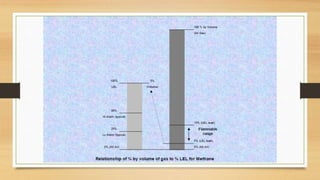
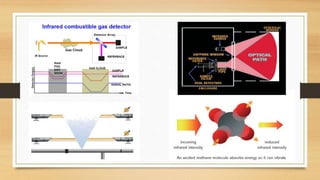
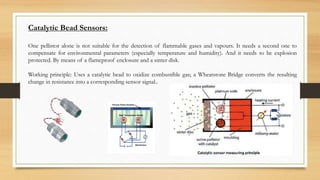
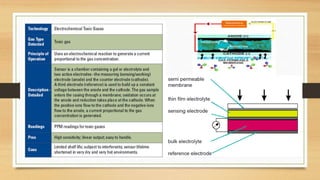
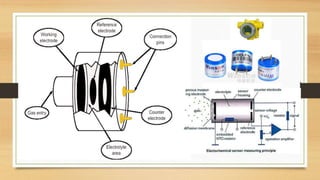
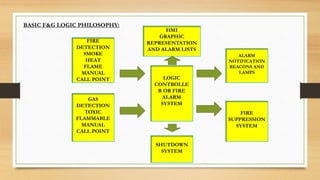
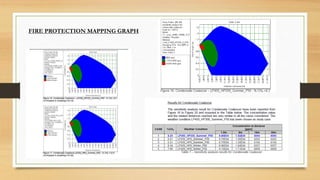
This document discusses fire and gas detection systems. It begins by defining fire and gases, explaining that fire is a chain reaction between fuel and oxygen that produces heat, light and other byproducts. Gases disperse and mix rapidly. Detection systems are needed to monitor hazardous gas levels and provide early warning before hazards form. They protect people, infrastructure and the environment per safety laws and codes. Detection considers flammable, toxic and asphyxiant gas risks. The document then covers gas properties, ignition risks, limits of flammability, and detection technology types like infrared and catalytic sensors. It stresses the importance of instrument calibration and certification to ensure accurate measurements. Examples of industrial accidents caused by gas leaks are also provided.