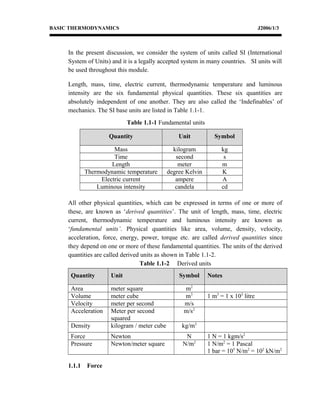
This document provides an introduction to basic thermodynamics concepts including units, dimensions, and conversions. It defines fundamental and derived physical quantities and their SI units. Examples are provided to demonstrate calculating force, pressure, work, power, and density using the appropriate units and conversion factors. The document also discusses dimensional homogeneity and using unit conversions and prefixes to change between units of the same physical quantity. Multiple practice problems are given for students to test their understanding of applying concepts of units, dimensions, and conversions.