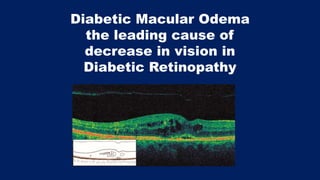
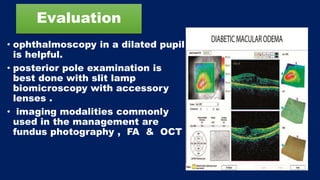
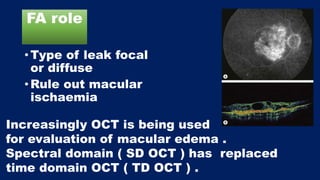
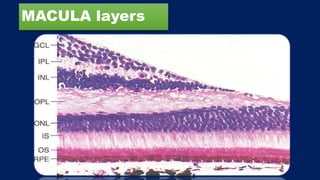
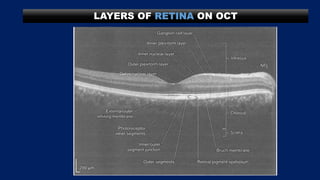
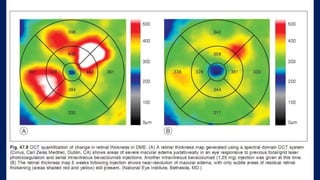
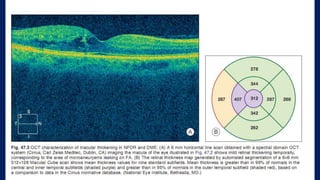
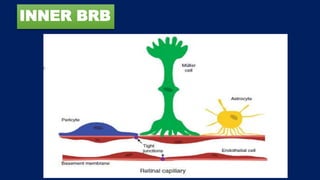
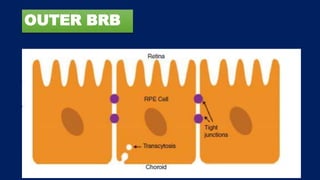
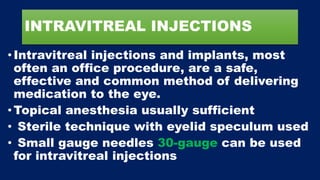
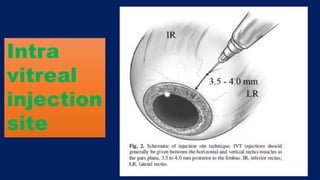
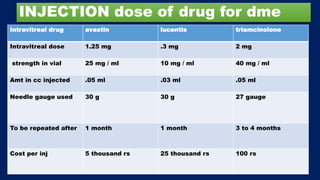
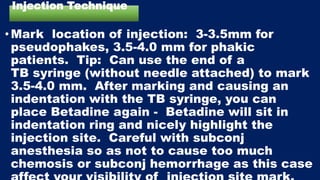
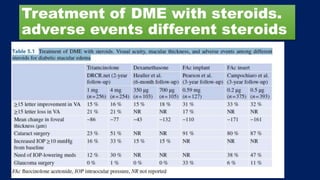
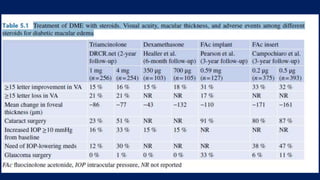
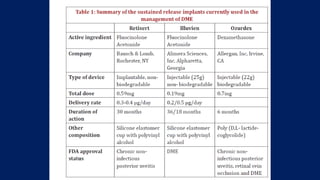
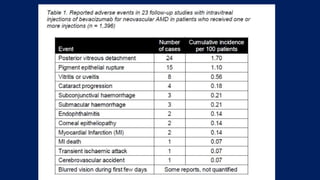
This document discusses diabetic macular edema (DME), including its prevalence, evaluation, treatment options, and intravitreal injection technique. DME is a leading cause of vision loss in diabetic retinopathy. Evaluation involves visual acuity testing, slit lamp examination, and imaging like OCT and FA. Treatment includes tight blood sugar and blood pressure control, as well as therapies like anti-VEGF agents, corticosteroids, and laser photocoagulation. Intravitreal injections are a common method to deliver these medications, with 30-gauge needles typically used for anti-VEGF drugs and 27-gauge for corticosteroids like triamcinolone.