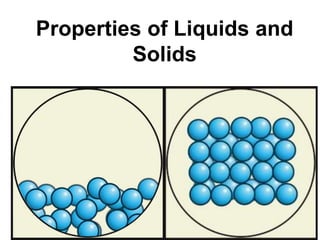
The document discusses how intermolecular forces affect the physical properties of liquids and solids. Specifically, it states that strong intermolecular forces lead to high surface tension, high viscosity, low vapor pressure, and high heat of vaporization. It provides examples of how hydrogen bonding between water molecules contributes to properties like surface tension and boiling point. The document also compares the intermolecular forces and properties of different compounds like glycerol, hexane, and decane.