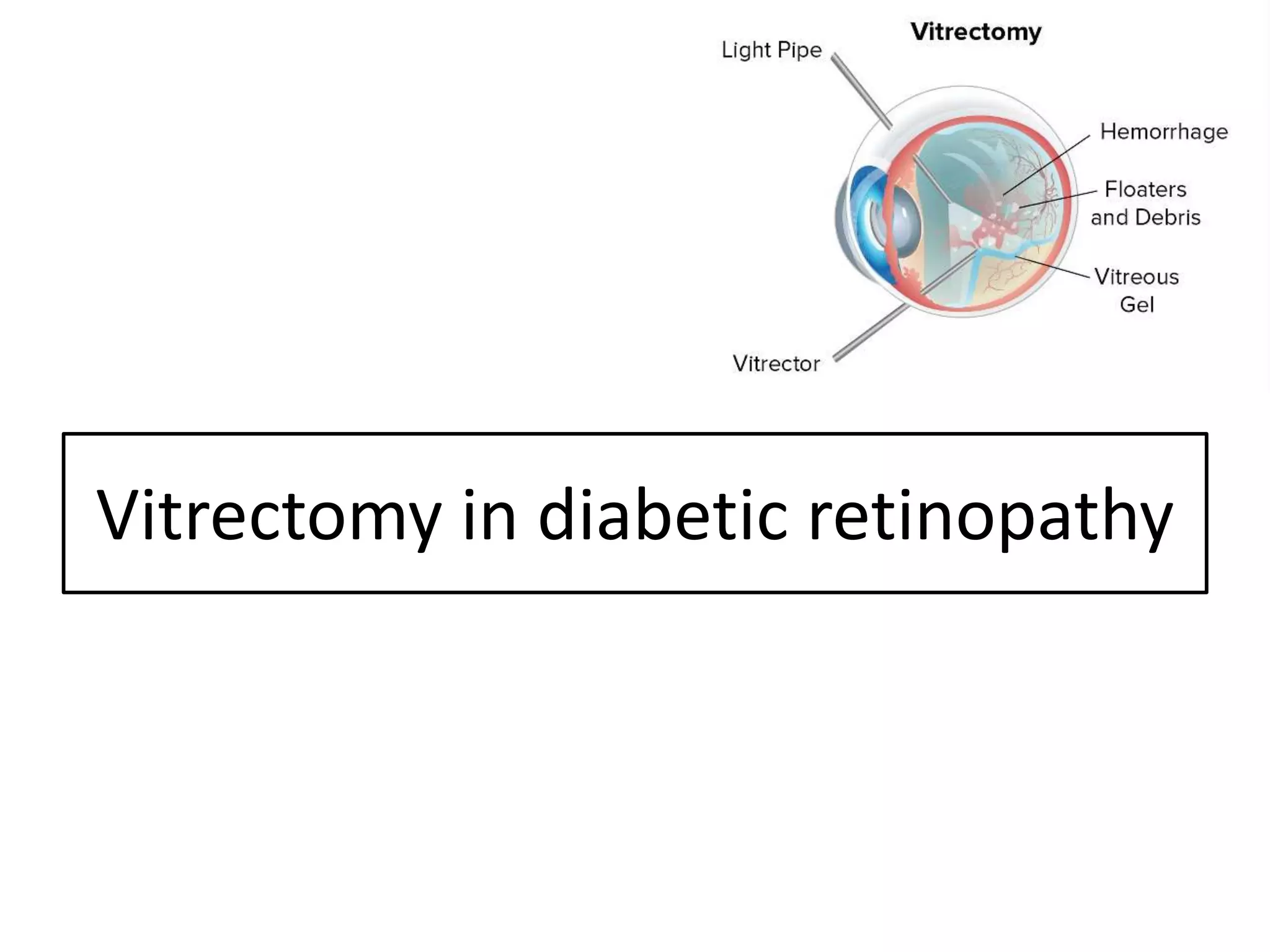
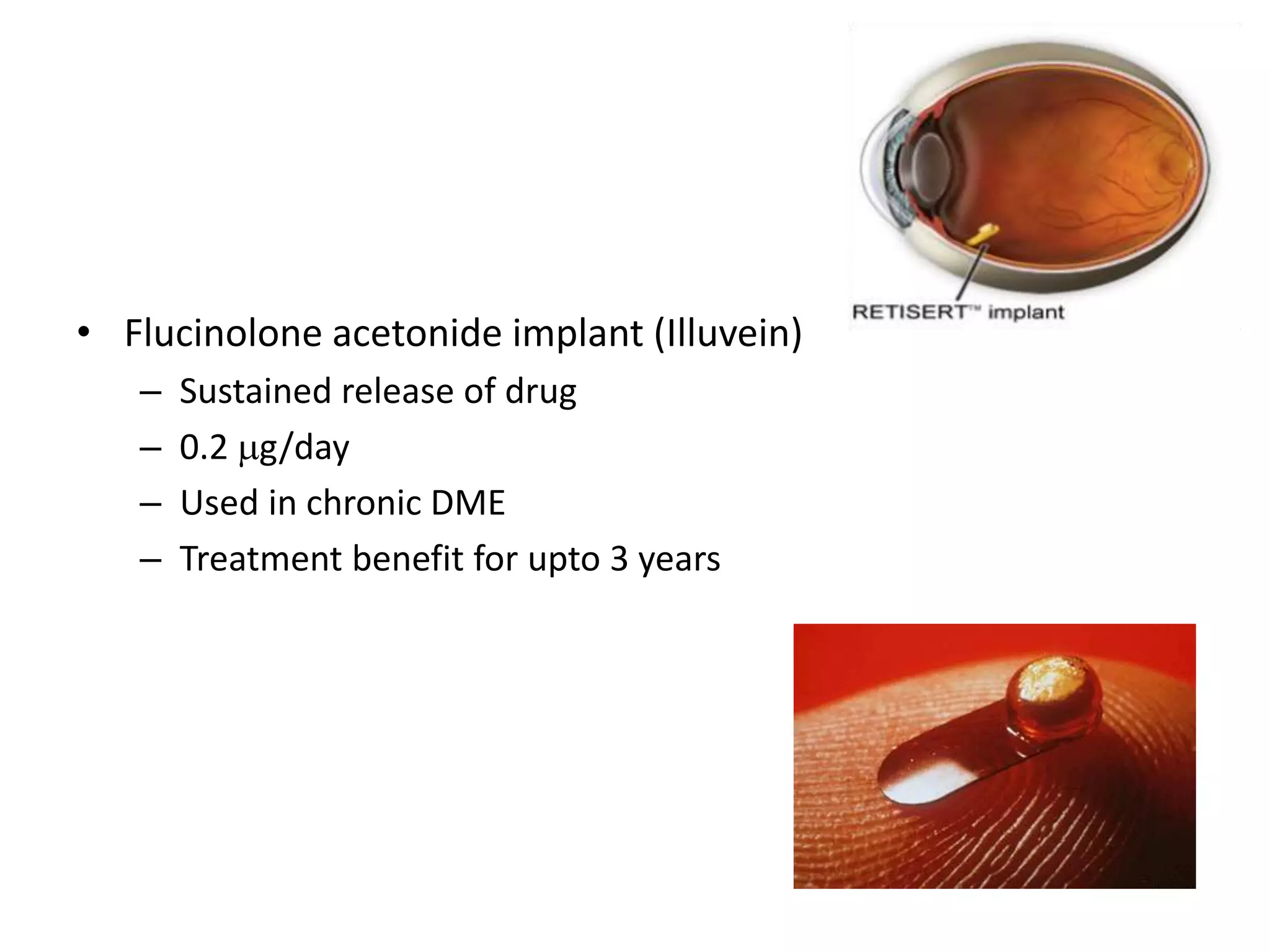
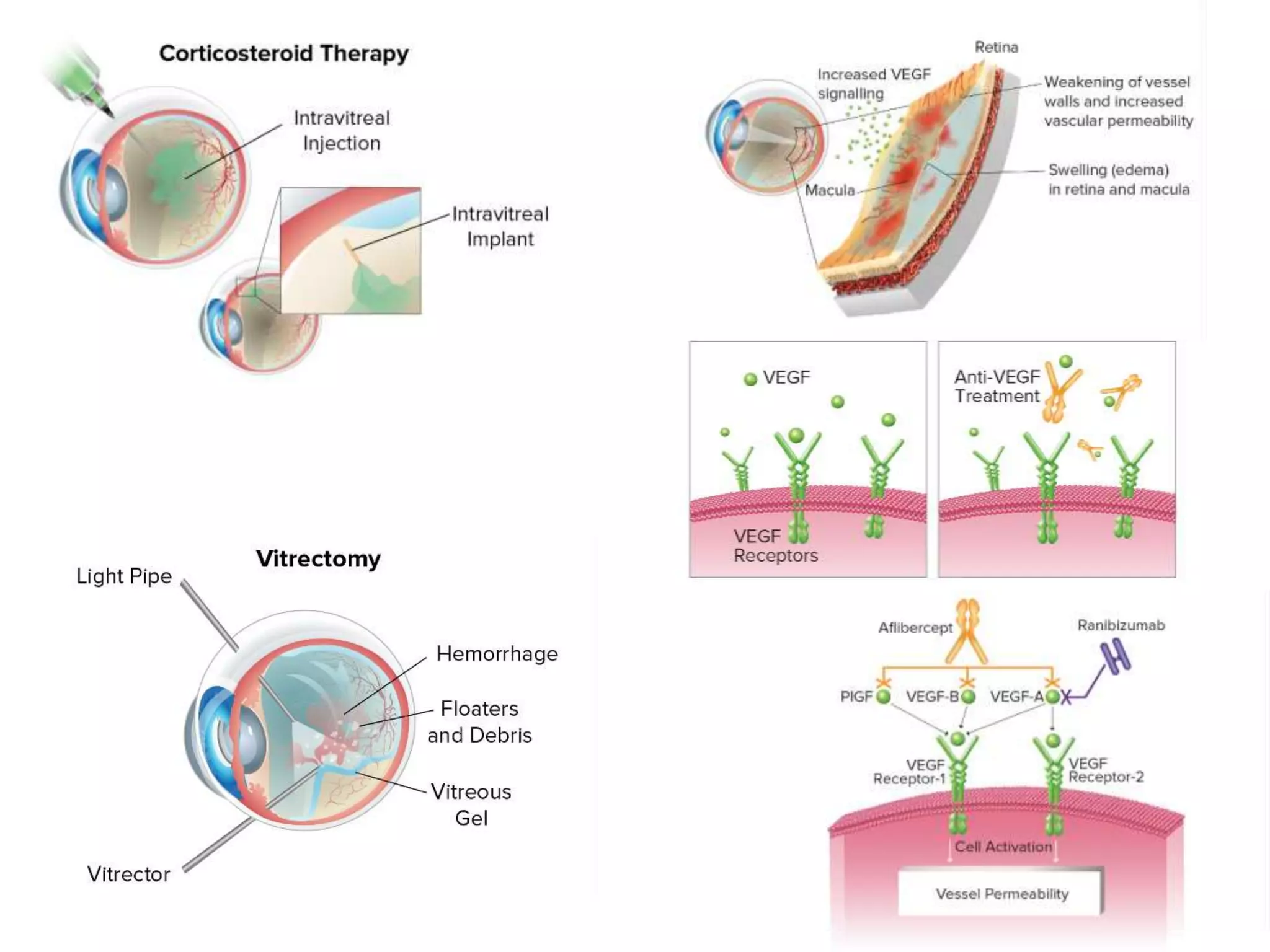
This document discusses diabetic retinopathy, including:
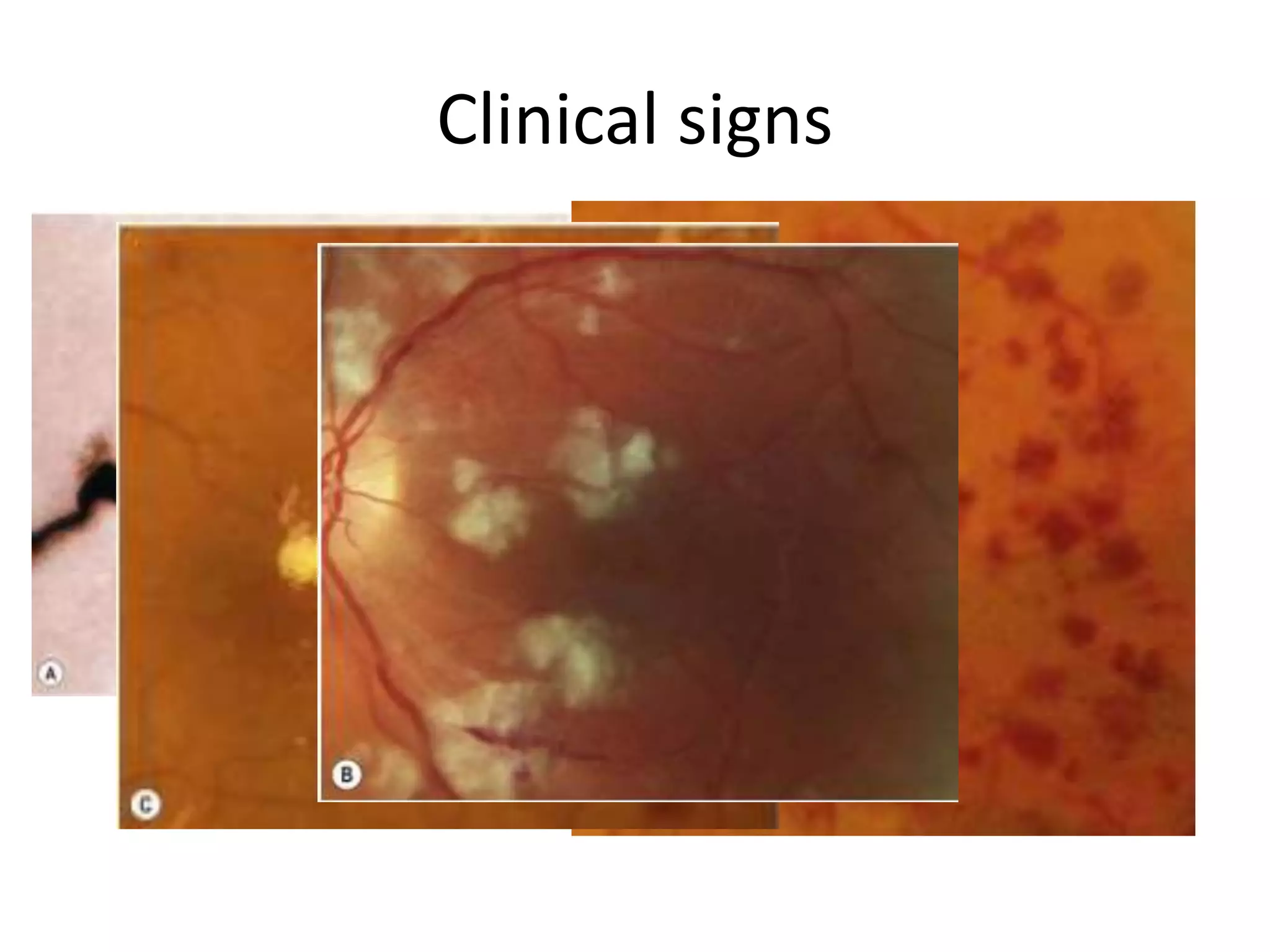
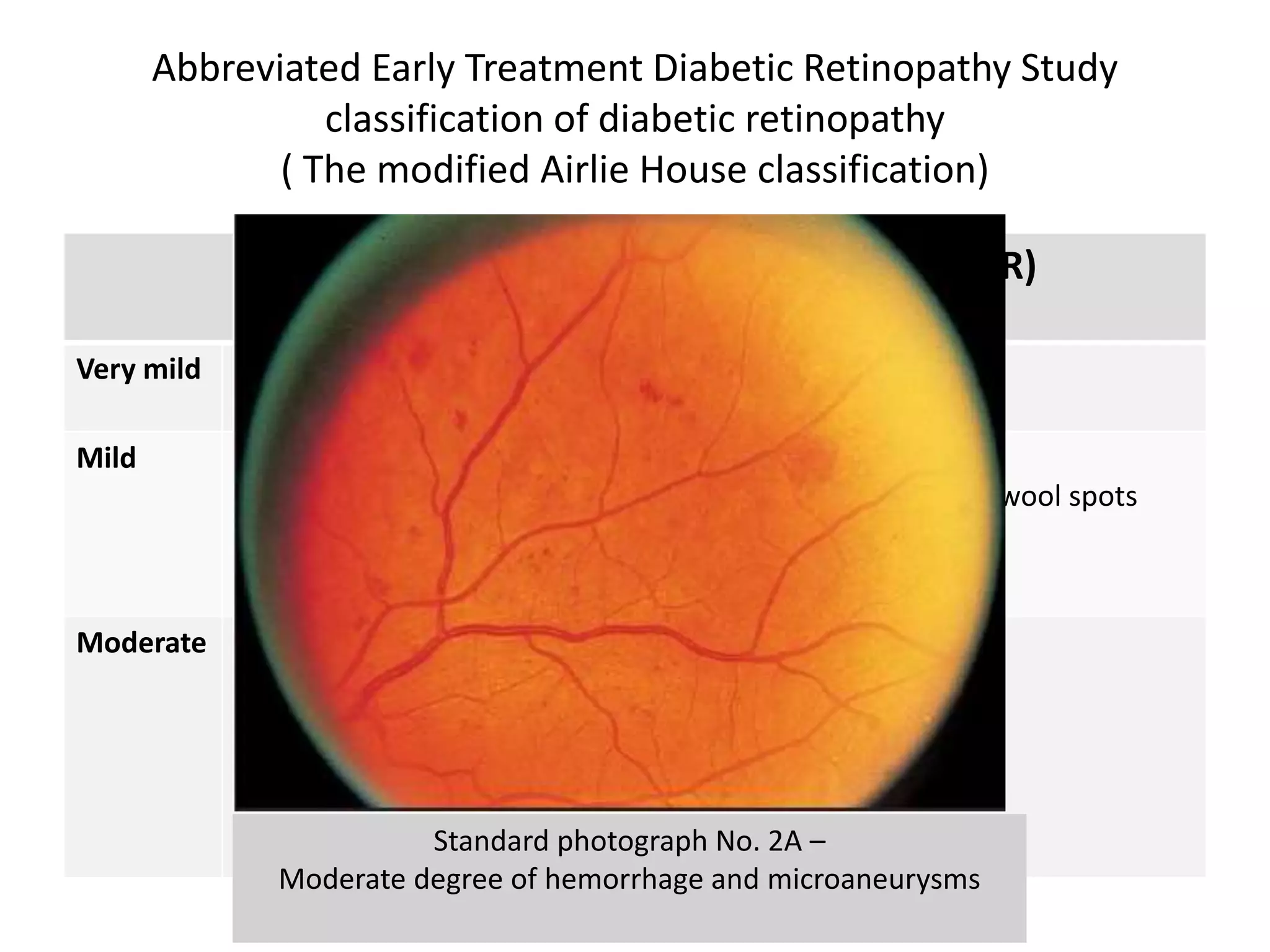
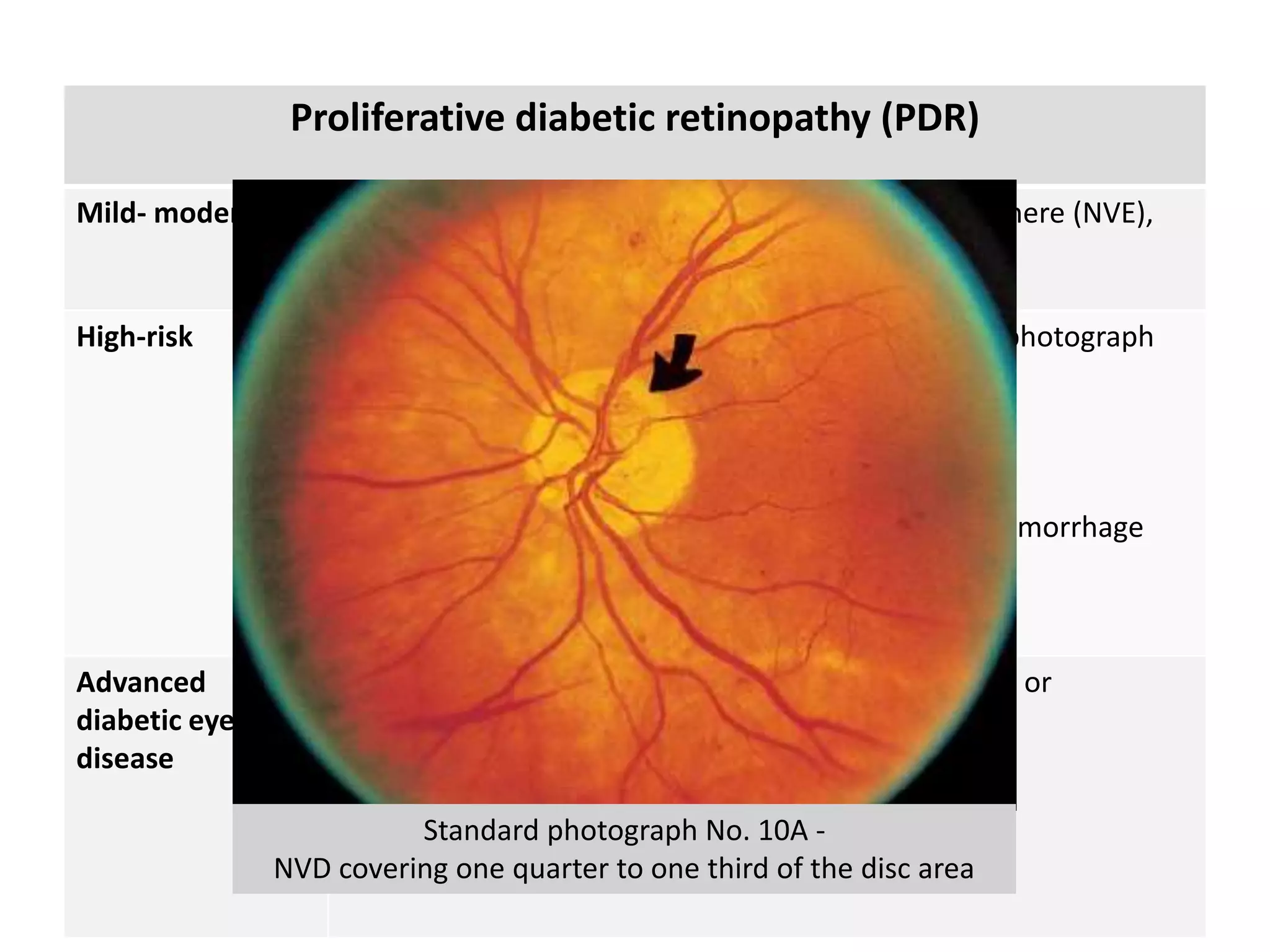
- DR is a major cause of blindness and is classified as non-proliferative or proliferative.
- Tight control of blood glucose, blood pressure, and lipids can slow progression of DR.
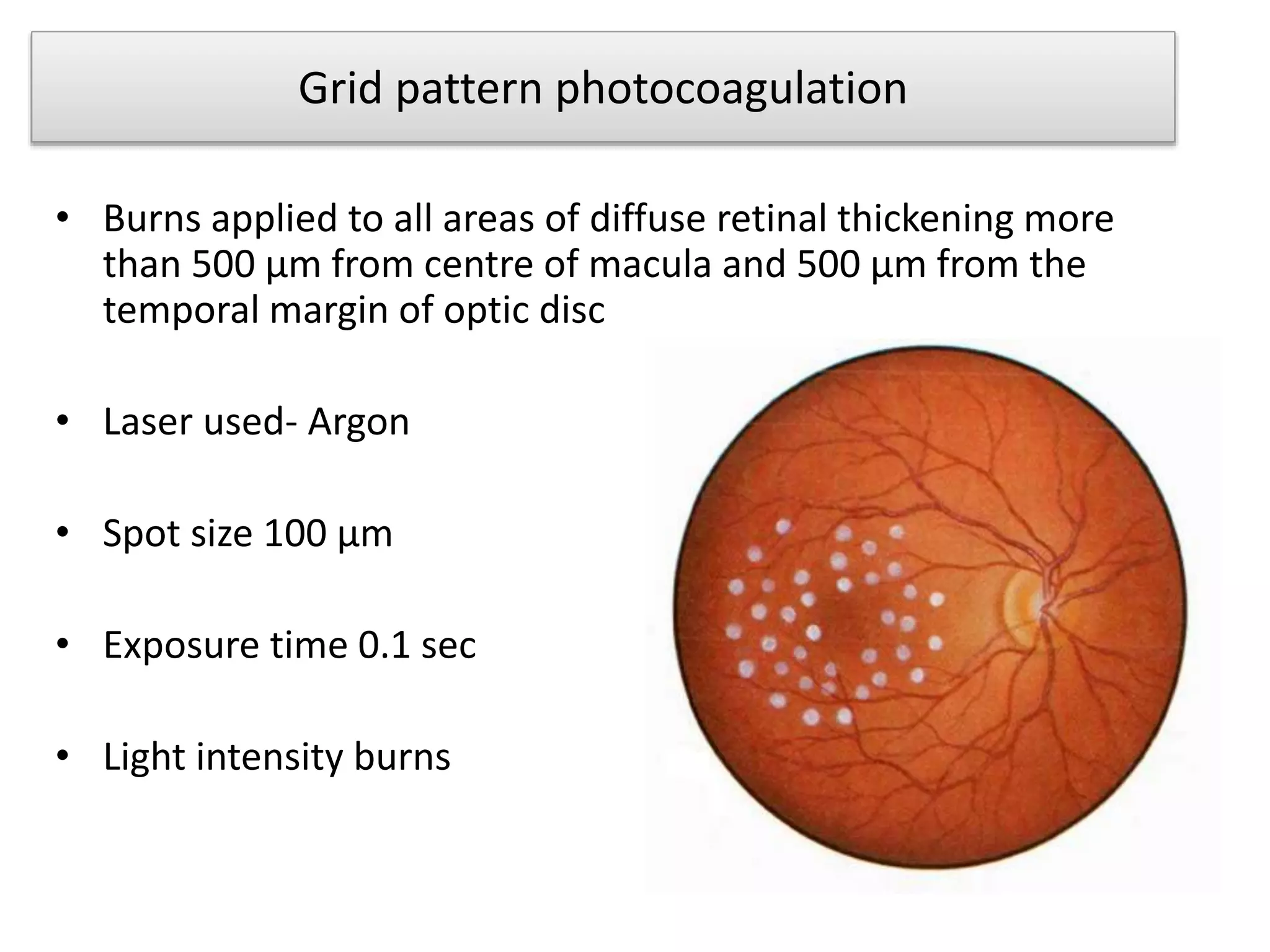
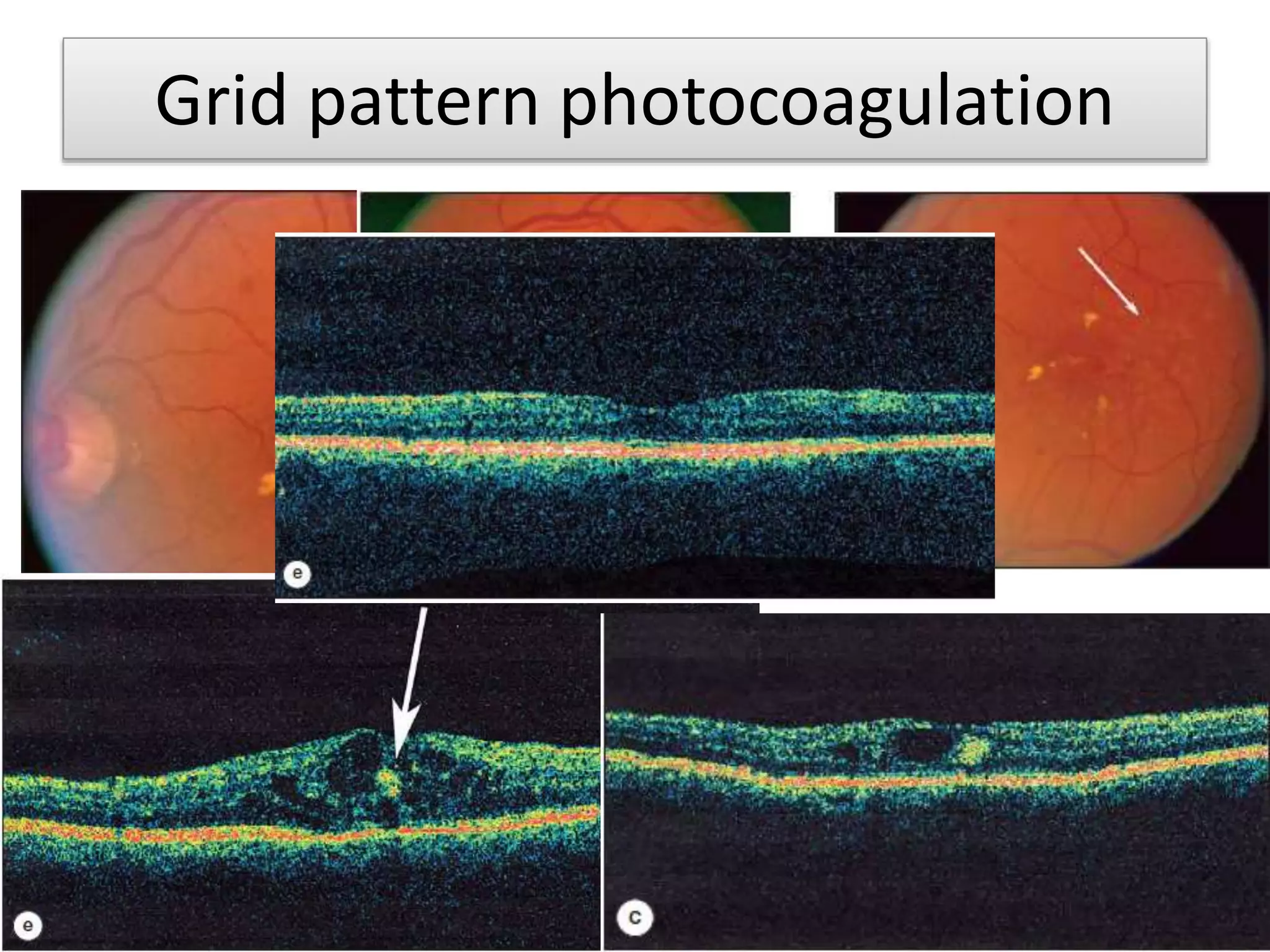
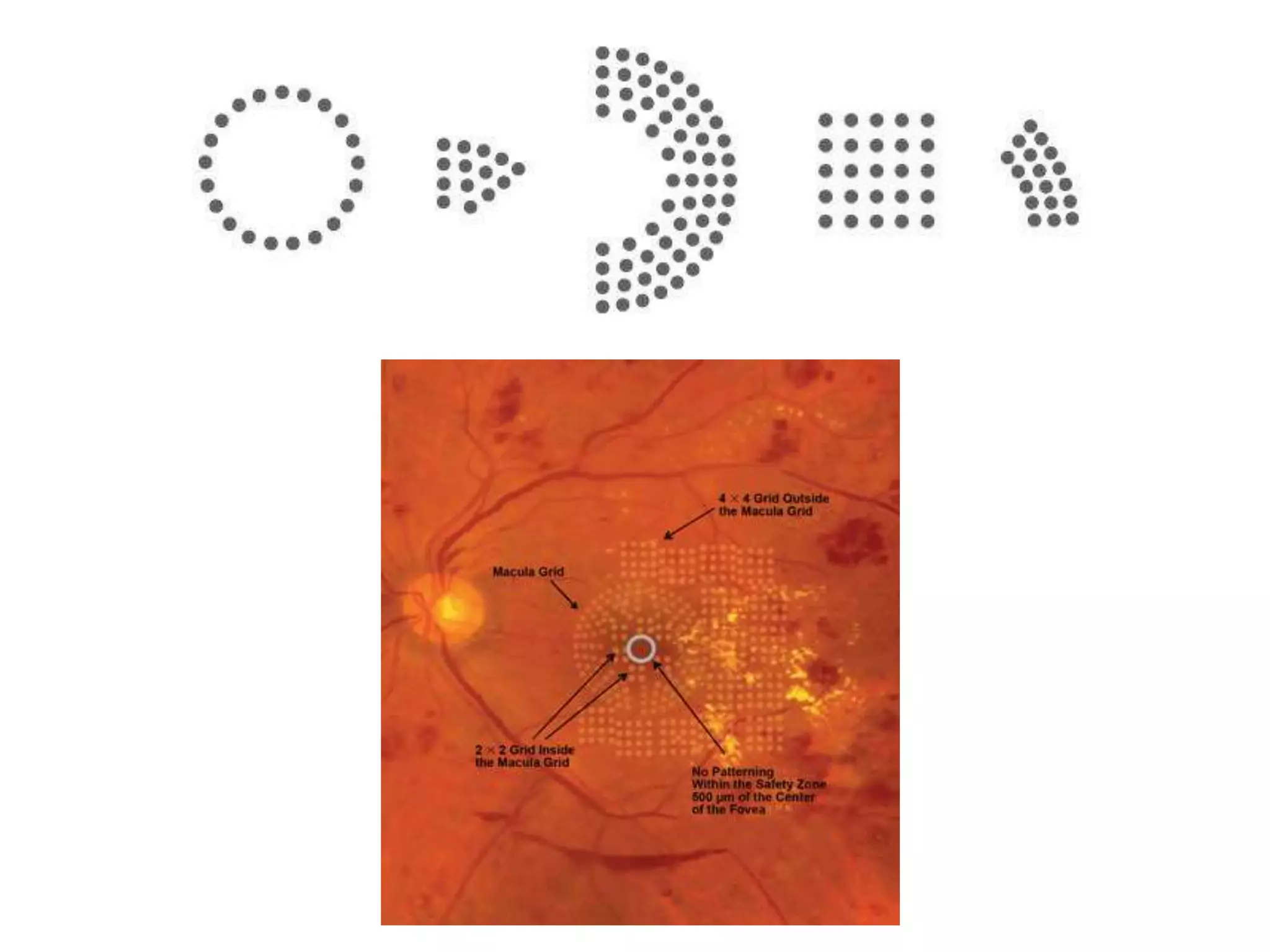
- Laser photocoagulation is effective for treating diabetic macular edema and proliferative DR. Focal laser is used for edema localized to the macula, while panretinal photocoagulation scatters wider burns throughout the retina to induce regression of new vessels.
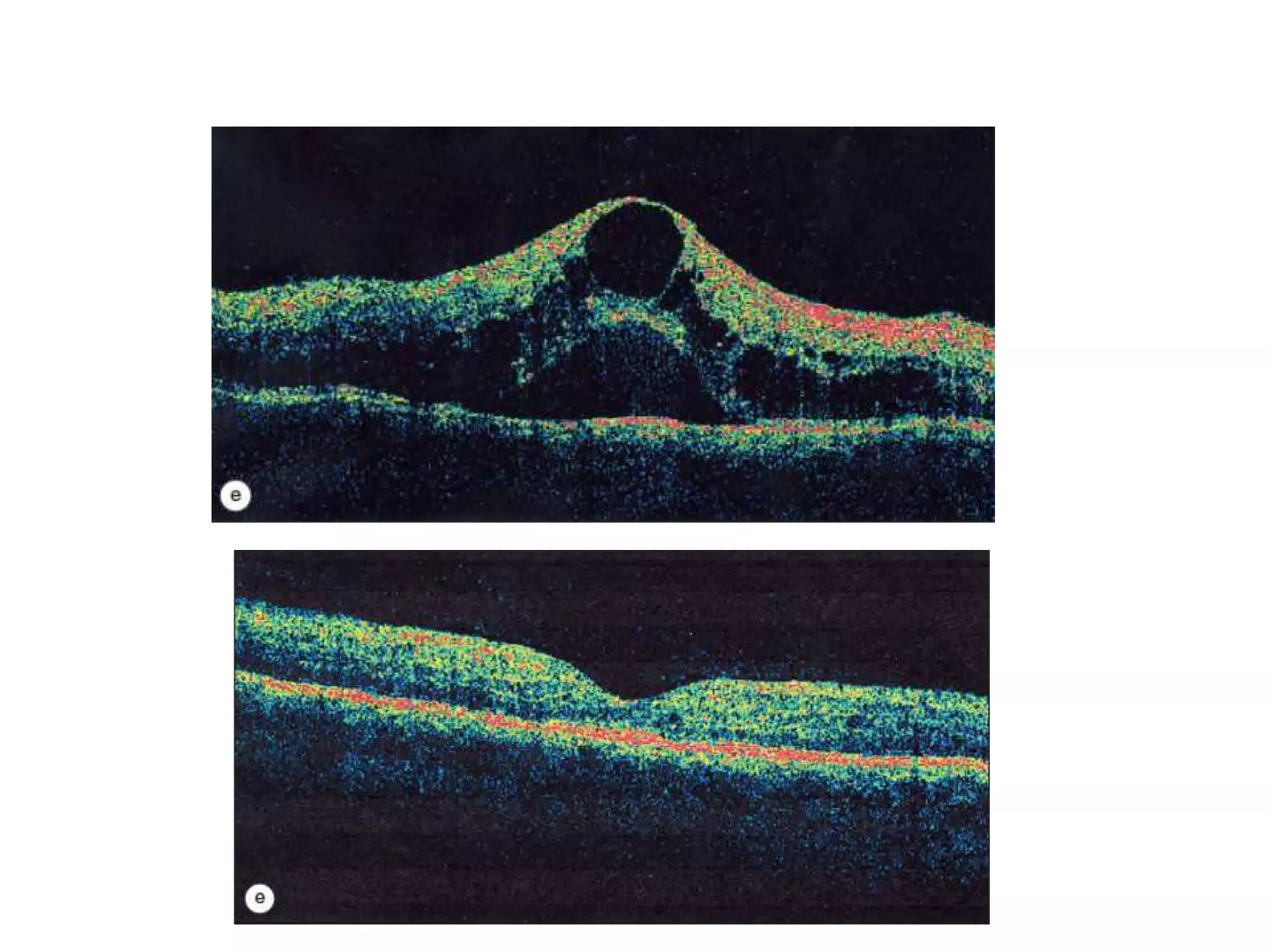
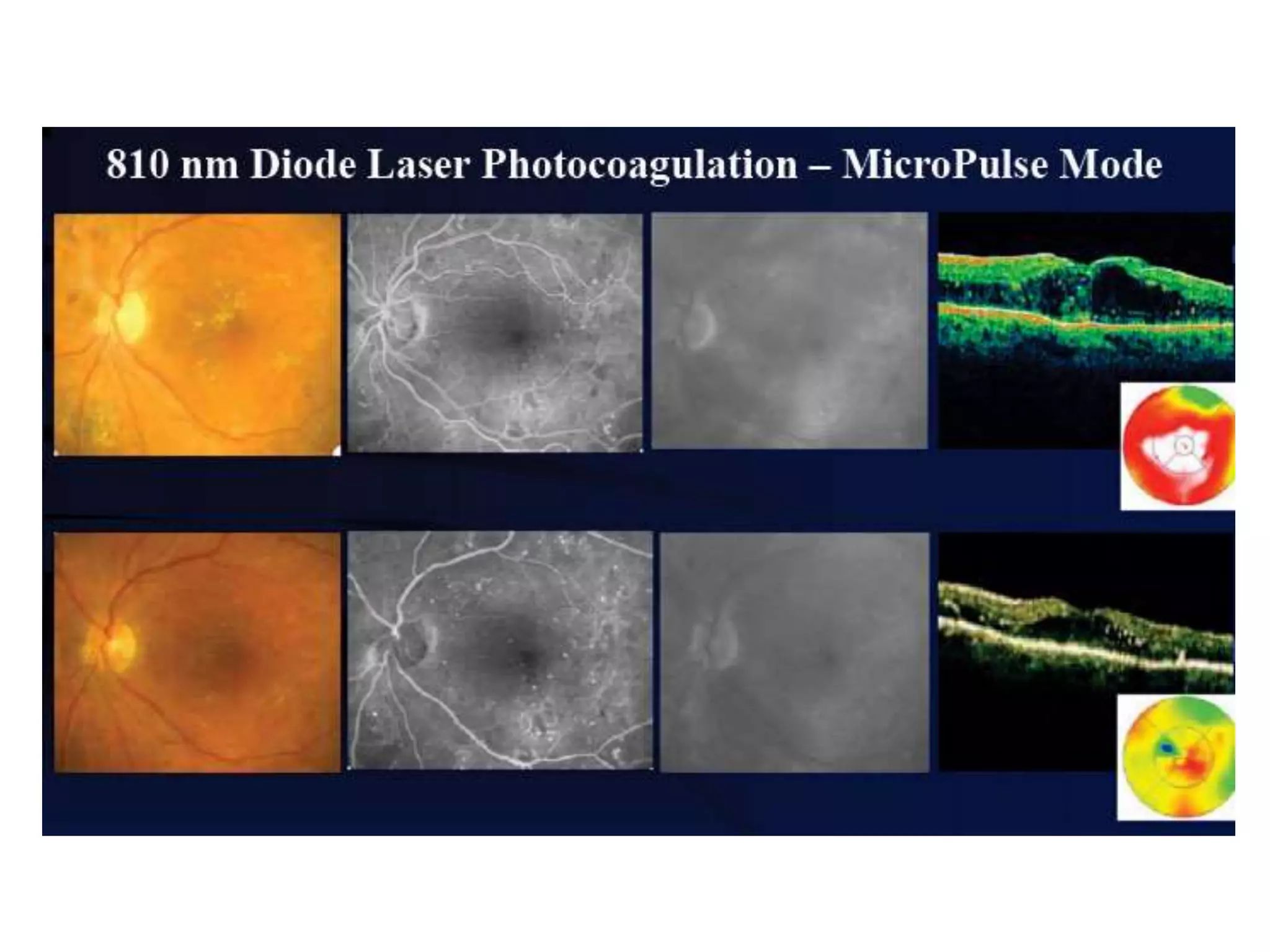
- Imaging like OCT and fluorescein angiography help diagnose and monitor DR and guide treatment.