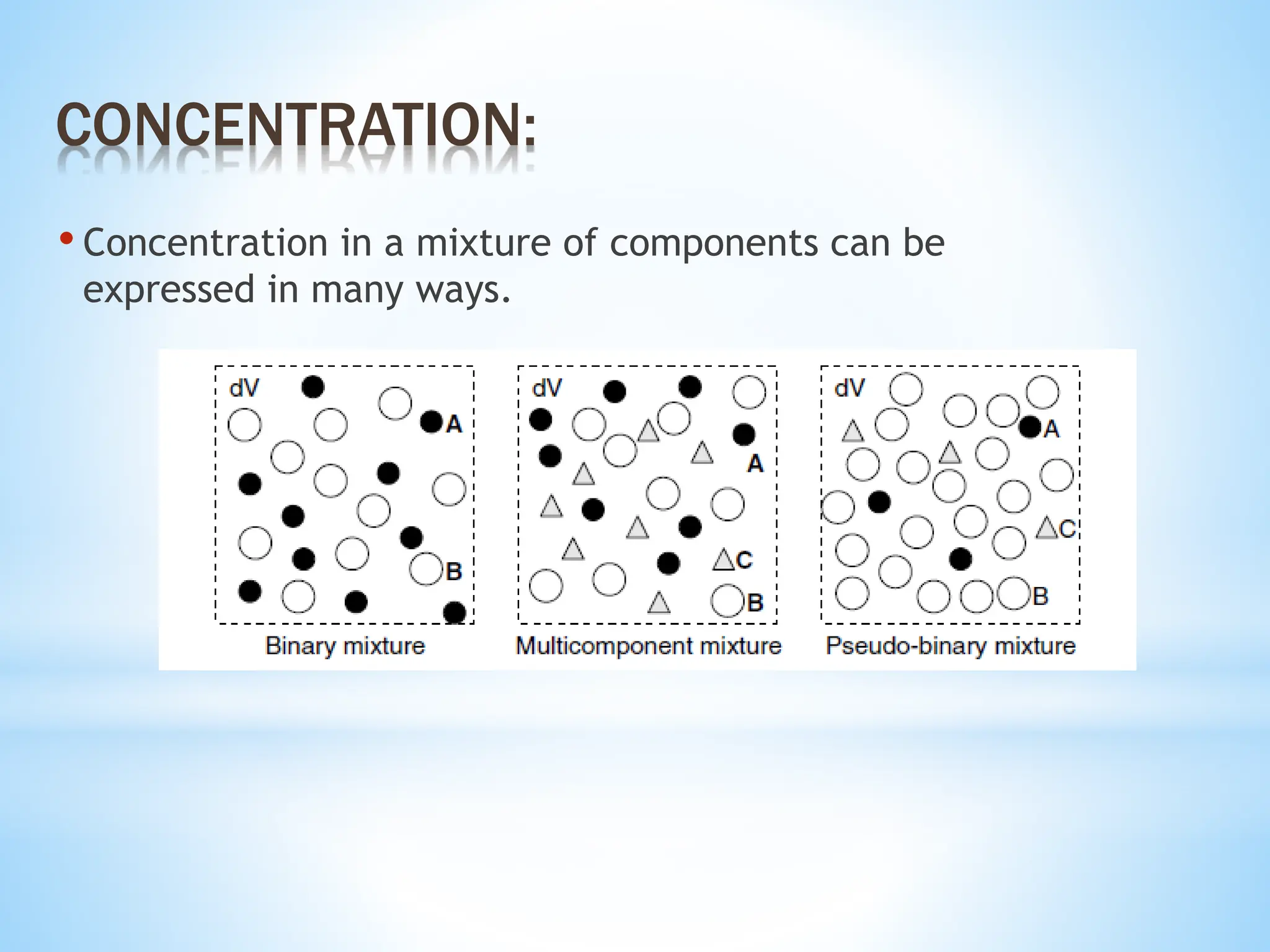
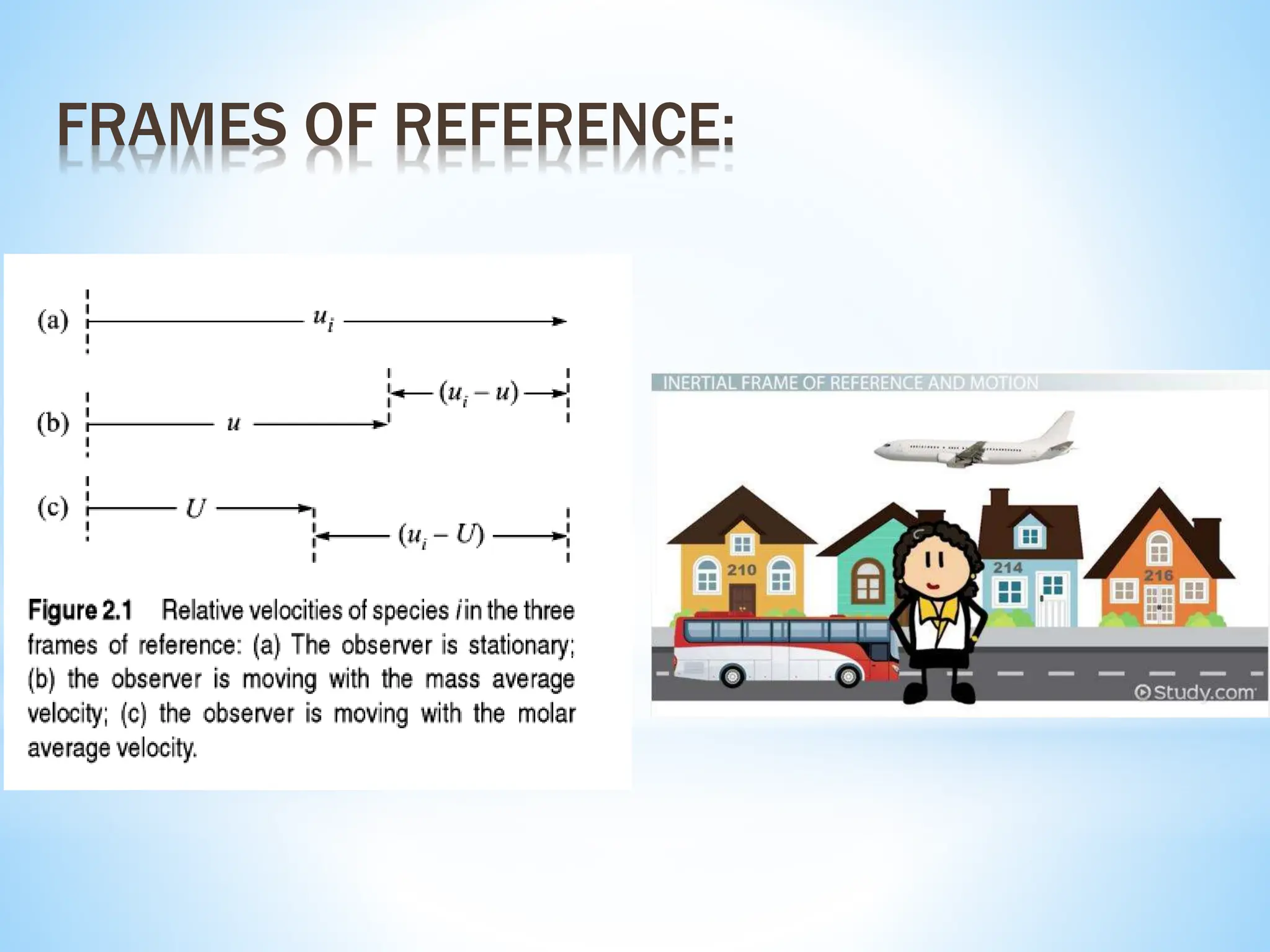
This document discusses mass transfer and diffusion fundamentals. It defines key terms like concentration, concentration gradient, steady state, and different types of concentrations used to measure species in a mixture. Fick's Laws of diffusion are also summarized, stating that the molar flux of a species is proportional to its concentration gradient, with diffusivity being the constant of proportionality. Different frames of reference for observing diffusion and equations for molar and mass flux and average velocities are provided.