Presentation1
•Download as PPTX, PDF•
0 likes•96 views
111111111111111111
Report
Share
Report
Share
Recommended
More Related Content
What's hot
What's hot (20)
Chemistry notes class 11 chapter 3 classification of elements and periodicity...

Chemistry notes class 11 chapter 3 classification of elements and periodicity...
Viewers also liked
Viewers also liked (13)
Open Homes for sale in Cheyenne, WY May 11 & May 12, 2013

Open Homes for sale in Cheyenne, WY May 11 & May 12, 2013
10558689 problemas-resueltos-genetica-de-poblaciones

10558689 problemas-resueltos-genetica-de-poblaciones
More from PRASANTH S
More from PRASANTH S (11)
Recently uploaded
Dwarka Sector 26 Call Girls | Delhi | 9999965857 🫦 Vanshika Verma More Our Se...

Dwarka Sector 26 Call Girls | Delhi | 9999965857 🫦 Vanshika Verma More Our Se...Call Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No AdvanceCall Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Rohini Sector 6 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 6 Call Girls Delhi 9999965857 @Sabina Saikh No AdvanceCall Girls In Delhi Whatsup 9873940964 Enjoy Unlimited Pleasure
Recently uploaded (20)
Delhi Call Girls Rohini 9711199171 ☎✔👌✔ Whatsapp Hard And Sexy Vip Call

Delhi Call Girls Rohini 9711199171 ☎✔👌✔ Whatsapp Hard And Sexy Vip Call
Dwarka Sector 26 Call Girls | Delhi | 9999965857 🫦 Vanshika Verma More Our Se...

Dwarka Sector 26 Call Girls | Delhi | 9999965857 🫦 Vanshika Verma More Our Se...
Call Girls Dubai Prolapsed O525547819 Call Girls In Dubai Princes$

Call Girls Dubai Prolapsed O525547819 Call Girls In Dubai Princes$
Call Now ☎ 8264348440 !! Call Girls in Shahpur Jat Escort Service Delhi N.C.R.

Call Now ☎ 8264348440 !! Call Girls in Shahpur Jat Escort Service Delhi N.C.R.
Moving Beyond Twitter/X and Facebook - Social Media for local news providers

Moving Beyond Twitter/X and Facebook - Social Media for local news providers
AlbaniaDreamin24 - How to easily use an API with Flows

AlbaniaDreamin24 - How to easily use an API with Flows
Call Girls In Pratap Nagar Delhi 💯Call Us 🔝8264348440🔝

Call Girls In Pratap Nagar Delhi 💯Call Us 🔝8264348440🔝
Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 26 Call Girls Delhi 9999965857 @Sabina Saikh No Advance
Low Rate Call Girls Kolkata Avani 🤌 8250192130 🚀 Vip Call Girls Kolkata

Low Rate Call Girls Kolkata Avani 🤌 8250192130 🚀 Vip Call Girls Kolkata
Call Girls In Defence Colony Delhi 💯Call Us 🔝8264348440🔝

Call Girls In Defence Colony Delhi 💯Call Us 🔝8264348440🔝
Call Girls In Ashram Chowk Delhi 💯Call Us 🔝8264348440🔝

Call Girls In Ashram Chowk Delhi 💯Call Us 🔝8264348440🔝
VIP Kolkata Call Girl Salt Lake 👉 8250192130 Available With Room

VIP Kolkata Call Girl Salt Lake 👉 8250192130 Available With Room
'Future Evolution of the Internet' delivered by Geoff Huston at Everything Op...

'Future Evolution of the Internet' delivered by Geoff Huston at Everything Op...
Call Girls In Model Towh Delhi 💯Call Us 🔝8264348440🔝

Call Girls In Model Towh Delhi 💯Call Us 🔝8264348440🔝
Rohini Sector 6 Call Girls Delhi 9999965857 @Sabina Saikh No Advance

Rohini Sector 6 Call Girls Delhi 9999965857 @Sabina Saikh No Advance
Challengers I Told Ya ShirtChallengers I Told Ya Shirt

Challengers I Told Ya ShirtChallengers I Told Ya Shirt
Chennai Call Girls Alwarpet Phone 🍆 8250192130 👅 celebrity escorts service

Chennai Call Girls Alwarpet Phone 🍆 8250192130 👅 celebrity escorts service
Chennai Call Girls Porur Phone 🍆 8250192130 👅 celebrity escorts service

Chennai Call Girls Porur Phone 🍆 8250192130 👅 celebrity escorts service
On Starlink, presented by Geoff Huston at NZNOG 2024

On Starlink, presented by Geoff Huston at NZNOG 2024
Presentation1
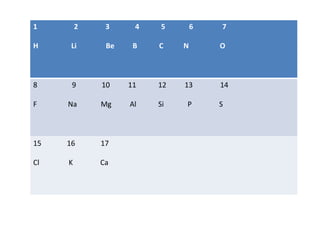
- 1. 1 2 3 4 5 6 7 H Li Be B C N O 8 9 10 11 12 13 14 F Na Mg Al Si P S 15 16 17 Cl K Ca
- 3. LAW OF OCTAVES When elements were arranged in the accenting order of their atomic masses , each eighth elements had similar properties as those of the first
- 4. Li 7 Na 23 K 39 Ca 40 Sr 87.6 Ba 137.3 Cl 35.5 Br 80 I 127
- 6. TRIADS The elements can be classified in groups of three on the basis of similarities of properties is known as triads.