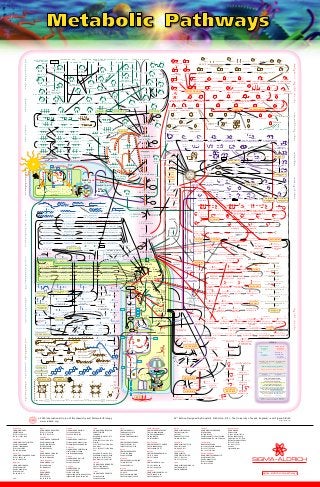
This document provides a detailed diagram of metabolic pathways in photosynthesis and cellular respiration. It depicts the major reactions, enzymes, and reactants/products involved in light-dependent and light-independent reactions of photosynthesis, the citric acid cycle, electron transport chain, and oxidative phosphorylation. Arrows show the direction of reactions and transport of electrons, protons, and ATP.