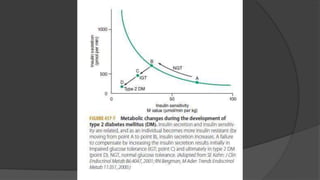
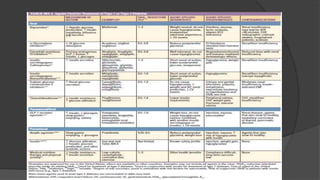
This document discusses diabetes mellitus (DM), including its classification, pathophysiology, and diagnosis. It describes the two main types of DM - type 1 resulting from autoimmune destruction of insulin-producing beta cells, and type 2 involving insulin resistance and impaired insulin secretion. Other types of DM exist that share features of type 1 and 2 or have distinct genetic causes. The classification of DM is based on the pathogenic processes leading to hyperglycemia. Precise diagnostic criteria exist involving hemoglobin A1c, fasting plasma glucose, and oral glucose tolerance tests.












![▶ The International Expert Committee with members appointed by the ADA,
the European Association for the Study of Diabetes, and the International
Diabetes Federation have issued diagnostic criteria for DM (Table 417-2)
based on the following premises:
▶ (1) the FPG, the response to an oral glucose challenge (oral glucose tolerance
test [OGTT]), and HbA1c differ among individuals
▶ (2) DM is defined as the level of glycemia at which diabetes-specific
complications occur rather than on deviations from a population-based
mean.
▶ A random plasma glucose concentration ≥11.1 mmol/L (200 mg/dL)
accompanied by classic symptoms of DM (polyuria, polydipsia, weight loss)
is also sufficient for the diagnosis of DM](https://image.slidesharecdn.com/diabetes-rupak1-3-240313130349-35e10776/85/Harrison-book-based-diabetes-Rupak1-3-pptx-13-320.jpg)








![▶ Pathophysiology:
▶ Although other islet cell types (alpha cells [glucagonproducing], delta cells
[somatostatin-producing], or PP cells [pancreatic polypeptide-producing]) are
functionally and embryologically similar to beta cells and express most of
same proteins as beta cells, they are spared from the autoimmune
destruction.
▶ Pathologically, the pancreatic islets have a modest infiltration of lymphocytes
(a process termed insulitis). After beta cells are destroyed, it is thought that
the inflammatory process abates and the islets become atrophic.](https://image.slidesharecdn.com/diabetes-rupak1-3-240313130349-35e10776/85/Harrison-book-based-diabetes-Rupak1-3-pptx-22-320.jpg)

![▶ Beta cells seem to be particularly susceptible to the toxic effect of some
cytokines (tumor necrosis factor α [TNF-α], interferon γ, and interleukin
1[IL-1]).
▶ The precise mechanisms of beta cell death are not known but may
involve formation of nitric oxide metabolites, apoptosis, and direct CD8+
CD8+ T cell cytotoxicity.
▶ The islet destruction is mediated by T lymphocytes rather than islet
autoantibodies, as these antibodies do not generally react with the cell
surface of islet cells and are not capable of transferring DM to animals.
▶ Efforts to suppress the autoimmune process at the time of diagnosis of
diabetes have largely been ineffective or only temporarily effective in
slowing beta cell destruction.](https://image.slidesharecdn.com/diabetes-rupak1-3-240313130349-35e10776/85/Harrison-book-based-diabetes-Rupak1-3-pptx-24-320.jpg)