Embed presentation







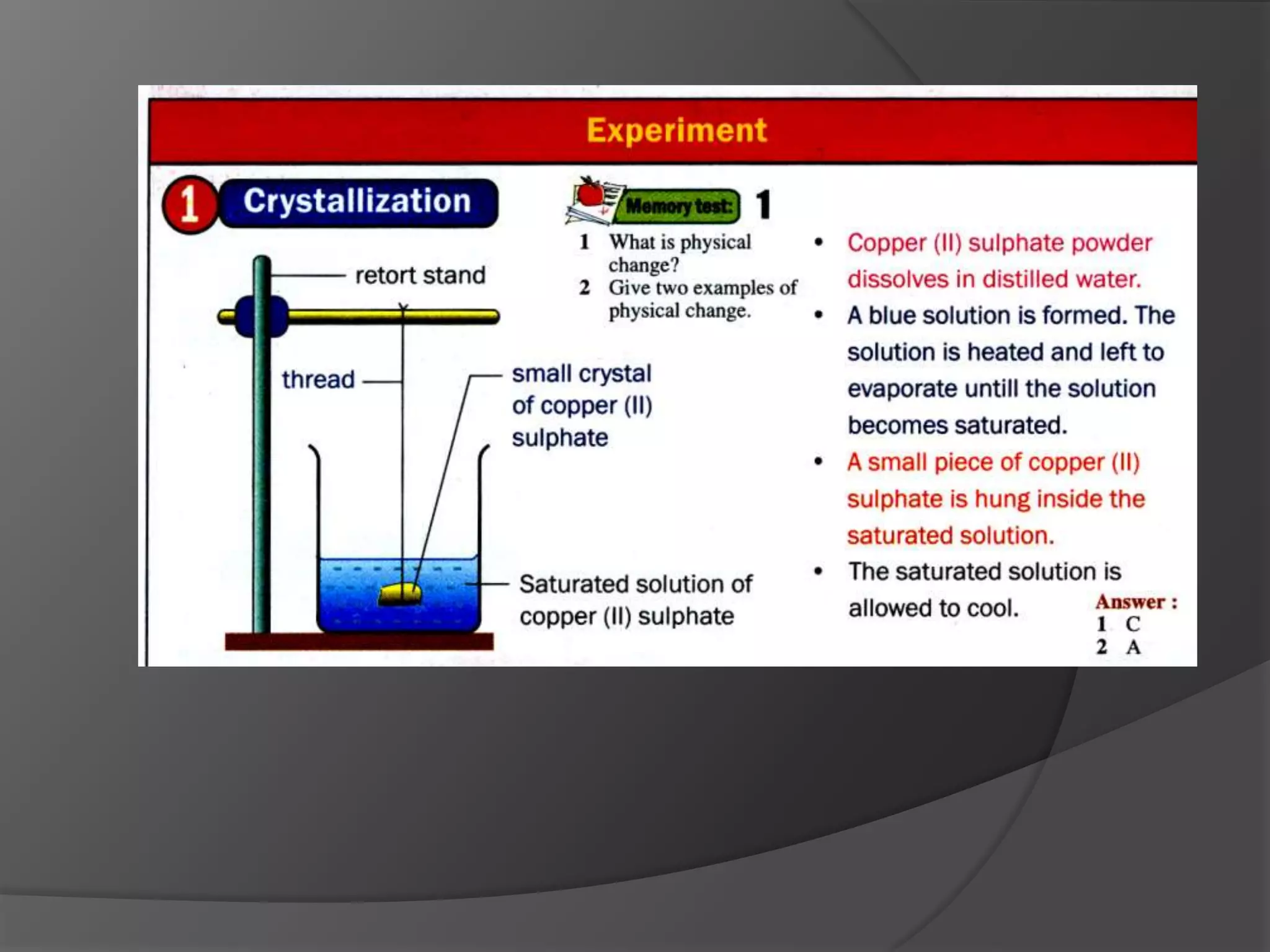
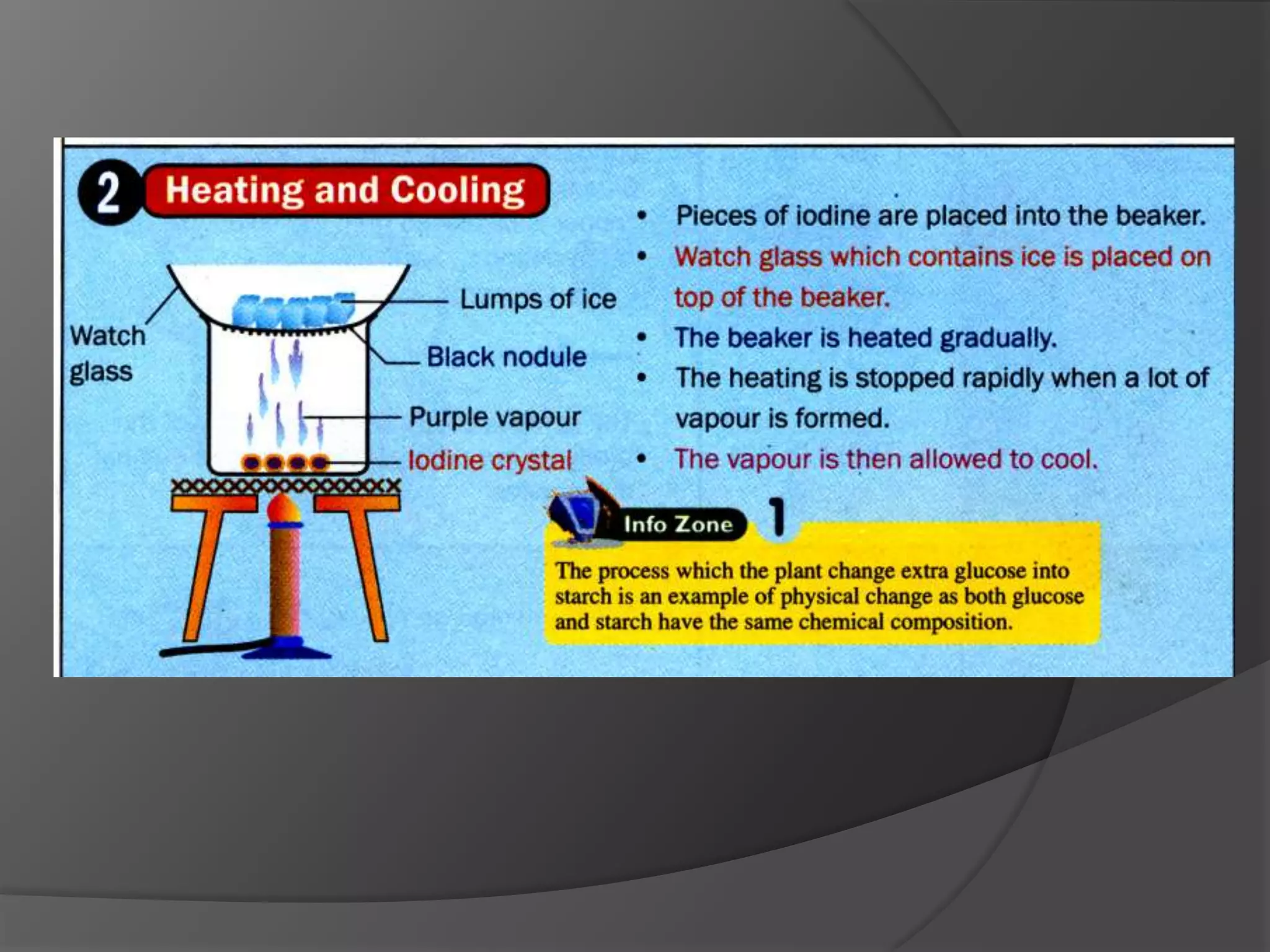
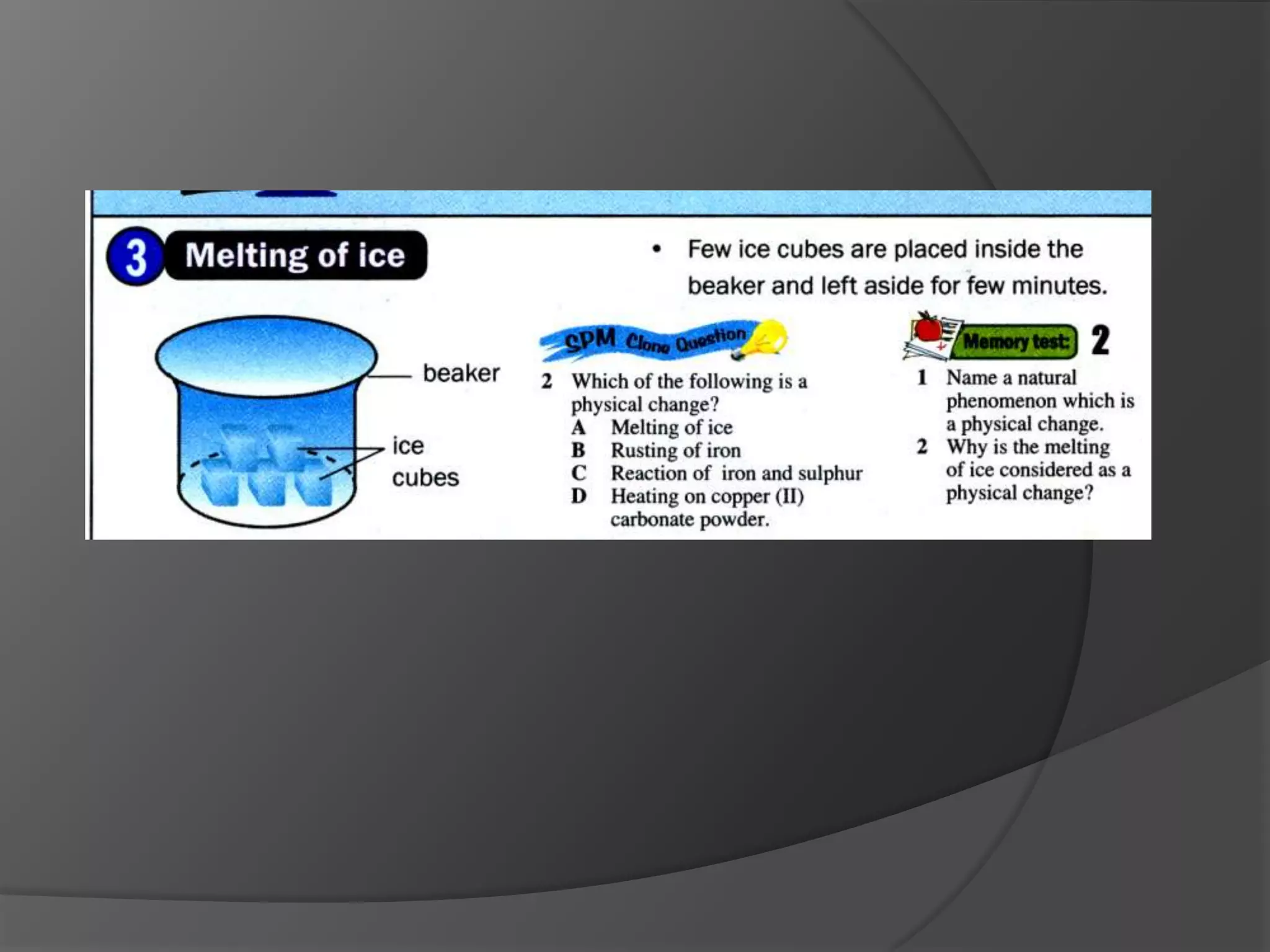
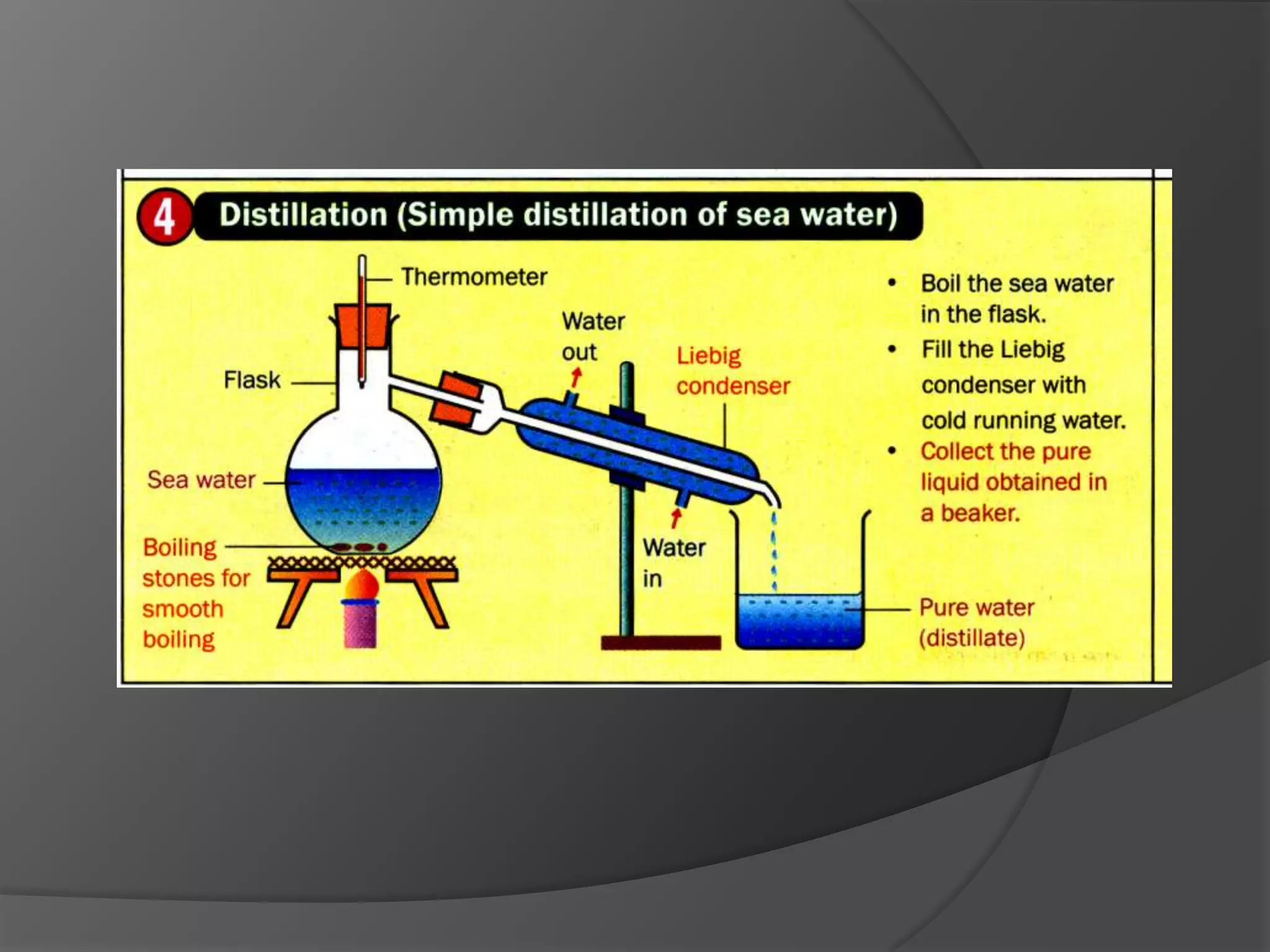



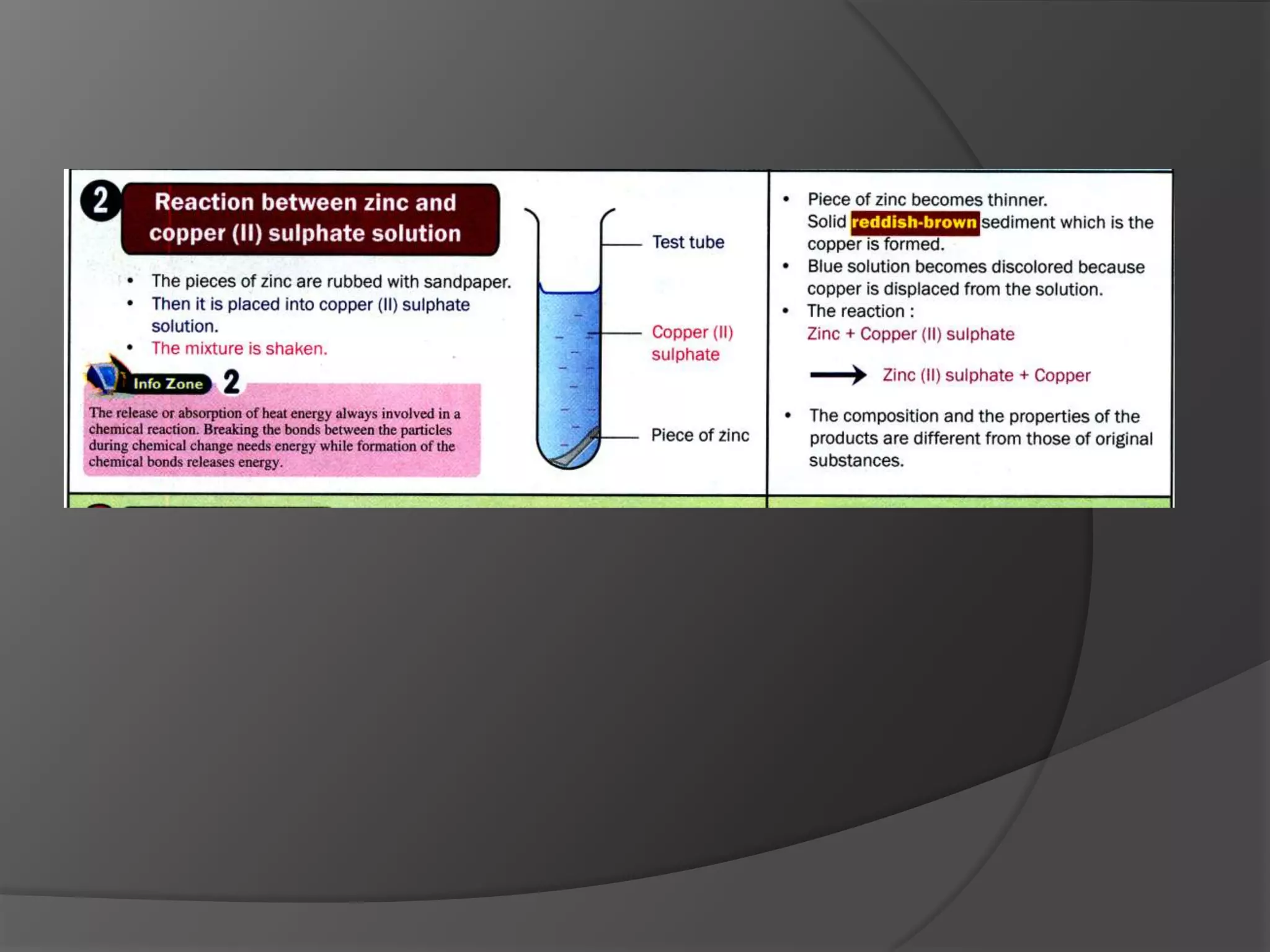
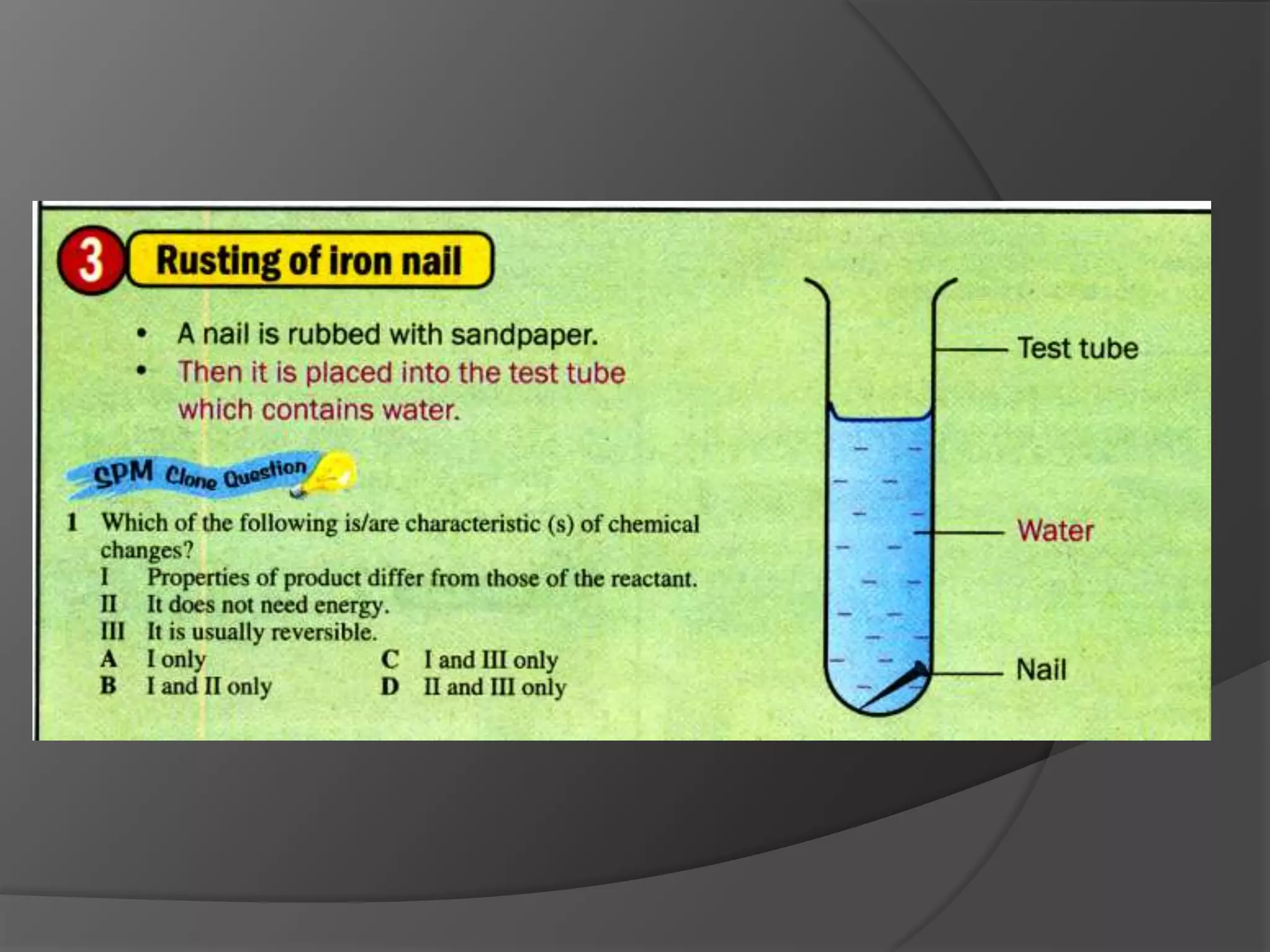
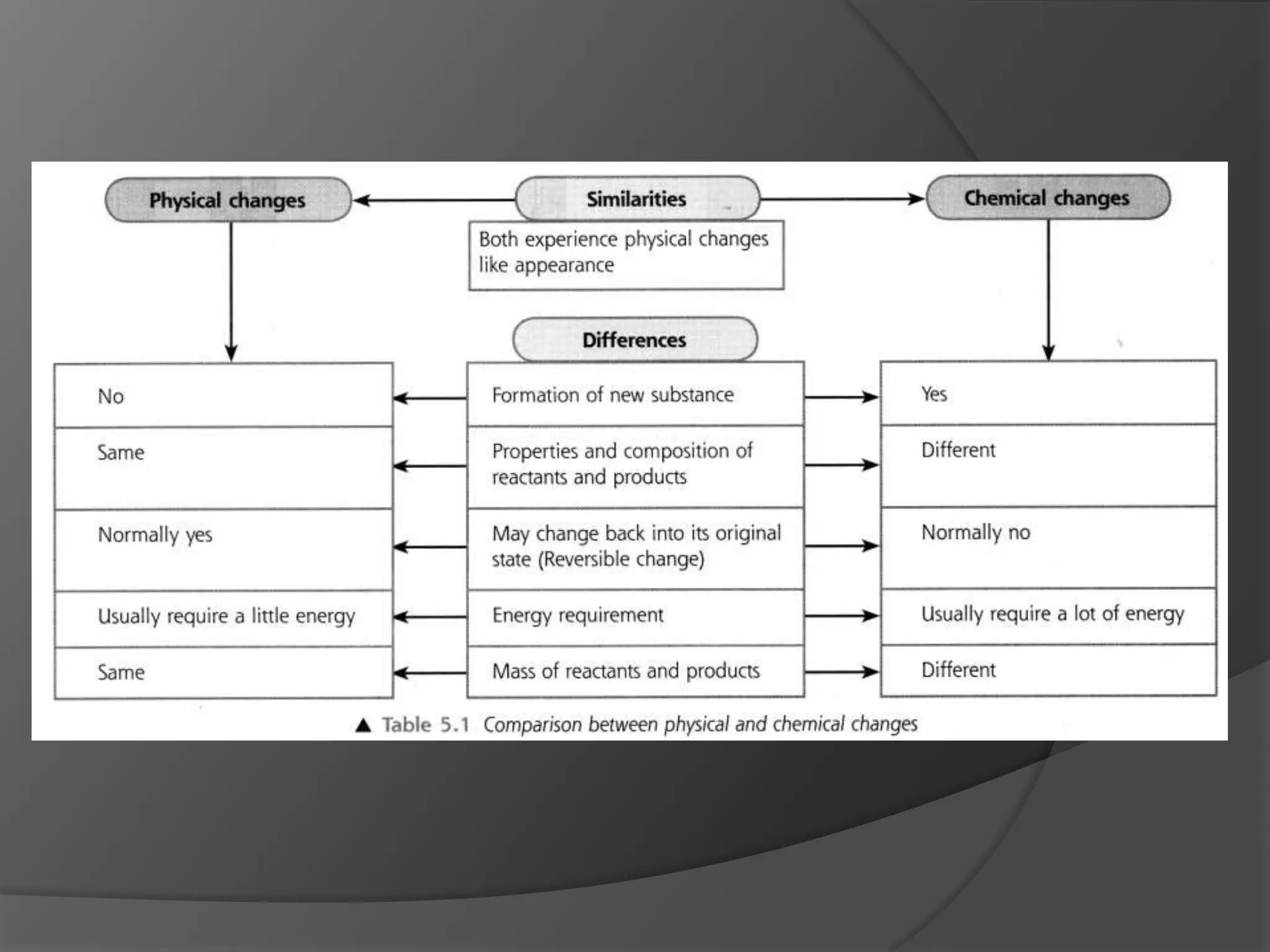
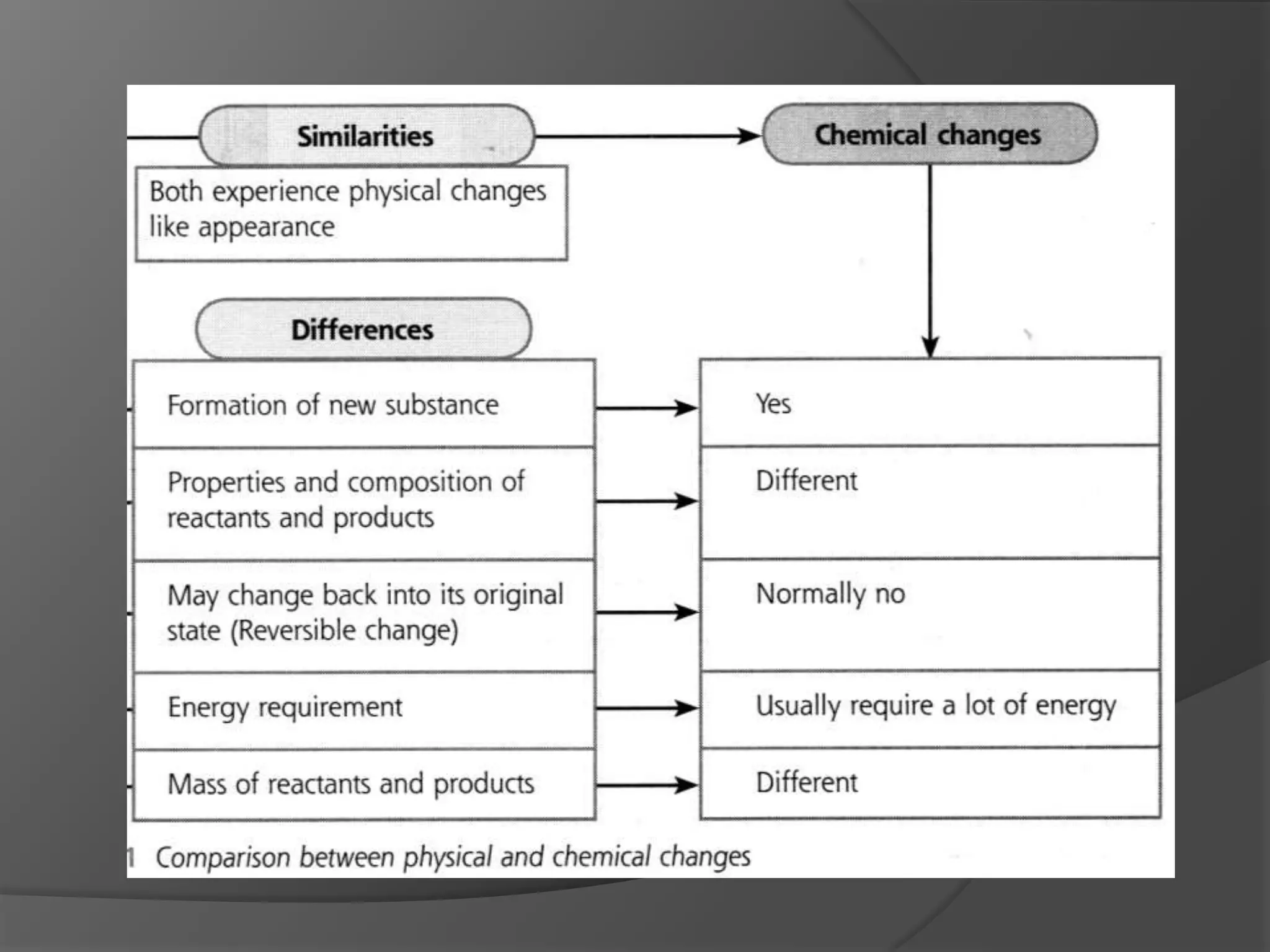






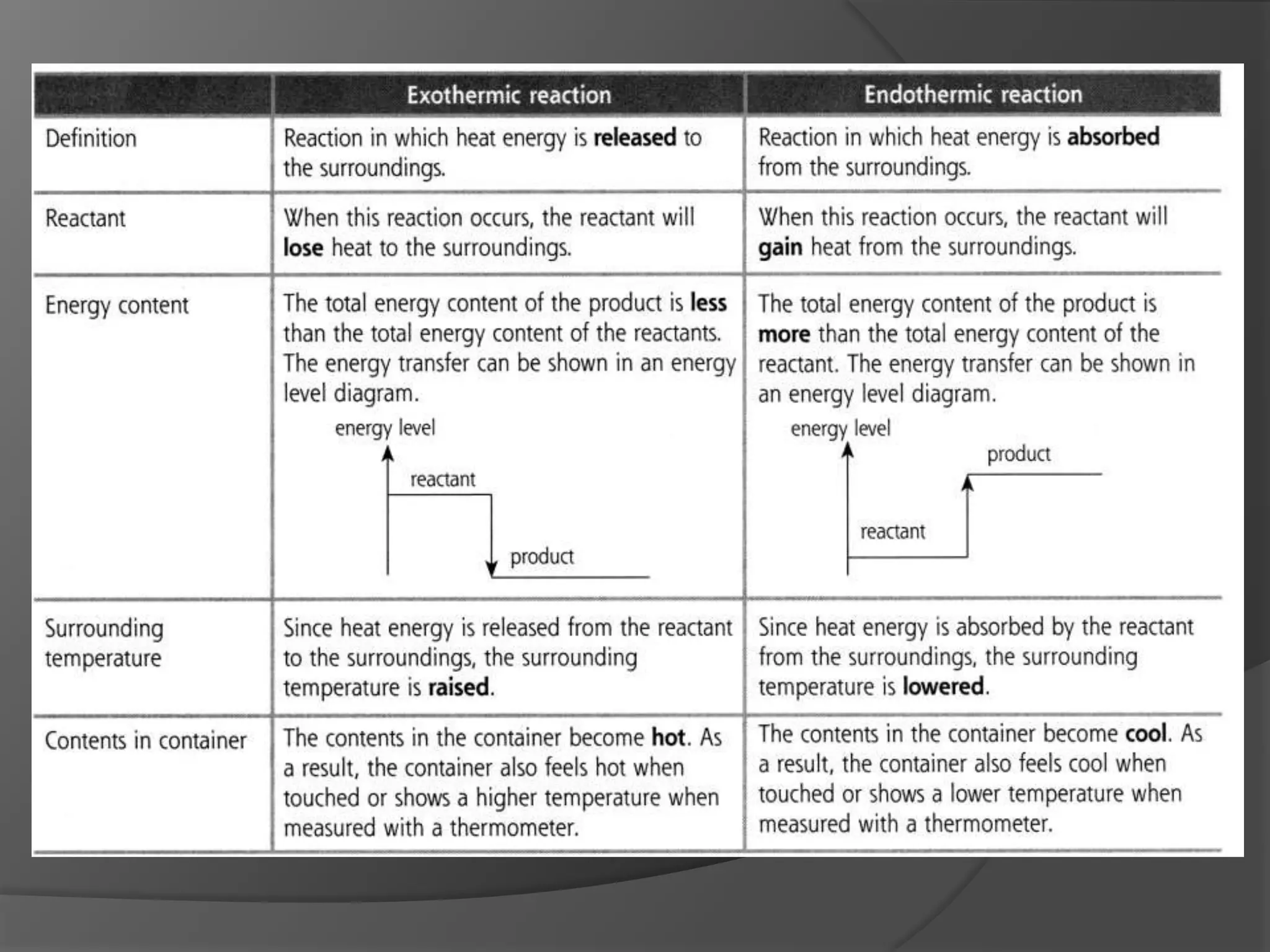

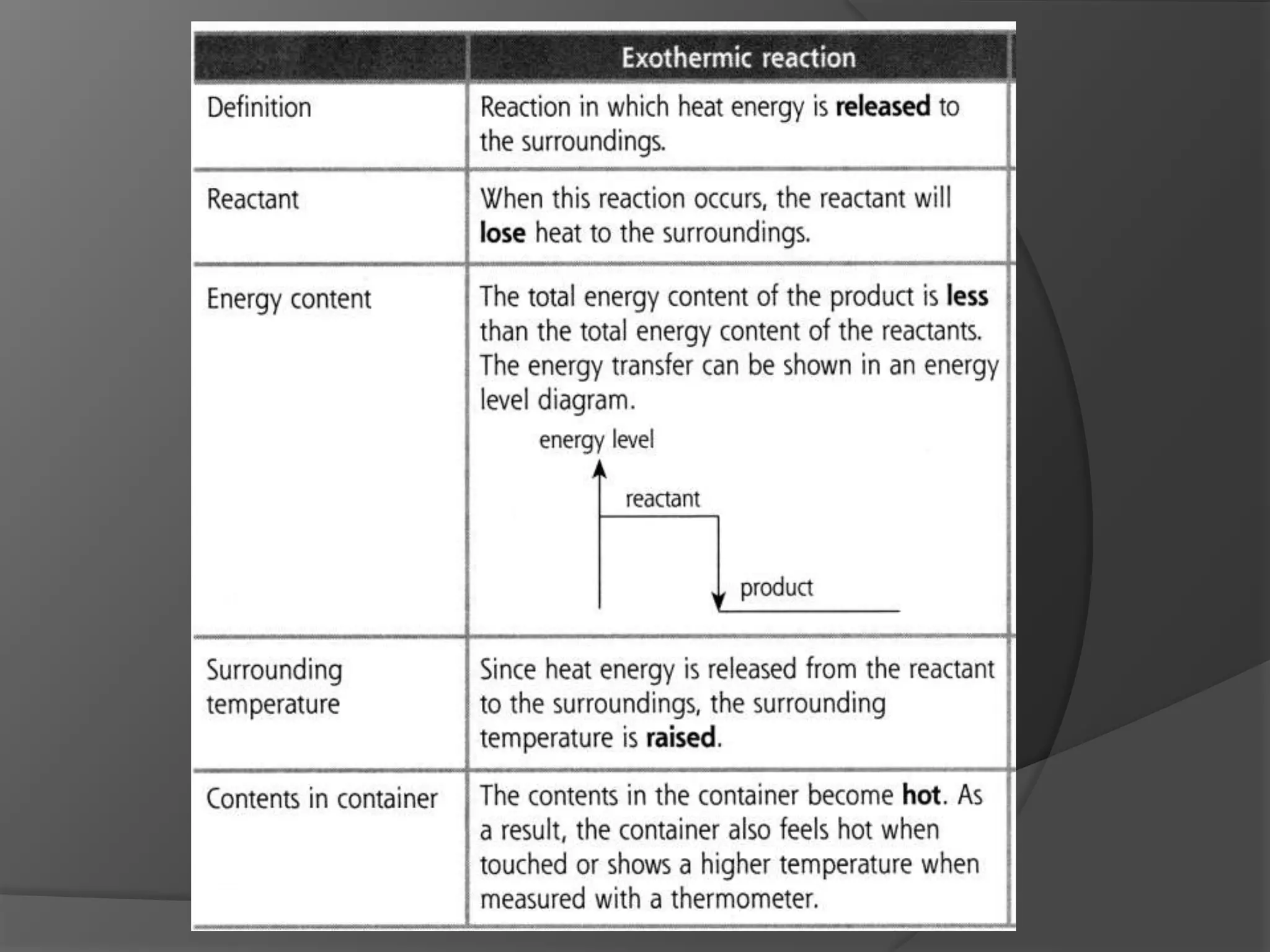
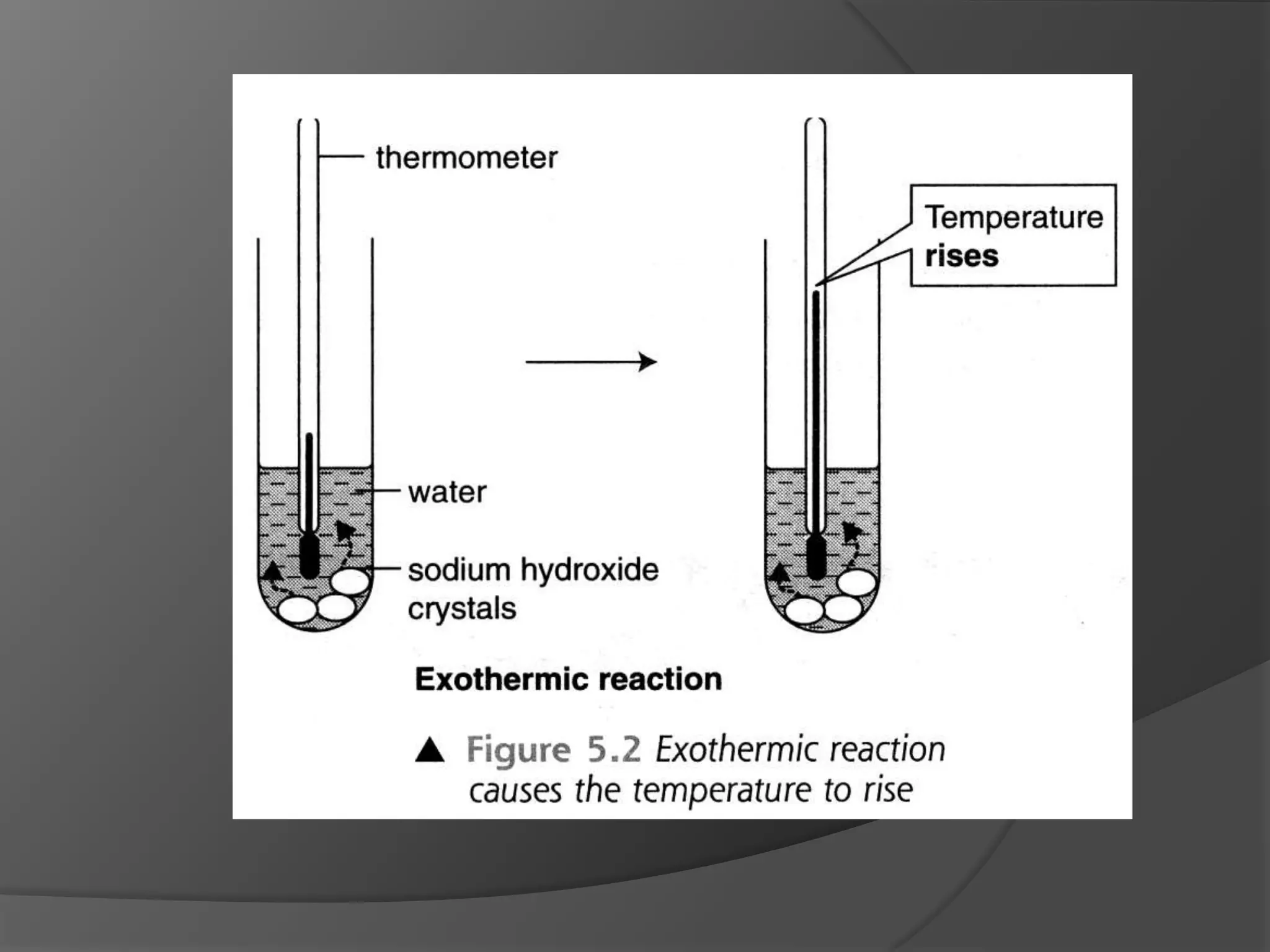

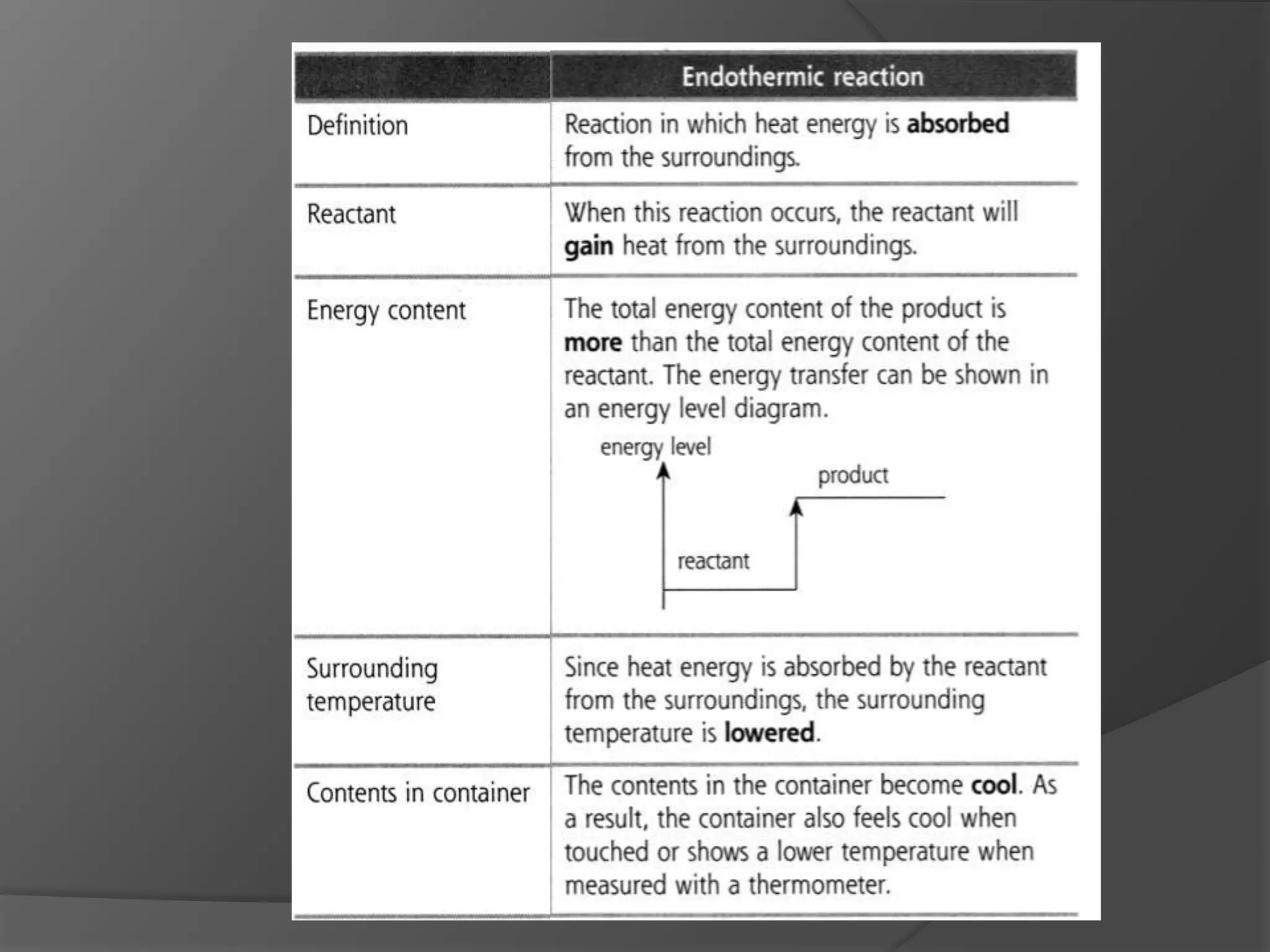

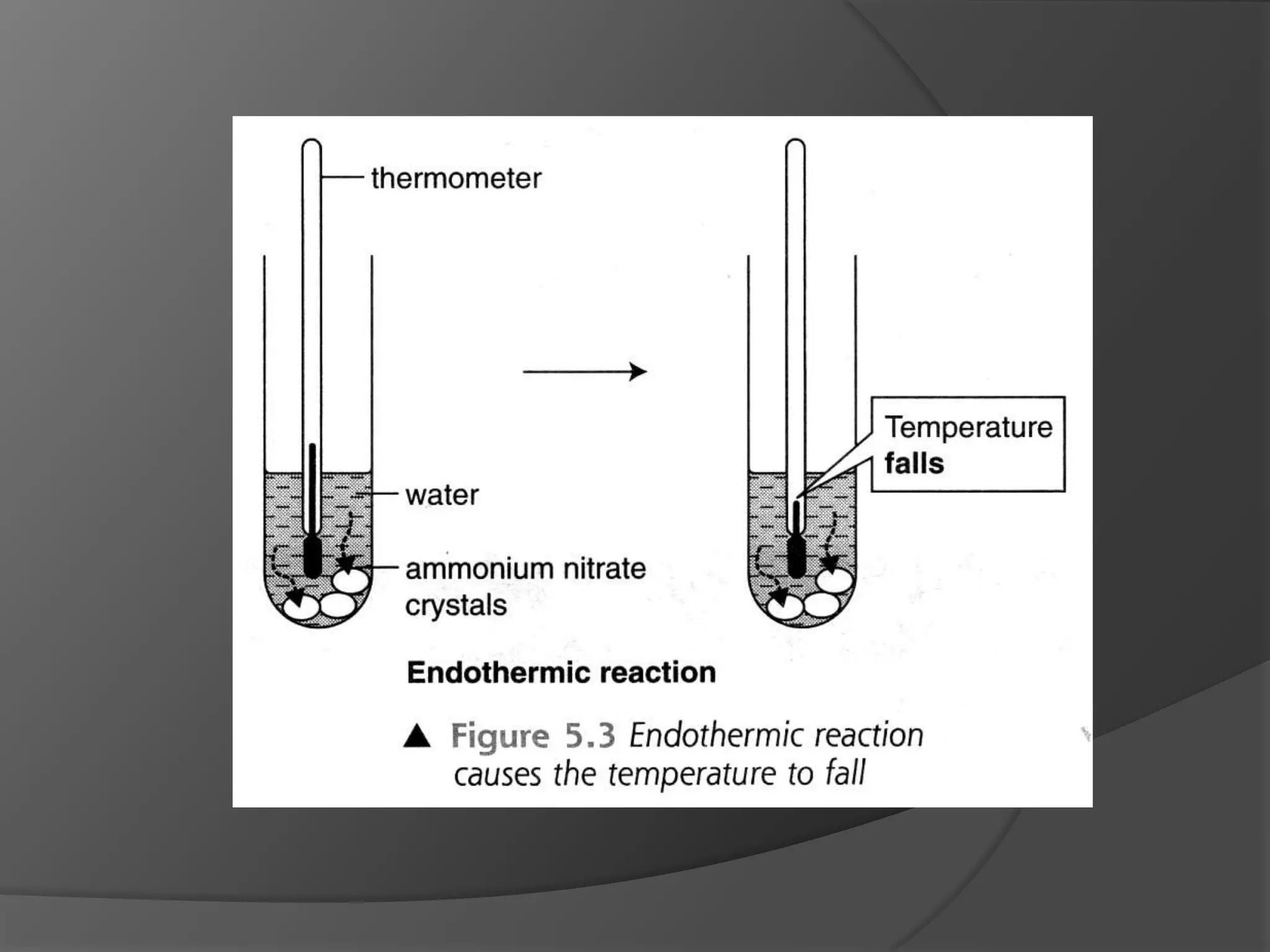



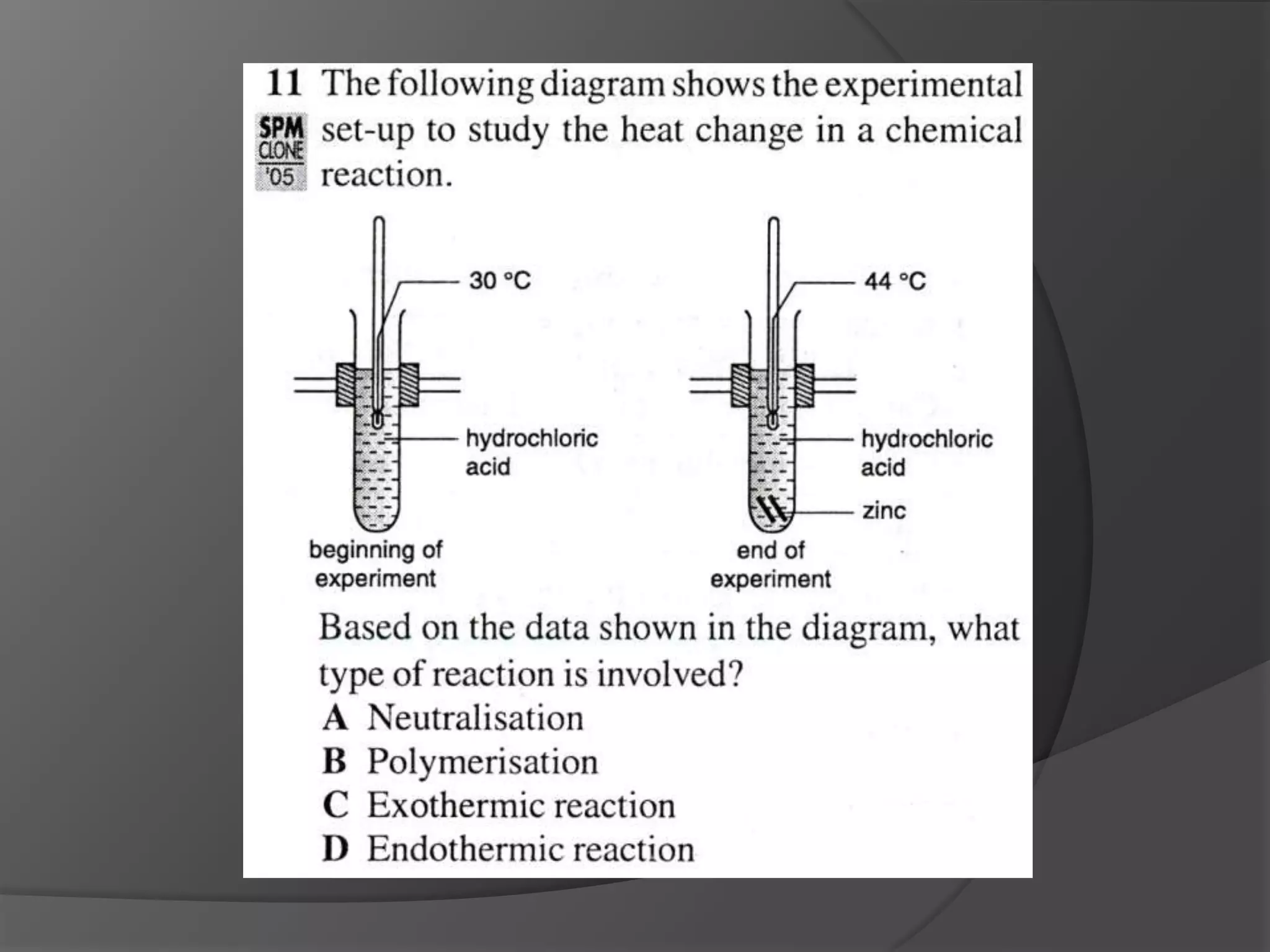







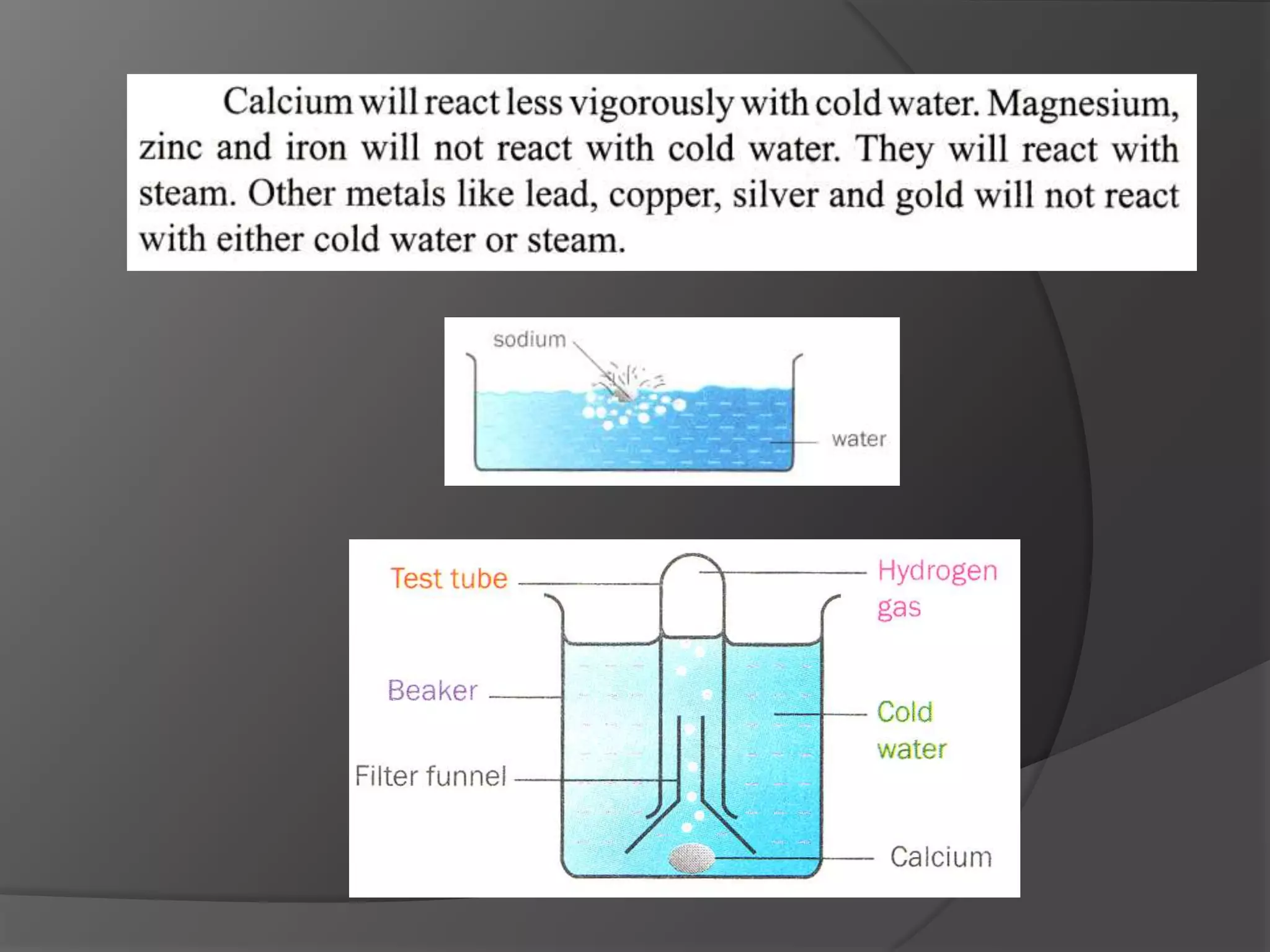
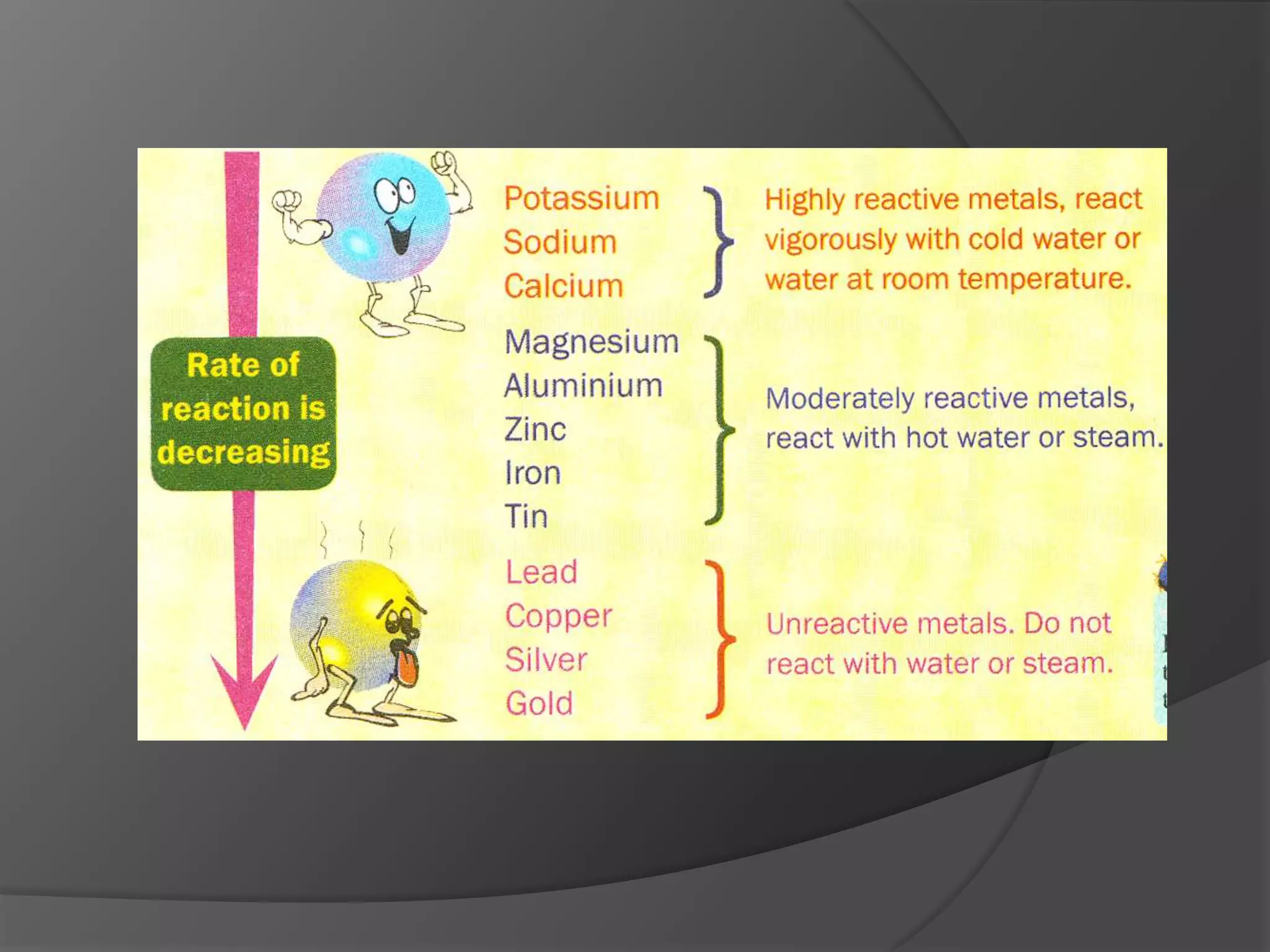





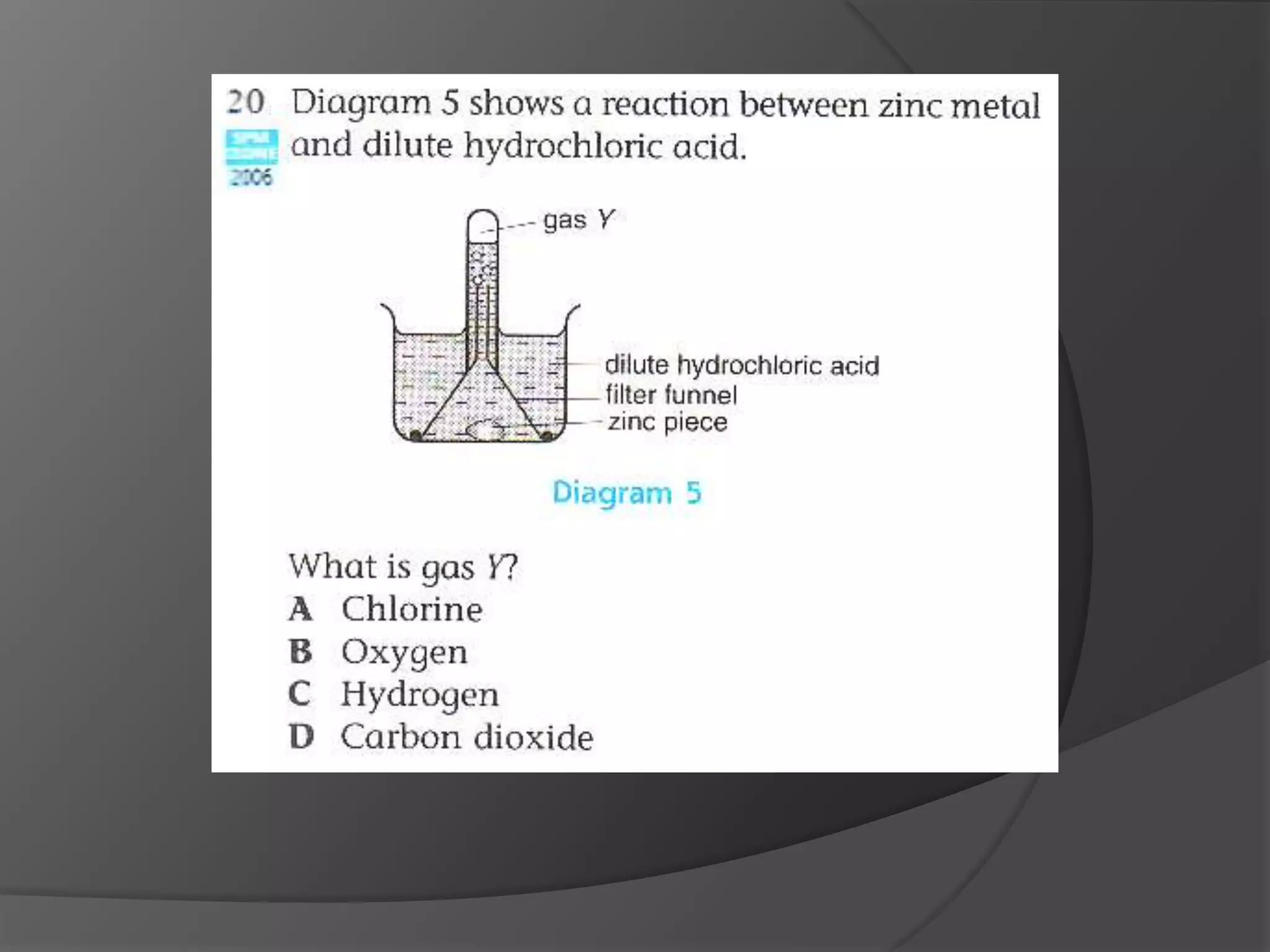


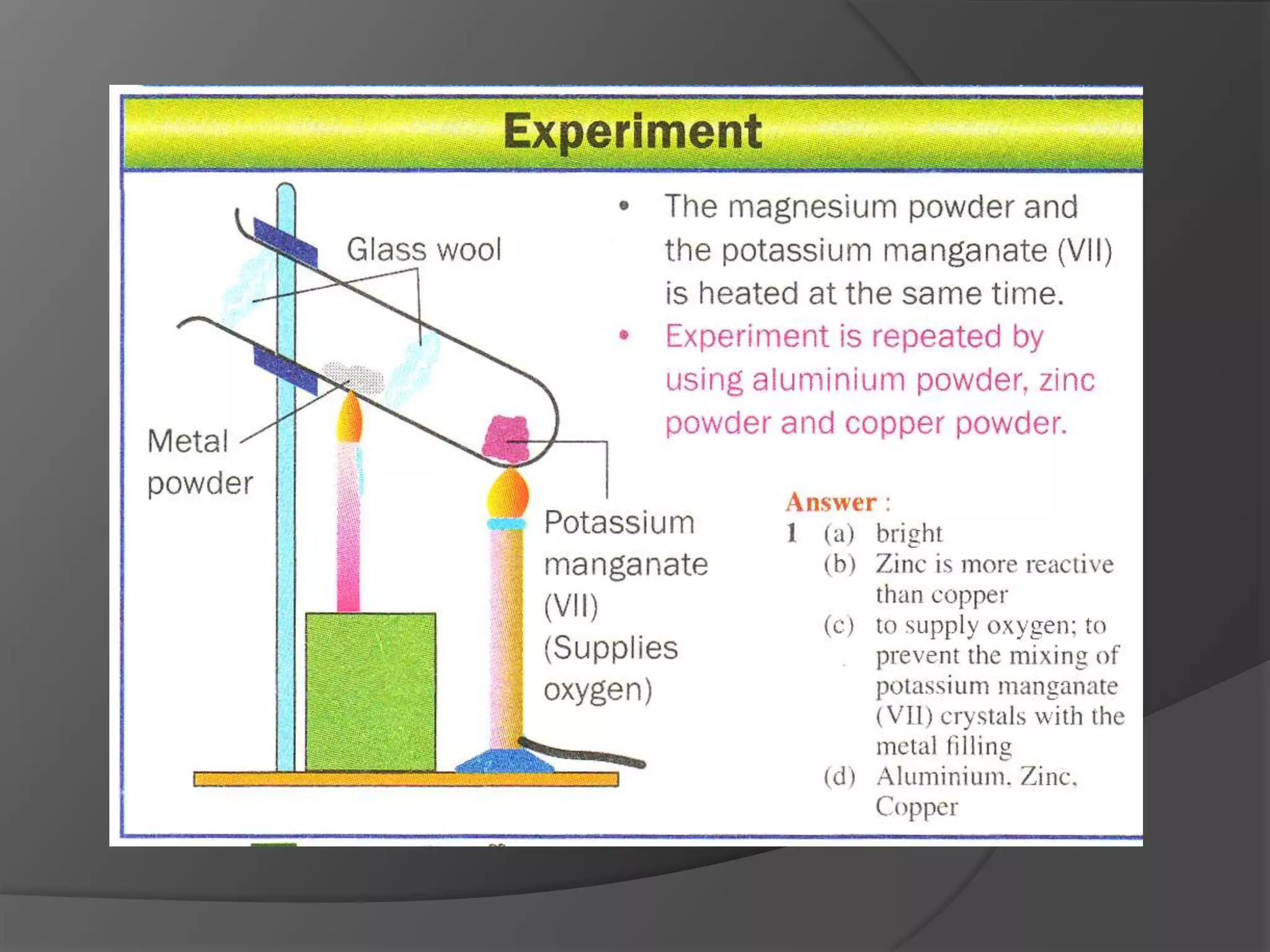



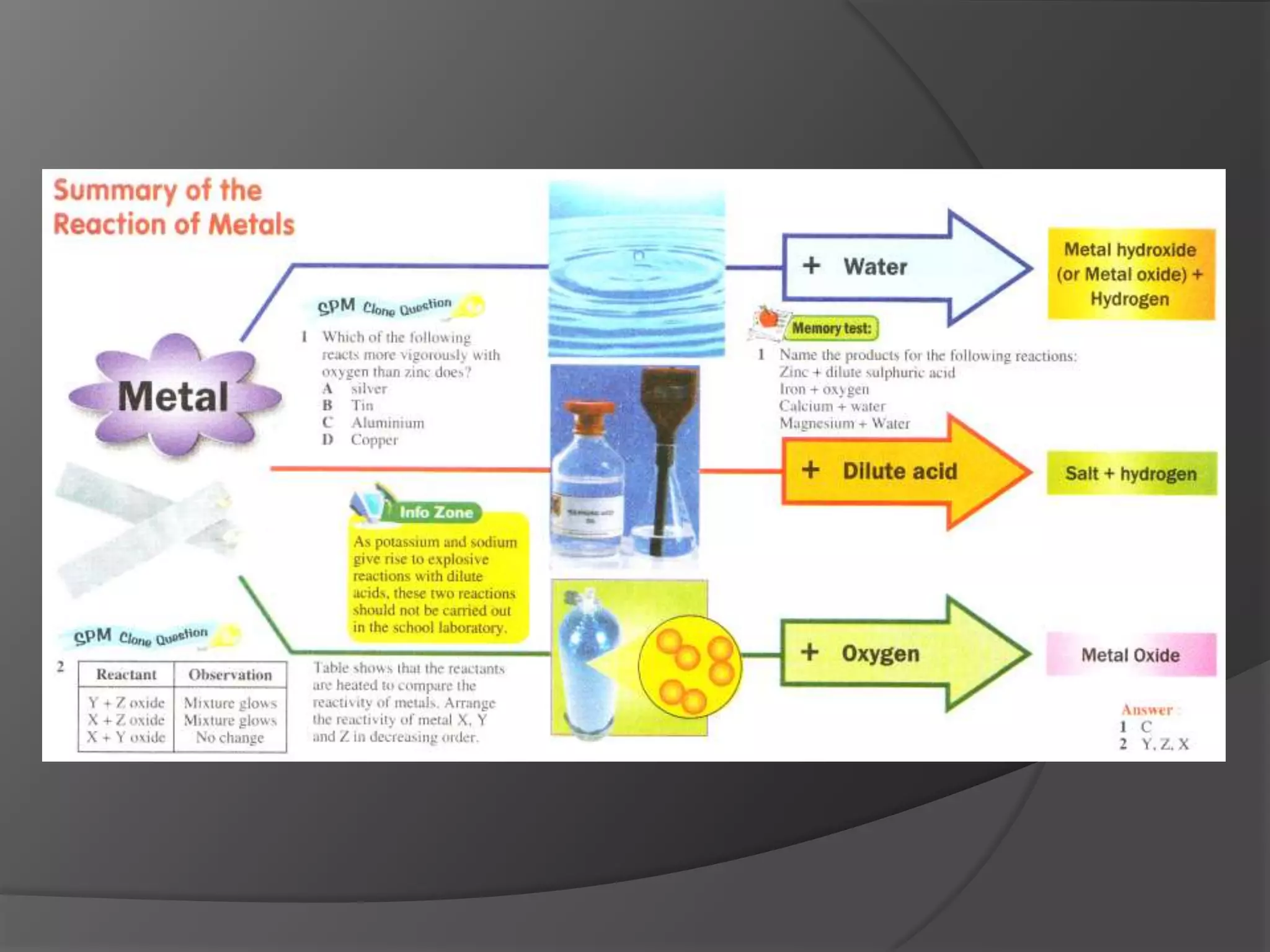
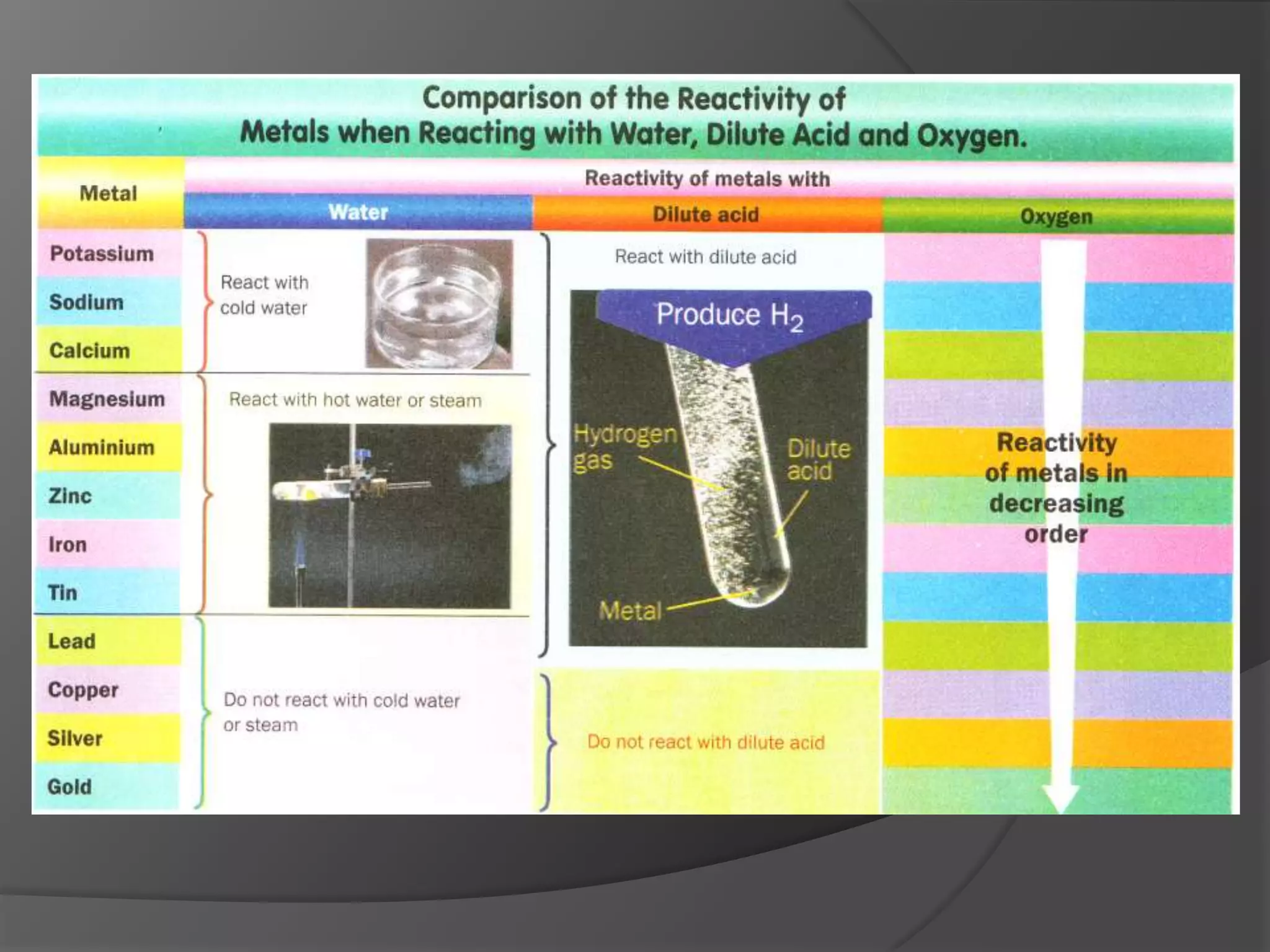

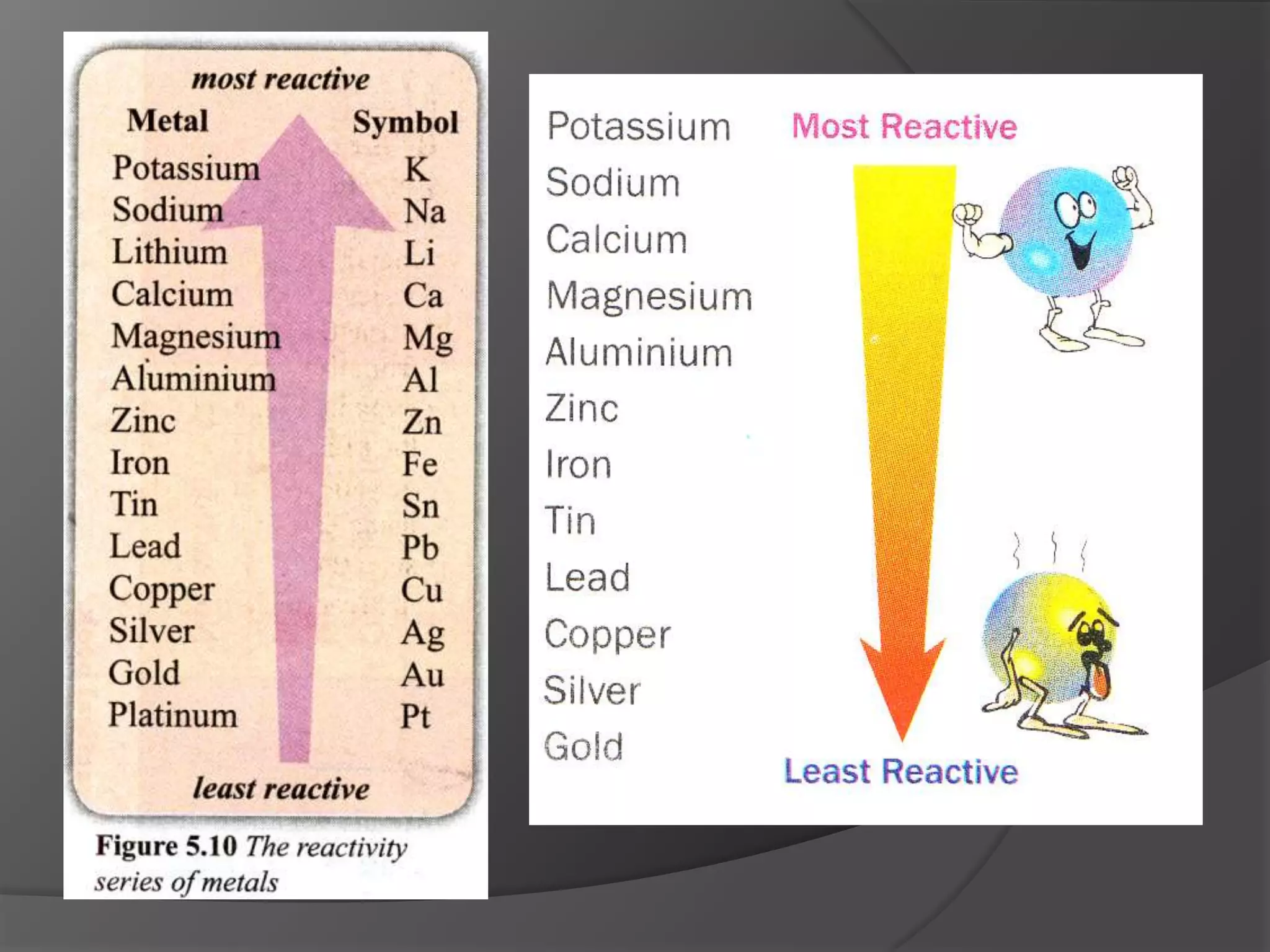





This chapter discusses physical and chemical changes, heat changes during chemical reactions, and the reactivity series of metals. Physical changes alter a substance's physical properties without changing its chemical composition, while chemical changes form new substances. Chemical reactions either absorb or release heat, changing the temperature of the surroundings. Metals exist in a reactivity series based on their tendency to undergo replacement reactions, with more reactive metals displacing less reactive ones.


































































