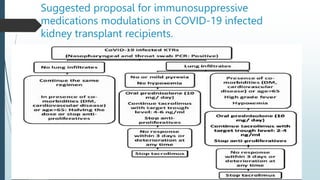
The document discusses management of kidney transplant recipients during the COVID-19 pandemic. It notes that transplantation programs have been reduced due to shortages of facilities and personnel devoted to COVID-19 care. Kidney transplant recipients are at high risk for COVID-19 infection due to immunosuppression. The challenge is controlling infection while maintaining immunosuppression to protect the transplant. The document reviews medications used to treat COVID-19 and potential interactions with immunosuppressive drugs in kidney transplant patients.





![Cut down of transplantation program
during COVID-19 outbreaks
Transplantation program faces many difficulties during this pandemic. These difficulties
included shortage in hospital facilities as most facilities are directed to treat COVID-19,
prioritization of ICU for COVID-19 patients, unknown risk of donor–recipient
transmission, unknown method for immunosuppressive medication modulation. and
the risk versus benefit of adding new immunocompromised patient to the society [5].
Kidney transplantation is not a safe procedure in pandemic areas except in emergencies
as in case of difficult vascular access
. In Middle East, including Egypt, transplantation rates have been decreased significantly
in comparison with 2019
Zidan A, Alabbad S, Tariq A. Position statement of transplant activity in the Middle East
in era of COVID-19 pandemic, transplantation. June 9, 2020. DOI:
10.1097/TP.0000000000003348](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-6-320.jpg)

![Hydroxychloroquine
Mechanism of action of hydroxychloroquine against COVID-19 is not well
established. However, it may induce changes in pH of cell membrane surface, thus
preventing viral fusion. Other theories suggest that it inhibit nucleic acid
replication, viral assembly, and release [26]. Since the beginning of the pandemic,
hydroxychloroquine became a basic medication in any treatment plan. A recent
French clinical trial reported significant reduction of viral load with
hydroxychloroquine [27]. Other reports showed conflicting results [28,29]. A report
was published in Lancet on May 22, 2020 against the use of hydroxychloroquine,
containing data from a large registry. However, it was retracted soon after owing to
significant methodology defect [30].](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-8-320.jpg)

![Hydroxychloroquine
Hydroxychloroquine may lead to different types of arrhythmias such as prolonged
QT interval and torsades de pointes. The incidence of arrhythmias reached 10% of
population in many studies [31]. The studies comparing between
hydroxychloroquine and placebo as prophylaxis showed no difference between
both groups, and patients who received hydroxychloroquine had more adverse
events [32,33]. Hydroxychloroquine undergoes hepatic metabolism as a substrate
of CYP2D6 and undergoes renal excretion via urine (15–25%) in metabolits and
unchanged drug forms [34]](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-10-320.jpg)

![ The COVID-19 treatment Guidelines Panel recommends against using lopinavir/ritonavir or other HIV protease inhibitors for the treatment of COVID-19,except in a clinical trial.
raise concerns about whether it is possible to achieve drug concentrations that can inhibit the SARS-CoV-2 proteases.
Lopinavir is boosted by RTV and cobicistat (COB), which inhibit cytochrome P3A4, thus affecting the trough level of different immunosuppressive medications [42]. Dramatic lowering of Table 1 Difference between
COVID-19, Pneumocystis jirovecii, and CMV infection among kidney transplant recipients (21–24) COVID-19 Pneumocystis jirovecii CMV Risky group All KTrs. Elderly, diabetic, cancer patients Severely
immunosuppressed patients with low CD4 count CMV IgG negative recipients with IgG positive donors Clinical data Fever is mild Cough, chest tightness Fever is mild to high, cough, chest tightness Fever, cough
chest tightness Extrapulmonary Acute kidney injury CMV can involve all organs Laboratory investigations Leucopenia Lymphocytopenia Positive throat and nasopharyngeal swabs for SARS-Cov-2 RNA Leucopenia
Lymphocytopenia Low CD4 count LDH increased Trophozites and cysts in BAL Leucopenia Neutopenia CMV inclusion bodies CMV RNA detection in blood Radiological findings Peripheral consolidation and ground
glass appearance Diffuse ground glass appearance which may be central and sparing periphery, rare consolidation Diffuse ground galss appearance and large consolidation Specific treatment No specific treatment
Sulfamethoxazole Pentamidine Clindamycin Caspofungin Ganciclovir, Valgancyclovir, Foscarnet, Cidofovir CMV, cytomegalovirus; IgG, immunoglobulin G; KTRs, kidney transplant recipients. 226 Journal of The
Egyptian Society of Nephrology and Transplantation, Vol. 20 No. 4, October-December 2020 [Downloaded free from http://www.jesnt.eg.net on Wednesday, October 28, 2020, IP: 196.154.141.223]
immunosuppressive medications (tacrolimus, cyclosporine and sirolimus) is needed to reach accepted trough level when combined with RTVboosted proteinase inhibitors [43,44]
Lopinavir/Ritonavir](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-12-320.jpg)
![Lopinavir/Ritonavir
. No changes in the dose of antiproliferatives are needed when combined with
ritonavir. However, it is suggested to stop antiproliferative during infection
episodes in general [45]](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-13-320.jpg)
![Tocilizumab
A hummanized monoclonal antibody called tocilizumab used in treating
autoimmune diseases can bind to interleukin-6 receptors (membrane type); thus, it
inhibits the release of inflammatory mediators and suppresses cytokine storm
triggered by hyperactivated immune system [49–51]. However, there are no data to
prove a drug-drug interaction between tocilizumab and immunosuppressive
medications [40]](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-14-320.jpg)







![Convalescent plasma
The use of convalescent plasma from recovered patients has been approved by US
Food and Drug Administration as a treatment for severe ill patients on an
individual basis [57]. Duan et al. [58] suggested therapeutic effect of convalescent
plasma in treating severely ill patients. The exact dose and timing is not well
defined. Another study by Shen et al. [59] reported that treatment of critical ill
cases responded to convalescent plasma. However, a large randomized clinical trial
(103 patients into two groups) by Ling et al. [60] found no statistical significant
improvement in time to clinical improvement between a group that used
convalescent plasma in addition to standard treatment and a control group that
used standard plasma alone.](https://image.slidesharecdn.com/covidscientificday-230530191055-e4130bbb/85/covid-scientific-day-pptx-22-320.jpg)