pharmaceutical organic chemistry enantiomeers
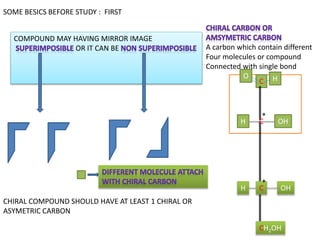
- 1. SOME BESICS BEFORE STUDY : FIRST COMPOUND MAY HAVING MIRROR IMAGE OR IT CAN BE A carbon which contain different Four molecules or compound Connected with single bond C O H C OH H H OH C CH2OH * * CHIRAL COMPOUND SHOULD HAVE AT LEAST 1 CHIRAL OR ASYMETRIC CARBON
- 2. CH3 CH3 Br H NOTE:INTERNAL COMPENSTATION MEANS OPTICAL INACTIVE COMPOUND WHICH IS OF MESOTYPE MESO TYPE COMPOUND ARE THOSE WHICH CONTAIN MORE THAN ONE OPTICALLY ACTIVE CENTER WHICH CANCLES EACH OTHER ACTIVE Br H CH3 H Br * * H Br CH3 MIRROR * *
- 3. COMPOUND NOT HAVING CHIRALITY CENTER BUT ARE ENANTIOMERIC ATROPISOMERISM
- 4. RACEMIC MIXTURE C CH2CH3 CH3 H HO C CH2CH3 CH3 H OH (S) (+) -BUTAN -2-OL A RECEMIC MIXTURE CONTAIN EQUAL AMOUNT OF TWO ENANTIOMERS [α]D=0. EQUAL QUANTITIES OF d AND l ENANTIOMERS. NOTATION= (dl) (+-) NO OPTICAL ACTIVITY THE MIXTURE MAY HAVE DIFFERENT BOILING POINT MELTING POINT FROM ENANTIOMERS (S) (+) -BUTAN -2-OL
- 5. PROPERTIES OF ENANTIOMERS THEY HAVE IDENTICAL PHYSICAL PROPERTIES BUT DIFFER IN DIRECTION OF THE ROTATION OF PLANE POLARIZED LIGHT 2-METHYL-1-BUTANOLS ENANTIOMERS SPECIFIC ROTAYION B.P REF.INDEX (+) +5.7S0 402K 1.41 (-) -5.7S0 402K 1.41 THE MAGNITUDE OF ROTATION OF POLARIZED LIGHT IS ALSO SAME BUT IT IS IN OPPOSITE DIRECTION
- 6. AS THE 4TH PRIORITY CONTAINING MOLECULE ON THE HORIZONTAL POSITION IT WILL CONVERT IT INTO OPPOSITE CONFIGURATION FOR EXAMPLE ……. C CH2OH CHO H HO C CH2 OH CHO H HO 90. ROTATION 90. ROTATION C CH2 OH CHO H HO (+) (+) (-)
- 7. C CH2OH CHO H HO CHO OH CH2OH H 1 4 2 3 In this structure there is OH which we should give 1st priority due to O which Has higher atomic number now second position should Give to CHO and 3rd position Give to the CH2OH and H Has last priority due to Lowest atomic number AS THE 4TH PRIORITY CONTAINING MOLECULE ON THE HORIZONTAL POSITION IT WILL CONVERT IT INTO OPPOSITE CONFIGURATION S(-) configuration but As we see least priority Containing group on the Horizontal position it Will convert into the r(+) Configuration If it least priority containing Group on the vertical line It was stay as s(-) configuration H HE LI BE B C N O F NE NA MG AL SI P S CL AR K CA SC TI V CR MN FE CO NI CU
- 8. Now ONE EXCHANGE OF GROUP C CH2 OH CHO H HO (+) C CH2 OH CHO H HO (-) One exchange of group C CH2 OH CHO H HO (+)
- 9. PROJECTION FORMULA FOR CHIRAL MOLECULE 1. Fischer projection 2 .Newman projection 3 .Sawhorse projection 4 .Flying wedge projection Fischer projection : ALWAYS IN VERTICAL POSITION C CH2OH OH H COOH BELOW THE PLAN OR AWAY FROM YOU ABOVE THE PLAN OR NEAR TO YOU
- 11. Above Below 180 degree or 60 degree 90 degree