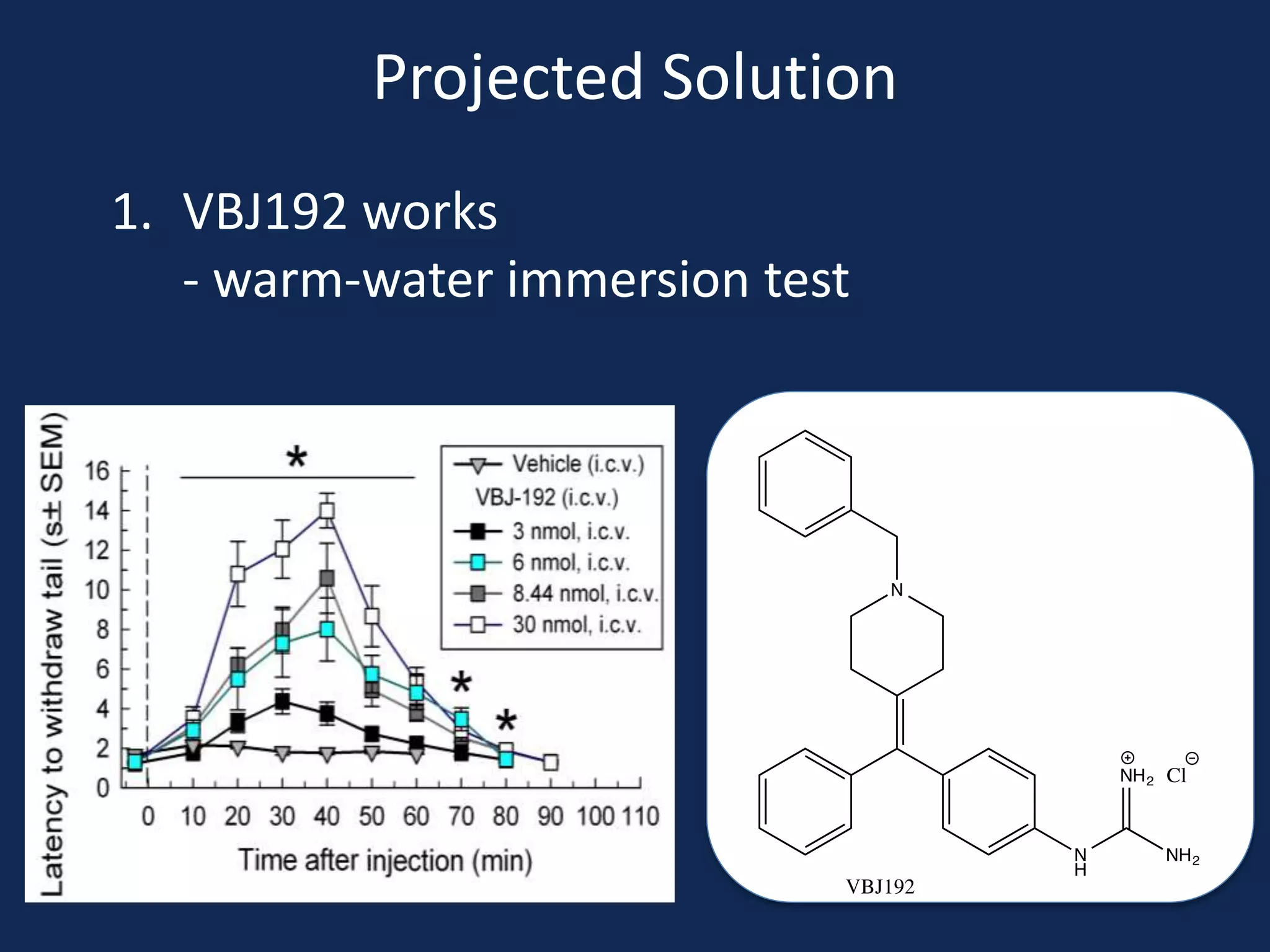
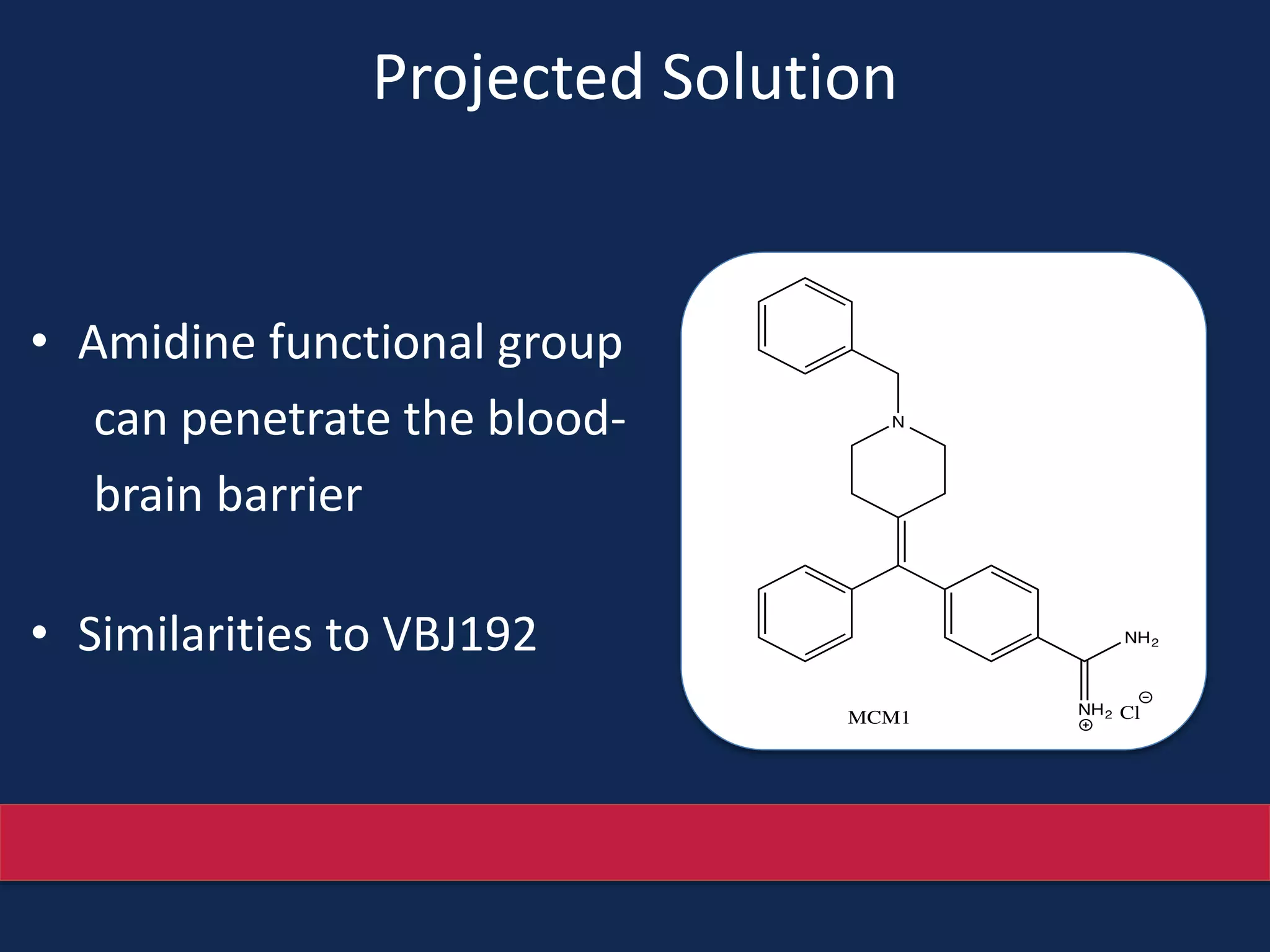
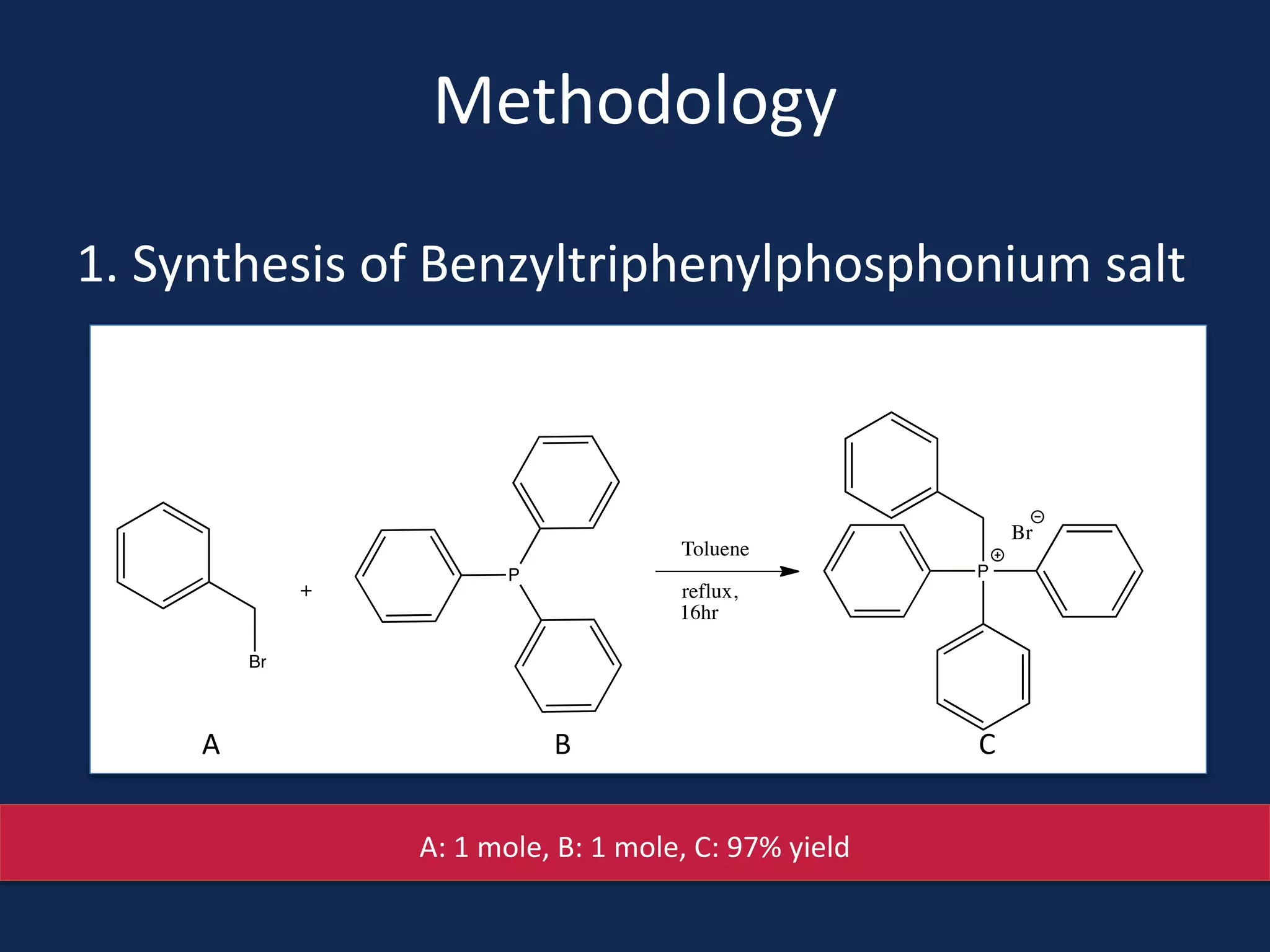
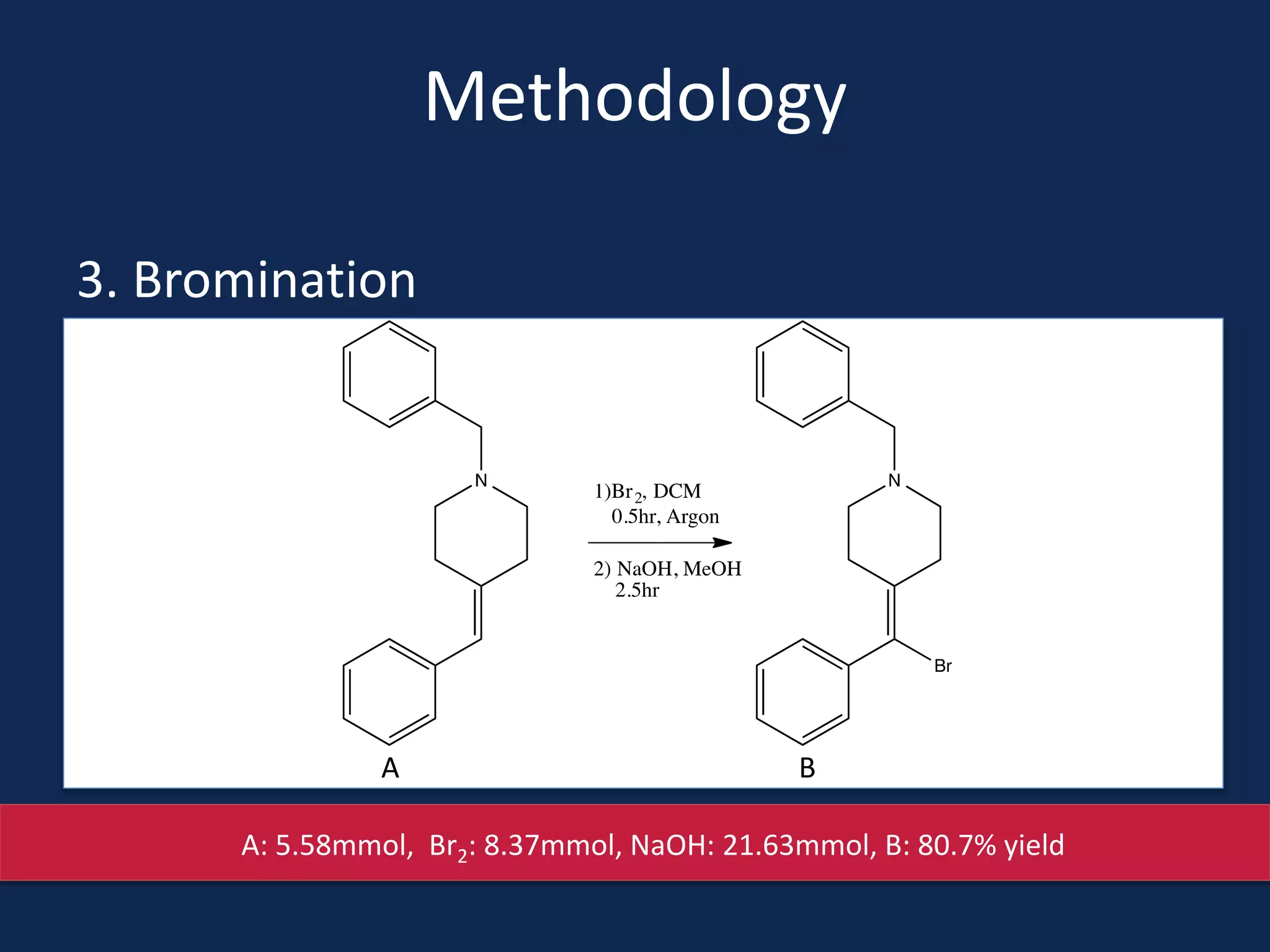
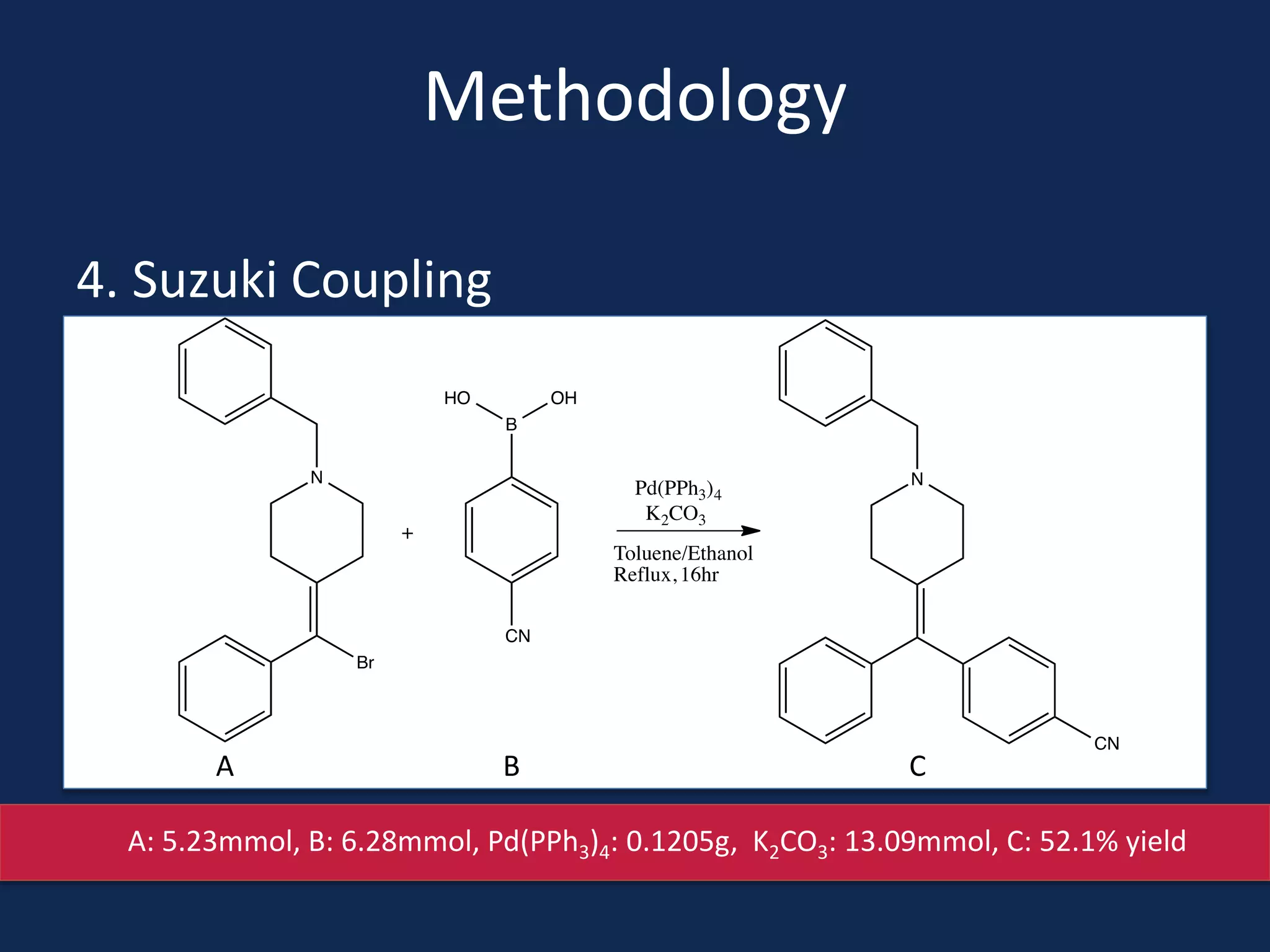
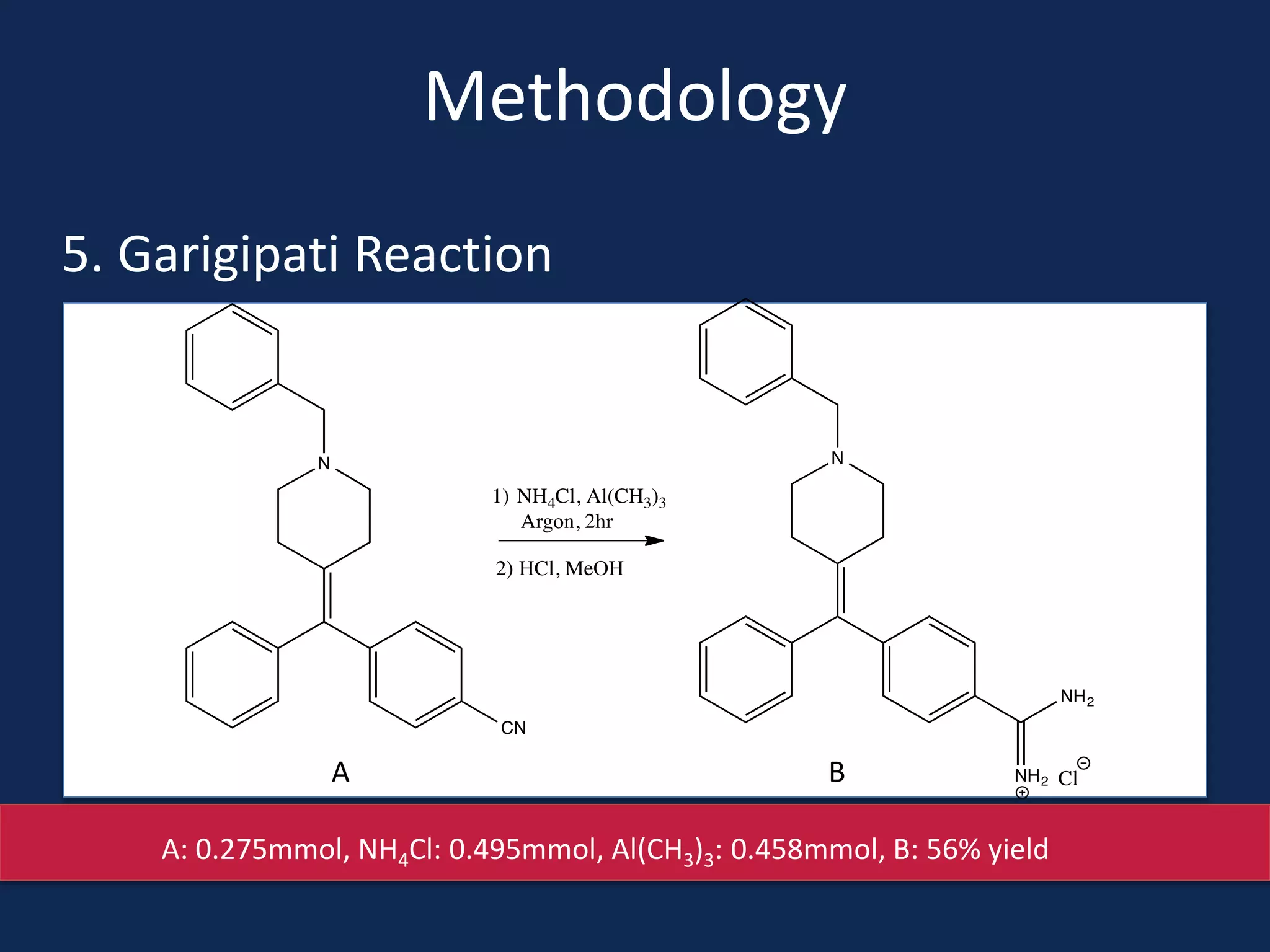
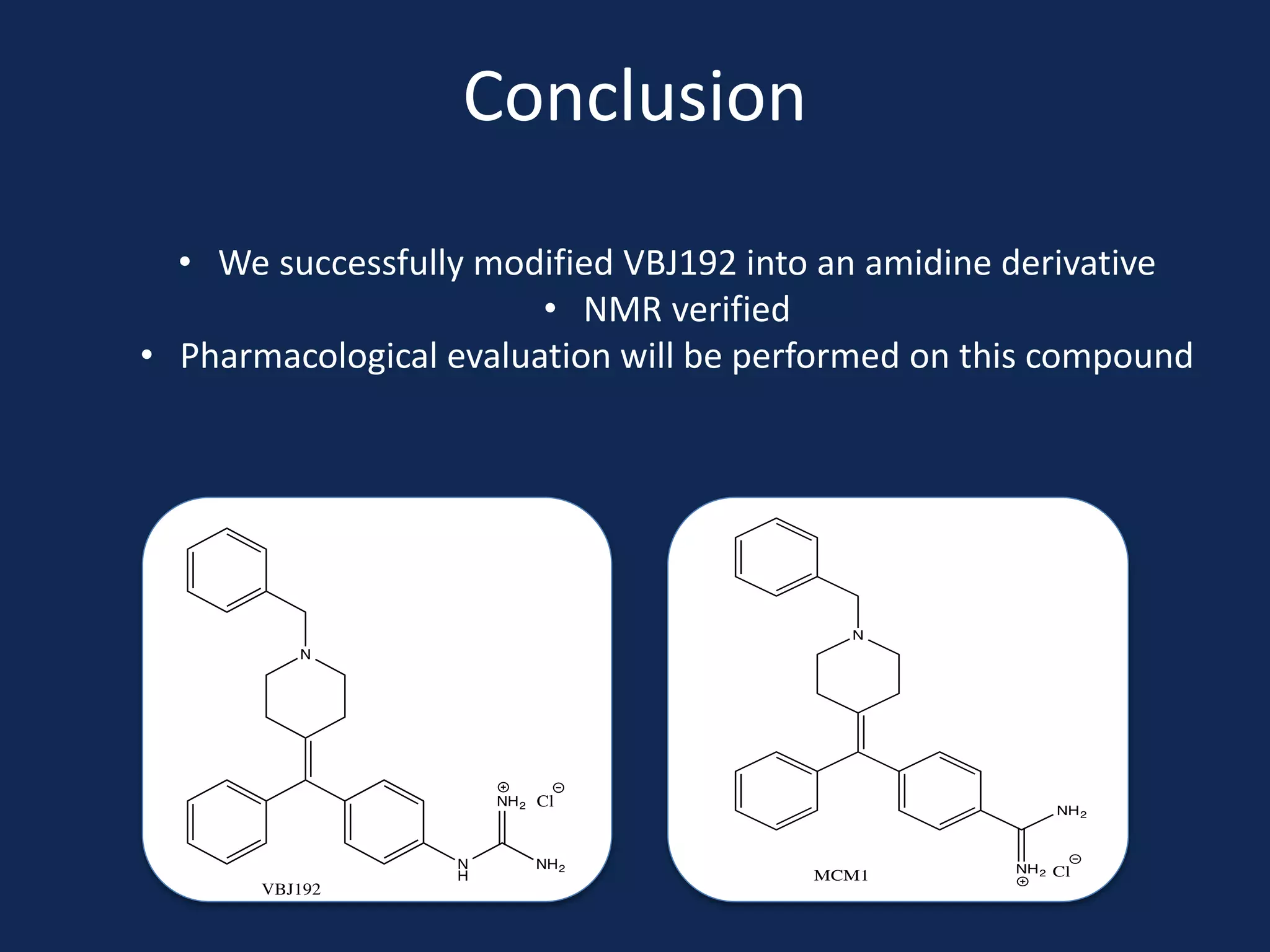
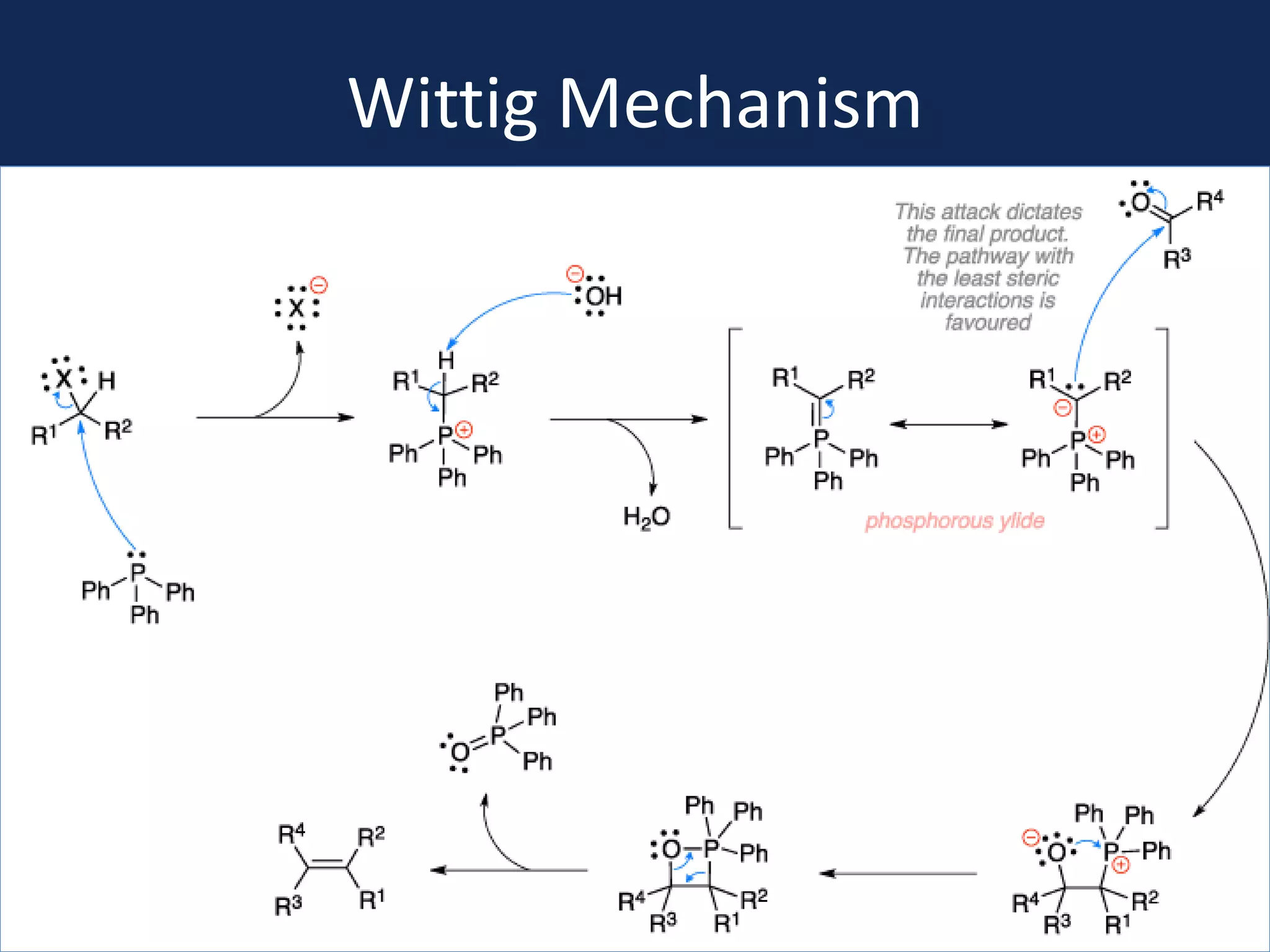
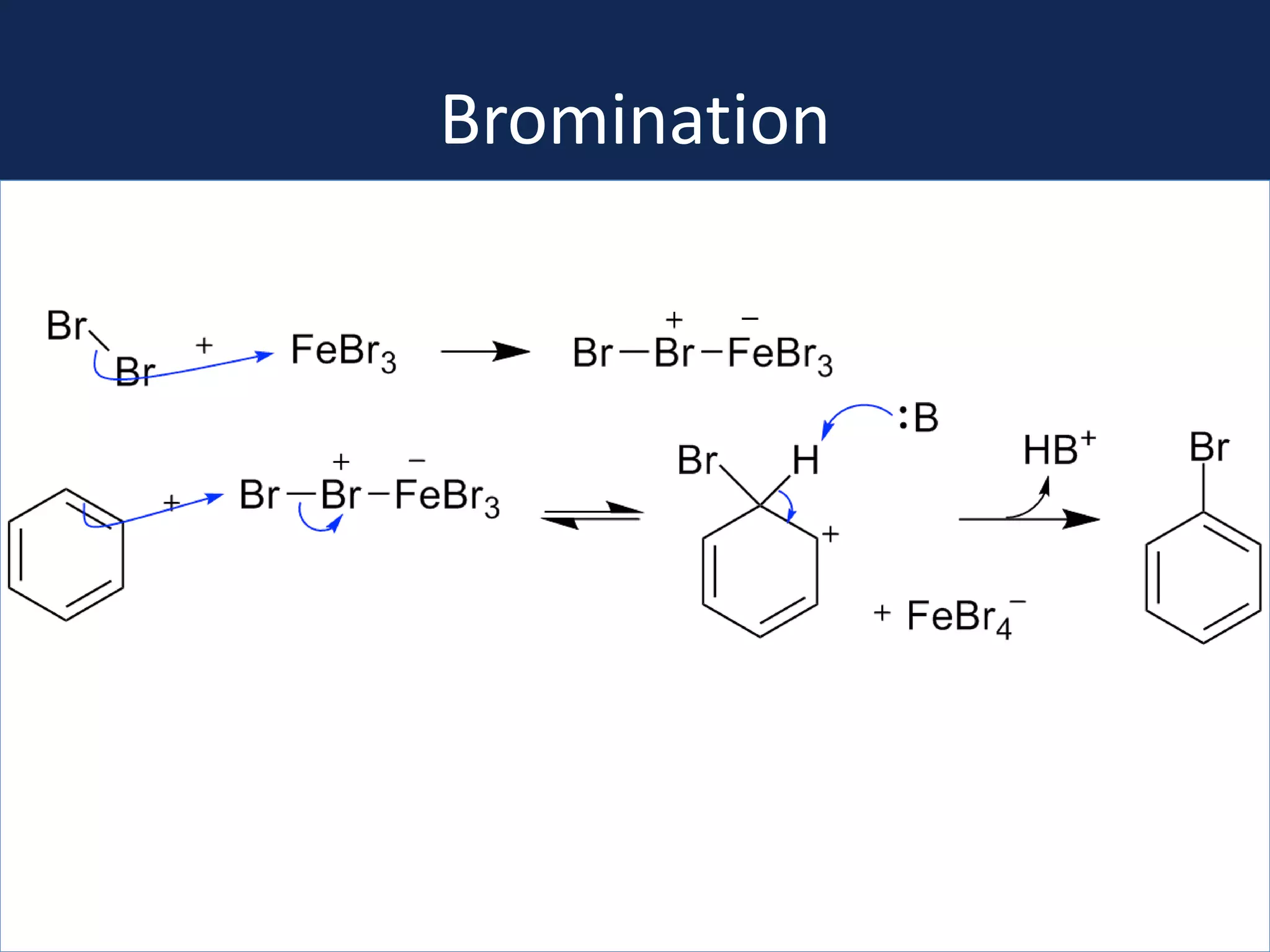
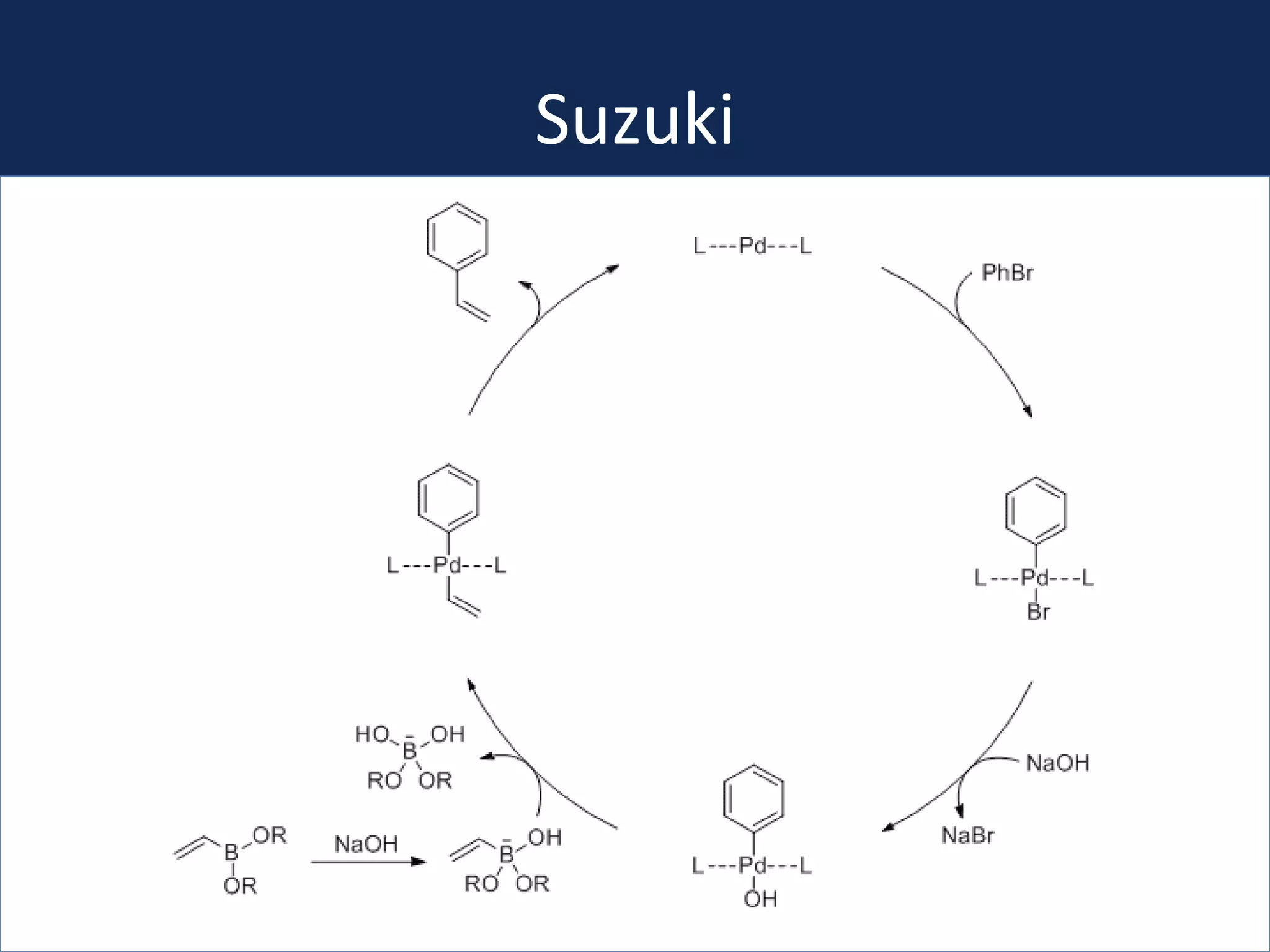
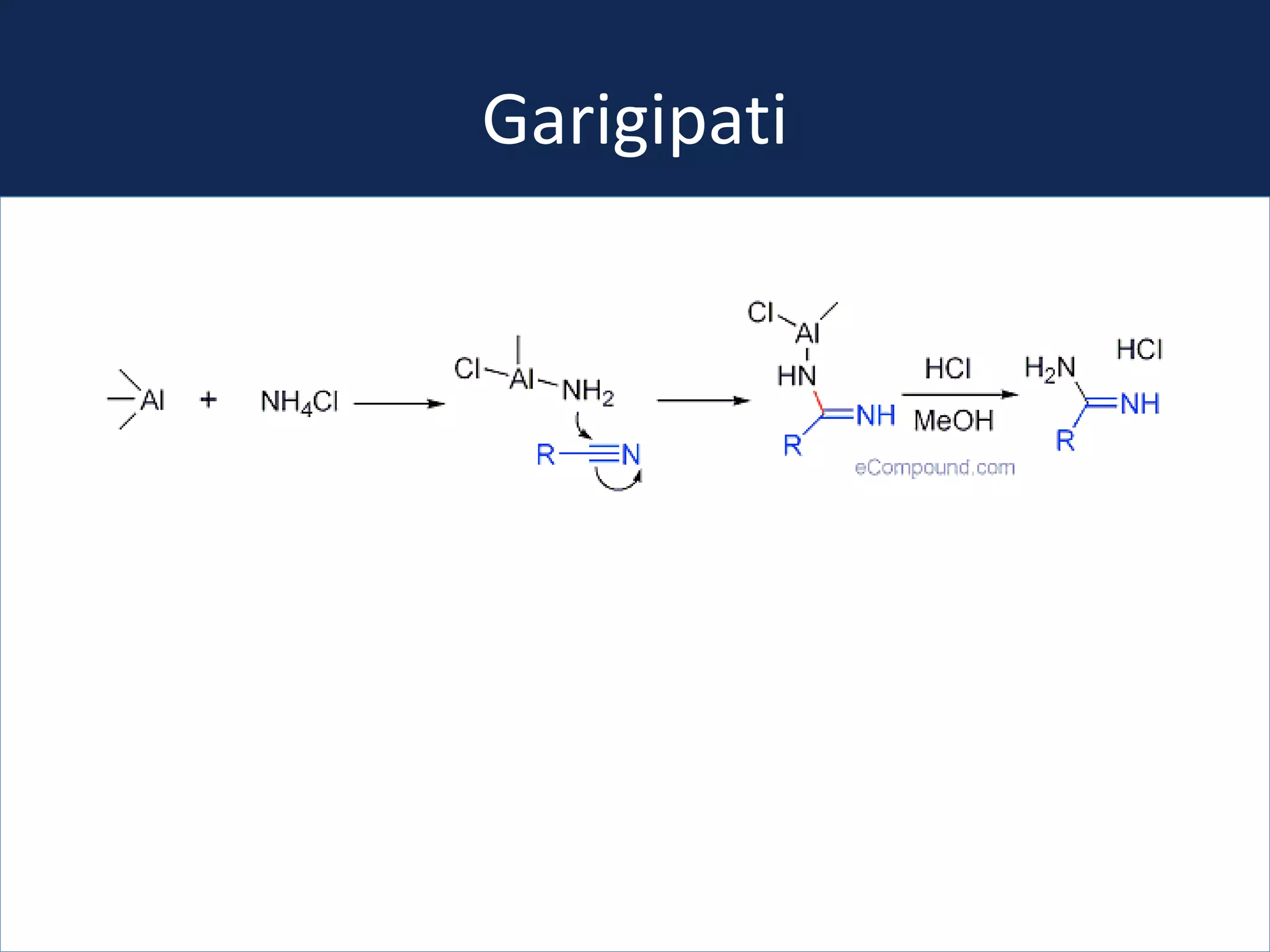
This document summarizes the synthesis of a novel compound with potential use as a painkiller with reduced side effects. The compound is a dual-acting ligand for opioid and neuropeptide FF receptors. It was synthesized in 5 steps: 1) a Wittig reaction, 2) bromination, 3) Suzuki coupling, 4) Garigipati reaction, and 5) purification via filtration and chromatography. Characterization using NMR and mass spectrometry confirmed the successful modification of an existing compound, VBJ192, into an amidine derivative for further pharmacological evaluation and testing as a painkiller with fewer side effects than traditional opioids.