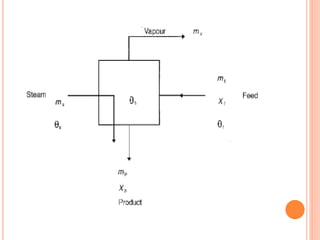
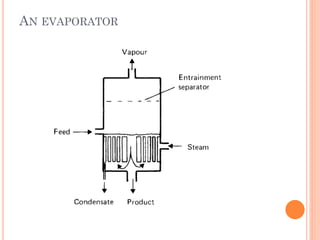
The document discusses food sterilization and preservation through heat processing. It describes sterilization as using high heat to destroy microbes and enzymes, giving foods a shelf life over 6 months. The factors that influence sterilization time include heat resistance of microbes, heating conditions, food acidity, and container size. Proper sterilization requires knowledge of microbe concentrations and heat resistance. Microbe death through heat follows a logarithmic order and can be measured using D-values and Z-values. Different container types and the rate of heat penetration must also be considered for effective sterilization.







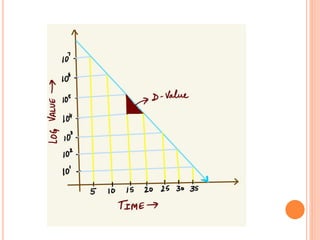



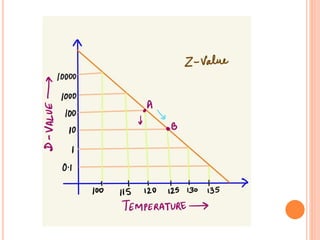


![Heat penetration rate
The rate of heat penetration is measured by measuring
the temperature at the centre of a container (the point
of slowest heating) ]food during processing
The rate will depend on the following factors;
Type of product
Size of container
Agitation of the container
Temperature of the retort
Shape of the container
Type of glass](https://image.slidesharecdn.com/foodprocessingandpreservation3-sterilization-240402094351-98046a7a/85/Food-Processing-and-preservation-3-Sterilization-pdf-15-320.jpg)