Embed presentation
Download to read offline
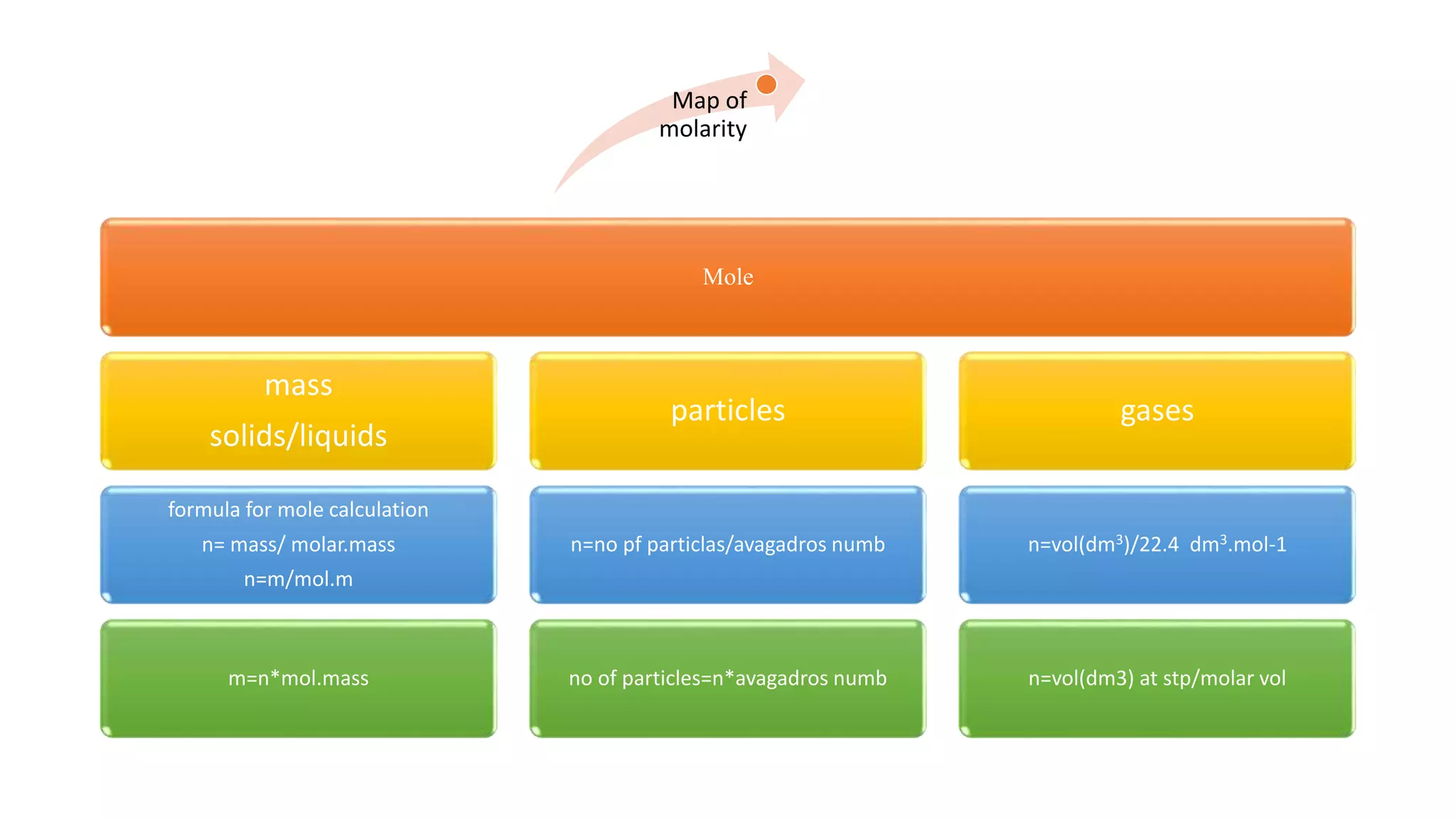


The document discusses molarity and calculations involving moles, molar mass, and number of particles. It provides examples of calculating the molecular masses of SO2 and caffeine based on given atomic masses. It also gives an example of calculating the number of moles in 5.68 g of iron using the formula n=mass/molar mass, finding the number of moles to be 0.1 mol.


