heavy Metal toxicology in environment science
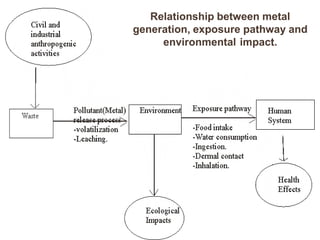
- 1. Relationship between metal generation, exposure pathway and environmental impact.
- 2. BIOACCUMULATION OF HEAVYMETALS Pollutants like heavy metals are CONSERVATIVE pollutants – i.e. they aren’t broken down by bacteria etc and are effectively permanent Most plants and animals can regulatetheir metal content to a certain point-but metals that can’t be excreted buildup in an organism over its lifetime
- 3. *short note BIOACCUMULATION Bioaccumulation is essentially the buildup of contaminants such as heavy metals or pesticides in living organisms
- 4. BIOMAGNIFICATION those animals feeding on bioaccumulators gain even higher inputs of contaminants and bioaccumulate even greater concentrations and so on.. with animals at the highest trophic level obtaining highest concentrations *short note
- 6. SPECIATION ANDTOXICITY OF HEAVY METALS Poisonous in their cations form Highly toxic when bonded to short chains of carbon atoms ex. Alkyl groups with mercury and lead attached
- 8. LEAD (Pb) Its atomic number is 82, atomic weight is 207.2g/mol Bluish-white in color. It is very soft, highly malleable, ductile and a relatively poor conductor of electricity. It melts at 327oC and its boiling point is 1750oC. Lead has low water solubility, is very resistant to corrosion but tarnishes upon exposure to air. Lead is a highly toxic trace element with no recognized biological requirement in organisms.
- 9. OCCURRENCE OF LEAD Lead occurs naturally in the earth crust, usually in ores such as: Galena (PbS) (majorore) Cerrusite(PbCO3) Anglesite(PbSO3) Lanarkite(PbO·PbSO3)
- 10. ENVIRONMENTAL SOURCES/USES OFLEAD Soundproofing in buildings Solder- alloy of lead and tin lining pipes, tanks, X-ray apparatus Weather-proofing buildings Protective shielding for radioactive material Smelters Sheathing electric cables Additives for gasoline Ammunitions Lead in solder to seal tin cans
- 11. ENVIRONMENTAL SOURCES/USES OFLEAD Lead in automobile batteries As additives in gasolines tetraethyl lead Pb(C2H5)4 and tetramethyl lead Pb(CH3)4 Lead salts as glazes in pots (PbO) Lead salts as pigments PbCrO4 -paints applied to school buses and yellow stripes on road Pb3O4 -as corrosion-resistant paints and has a bright red color Pb(CH2COOH)2 -used in preparations to cover gray hair Pb (CO ) (OH) -white lead
- 12. EFFECTS OF LEAD IN HUMAN REPRODUCTION AND INTELLIGENCE • Pb2+ replaces Ca2+ inbones • Pb2+ remains in bones for decade • High levels of inorganic Pb2+ is general poison • Deleterious effects on children's behaviorand attentiveness and Iqs • Dysfunctional sperm inmales • Miscarriages and stillbirth in pregnantwomen
- 13. HEALTH EFFETC IN ADULTS AND CHILDREN Blood levels lead Adults Children 10 g/dL Hypertension may occur •Crosses placenta •Impairment IQ, growth •Partial inhibition of heme synthesis 20 g/dL Inhibition of heme synthesis Increased erythrocyte protoporphyrin Beginning impairment of nerve conduction velocity 30 g/dL •Systolic hypertension •Impaired hearing( ) Impaired vitamin D metabolism 40 g/dL •Infertility in males •Renal effects •Neuropathy •Fatigue, headache, abd pain Hemoglobin synthesis inhibition 50 g/dL Anemia, GI sx, headache, tremor Colicky abd pain, neuropathy 100 g/dL Lethargy, seizures, encephalopathy Encephalopathy, anemia, nephropathy, seizures
- 15. MERCURY • Symbol Hg • From the Latin word, hydrargyrum, “liquid silver,” • Shining, mobile liquid, silvery-white in color • It is liquid at -38.9oC • Becomes solid when subjected to a pressure of 7,640 atmospheres • Freezing point of about -39°C (about -38°F), a boiling point of about 357°C • Density of 13.55 grams per cu cm. The atomic weight of mercury is 200.59gmol-1 • Conducts electricity well • Its principal ore is cinnabar
- 16. ENVIRONMENTAL SOURCES/USES OFMERCURY Electrical switches in automobilesbuilt before 2000 In fluorescent light bulbs and in streetlamps before dental amalgam Gold and silver mining Pesticides Used to make felt forhats
- 17. ENVIRONMENTAL SOURCES/USES OFMERCURY (cont.) Fossil fuel coal lignite(100ppb) Discarded laboratorychemicals Pharmaceuticalproducts Crematoria Wastewater from dentists’offices Preservepaint Used as slimicides in pulp-and-paper industry As topical antiseptic and in cosmetics
- 18. MERCURY ANDTHE INDUSTRIAL CHLOR-ALKALI PROCESS amalgam of sodium and mercury is used in some industrial chlor-alkali plants in process that converts aqueos sodium chloride into the commercial products chlorine and sodium hydroxide and gaseous hydrogen by electrolysis. Flowing mercury is used as the cathode Some of the mercury find its way to the air and water
- 19. MERCURY VAPOR It is composed of free, neutralatoms. If inhaled the atoms diffuse from the lungsto the bloodstream liquid mercury is not highly toxic, when ingested most are excreted
- 20. METHYLMERCURY TOXICITY Methyl anion combines with Hg2+ to yield volatile molecular liquid dimethylmercury. The formation occurs in muddy sediments of rivers and lakes, under anaerobic condition Methylmercury(or monomethylmercury) • Its production predominates over dimethylmercury in acidic or neutral aqueous systems • Sulfate ions stimulates sulfate-reducing bacteria that methylate mercury • Sulfide ions results in formation of mercury sulfide complexes that do not undergo methylation
- 22. (cont.) between methylmercury in fish muscle and that dissolved in the water: 1 million to 1, and can exceed 10 million to 1 High methylmercury concentration inlarge, long-lived predatory marine species
- 23. METHYLMERCURY ACCUMULATION INTHE ENVIRONMENT AND IN THEHUMANBODY • Half-life of methylmercury in human bodyis 70days • Cumulativepoison • dialkylmercury including dimethylmercuryis supertoxic
- 24. MINAMATA DISEASE • Occurred in a village in Minamata, Japan • CH3Hg-SCH3 • 100ppm of mercury in fish tissues • Onset symptoms were first seen in cats • Symptoms in humans arise from dysfunctions of the central nervous system • Methylmercury can be passed to fetus • U.S. has EPA maximum conc of 0.3ppm in fish tissue
- 25. SOURCES OFMETHYLMERCURY Fungicide in agriculture andindustry Leaching from rocks and soil intowater systems by natural processes PreservativeThimerosal
- 26. SAFE LEVEL OF MERCURY IN THE BODY 10-20ppm of CH3Hg+ in hair can be dangerous INTERNATIONAL CONTROL ONMERCURY United Nations Environment Programme in 2005 devised a global treaty to curb the production if mercury and to ban completely the export of mercury between countries
- 28. Drinking Water Guidelines for Heavy Metals
- 30. *AdditionalNoteon: THE WOOD PRESERVATIVECCA Chromated CopperArsenate A waterborne mixture of metal oxideswith which wood is treated using vacuum- pressure impregnation process 10% of the mass of thelumber Cr(IV) is usedhere Protects woodenstructures Replaced creosote andpentachlorophenol