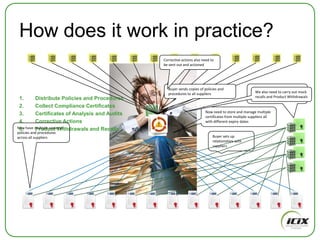
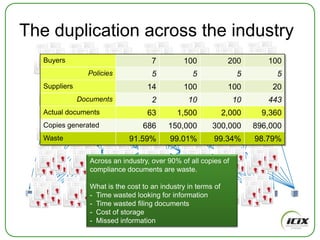
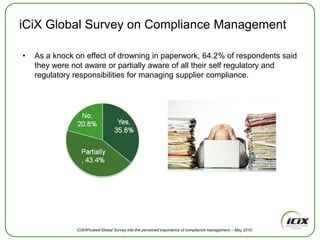
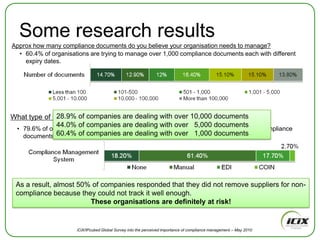
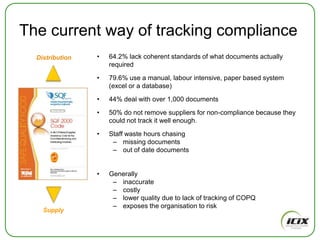
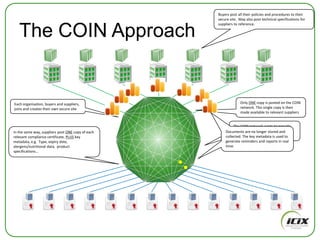
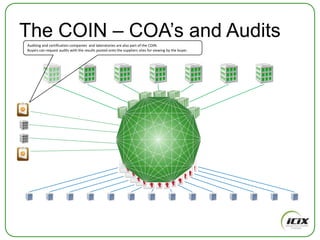
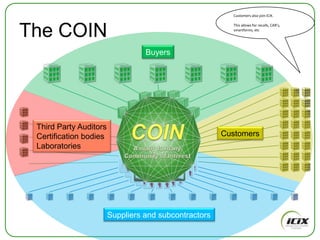
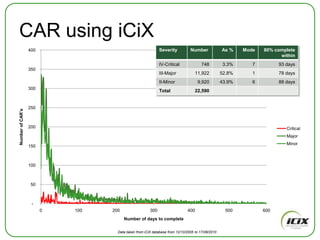
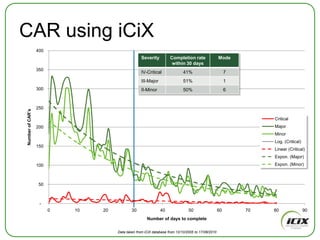
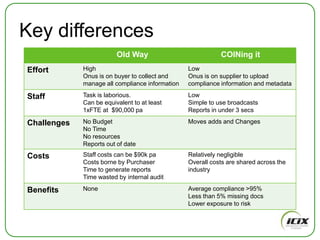
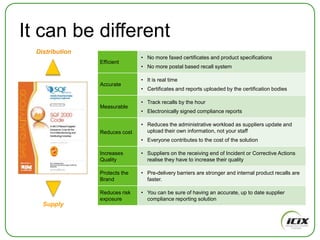
The document discusses how Community of Interest (COIN) networks can be used to more efficiently manage supplier compliance documentation by allowing buyers and suppliers to post policies, procedures, certificates and other documents to a shared network, reducing duplication and improving visibility of compliance information in real-time. It notes that currently most organizations rely on inefficient manual systems to track thousands of compliance documents from numerous suppliers. COIN networks aim to address the challenges of insufficient time, budget and resources that many organizations face in effectively managing supplier compliance.