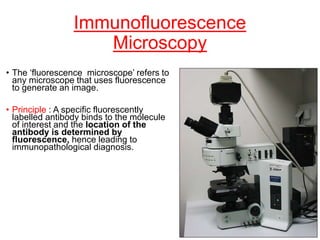
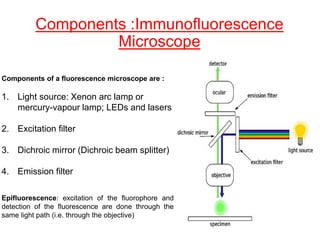
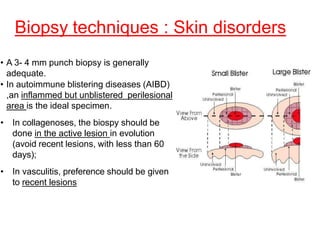
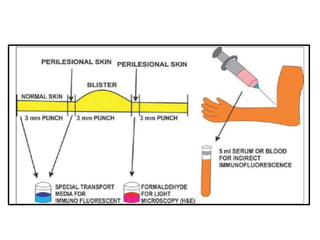
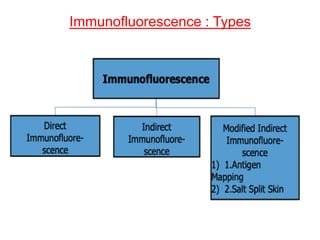
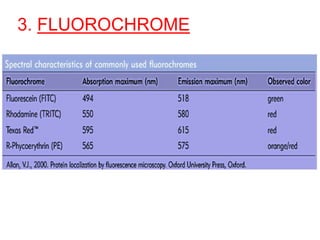
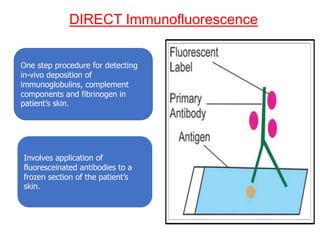
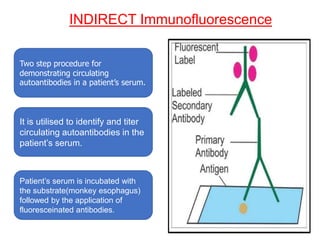
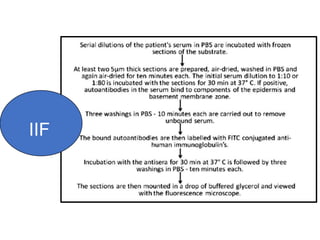
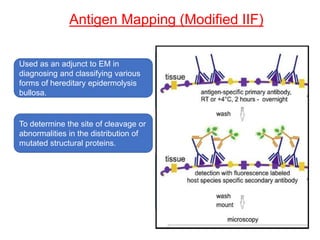
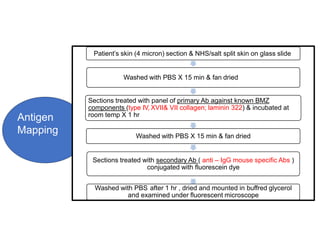
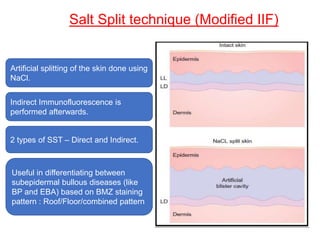
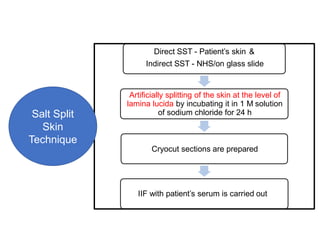
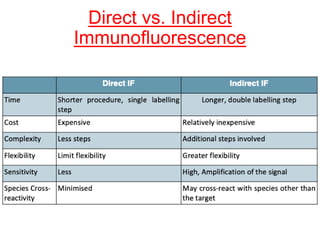
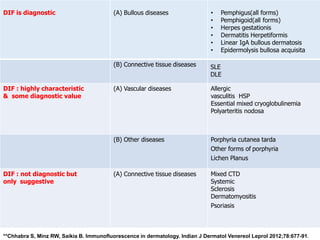
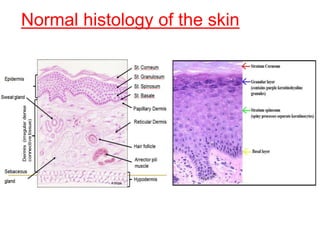
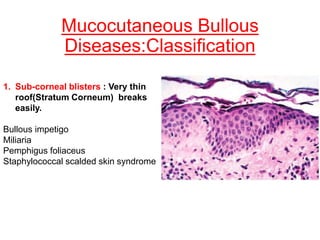
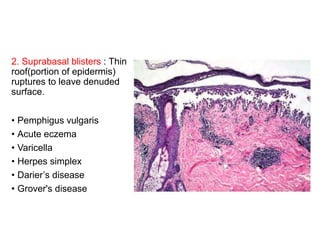
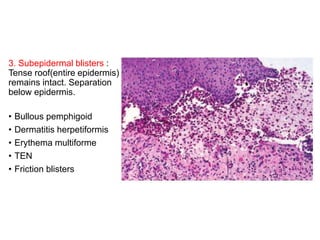
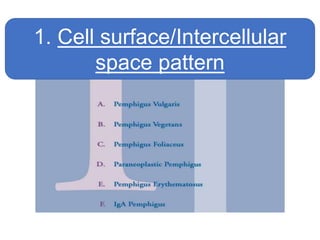
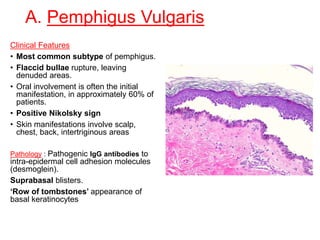
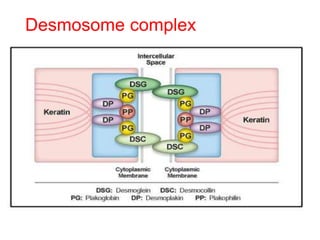
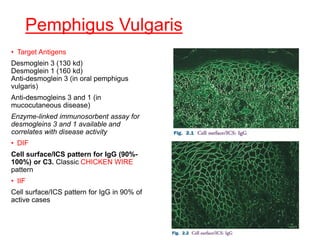
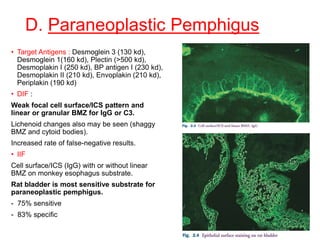
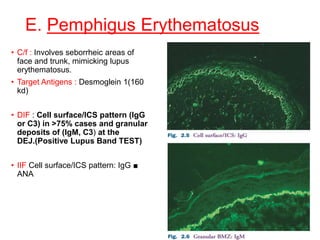
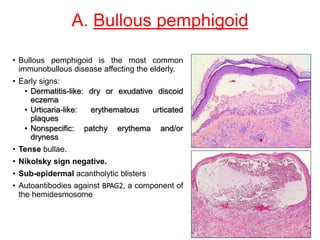
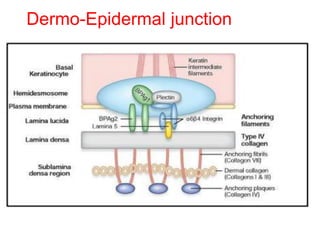
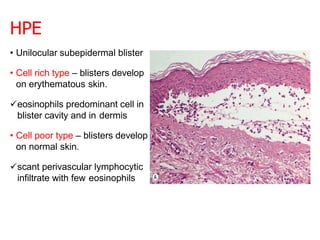
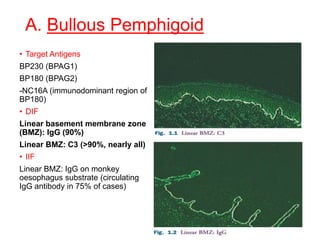
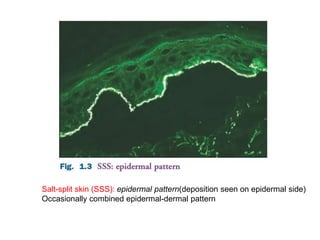
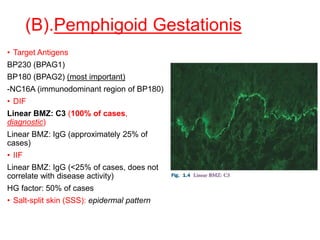
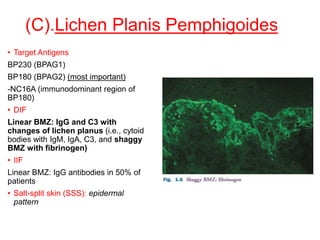
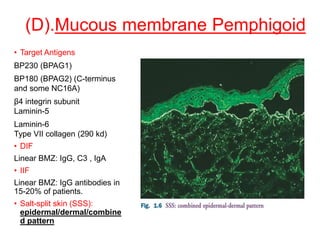
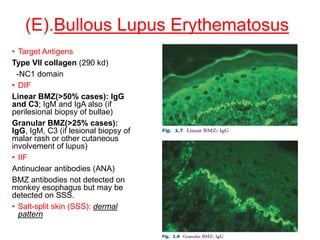
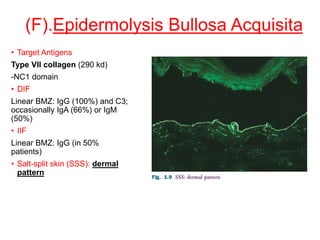
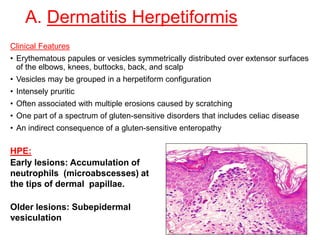
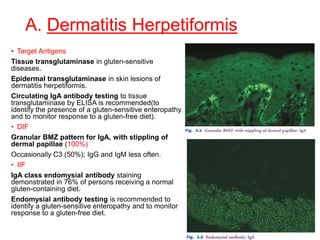
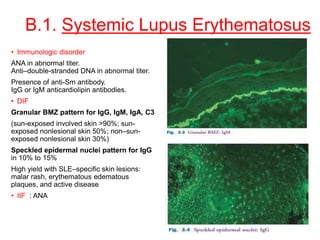
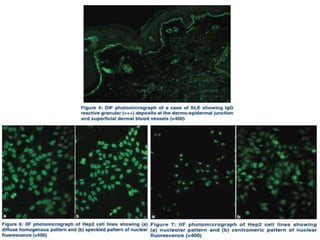
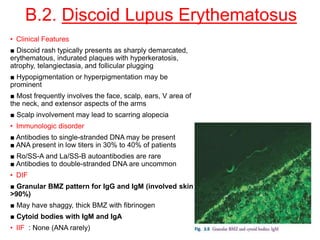
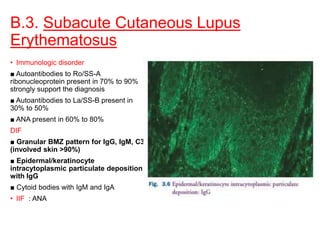
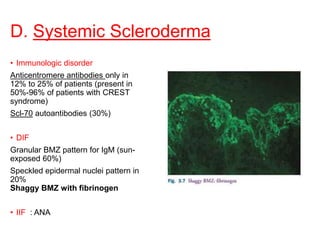
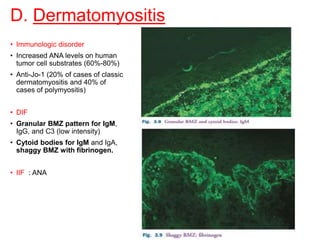
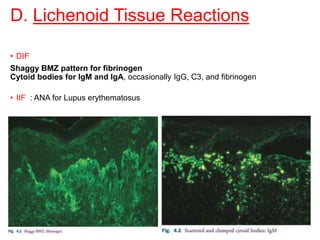
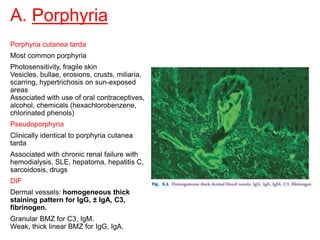
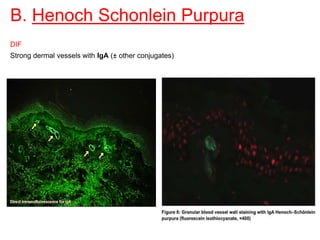
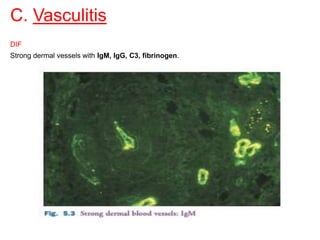
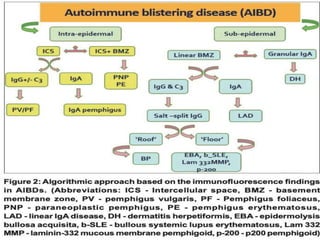
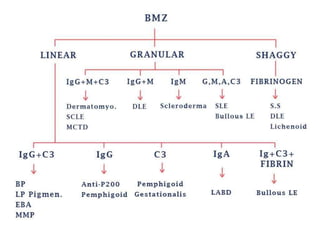
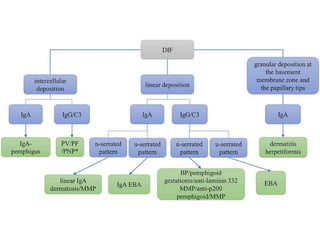
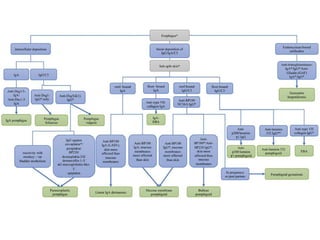
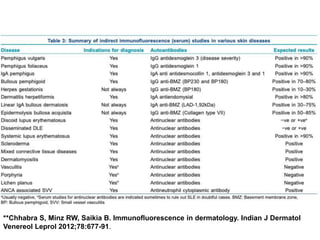
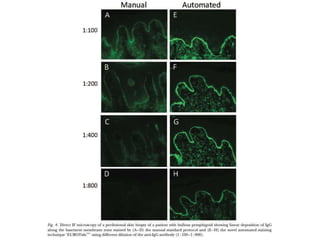
This document provides information about immunofluorescence techniques used in dermatopathology. It begins with the history and components of immunofluorescence microscopy. It then discusses the various applications of immunofluorescence including in skin, muscle, and renal biopsies. Specific techniques for skin biopsies like direct immunofluorescence, indirect immunofluorescence, antigen mapping, and salt split skin technique are explained. Common staining patterns seen in dermatological conditions like pemphigus vulgaris, pemphigus vegetans, pemphigus foliaceus, and paraneoplastic pemphigus are summarized. The document emphasizes the utility of immunofluorescence in diagnosing and classifying autoimmune bullous diseases and connective tissue disorders.