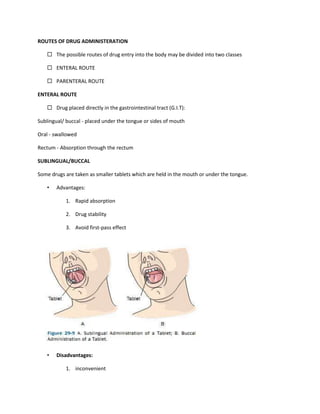
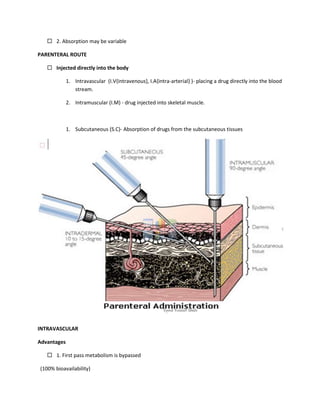
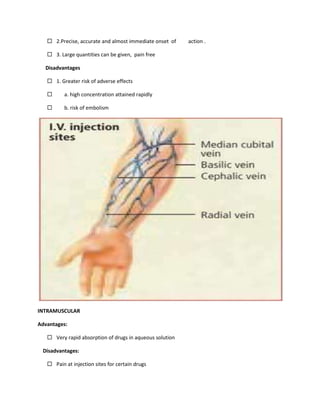
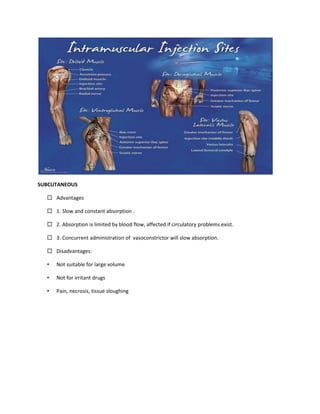
This document provides an introduction to pharmacology. It defines key terms like efficacy, potency, therapeutic index, and adverse drug reactions. It describes pharmacokinetic principles such as absorption, distribution, metabolism and elimination of drugs. It also discusses pharmacodynamics concepts like agonists, antagonists, and drug-receptor interactions. The document outlines various drug classifications and therapeutic categories. It discusses routes of drug administration including enteral and parental routes. It also covers topics like drug interactions, drug labeling, and patient records.