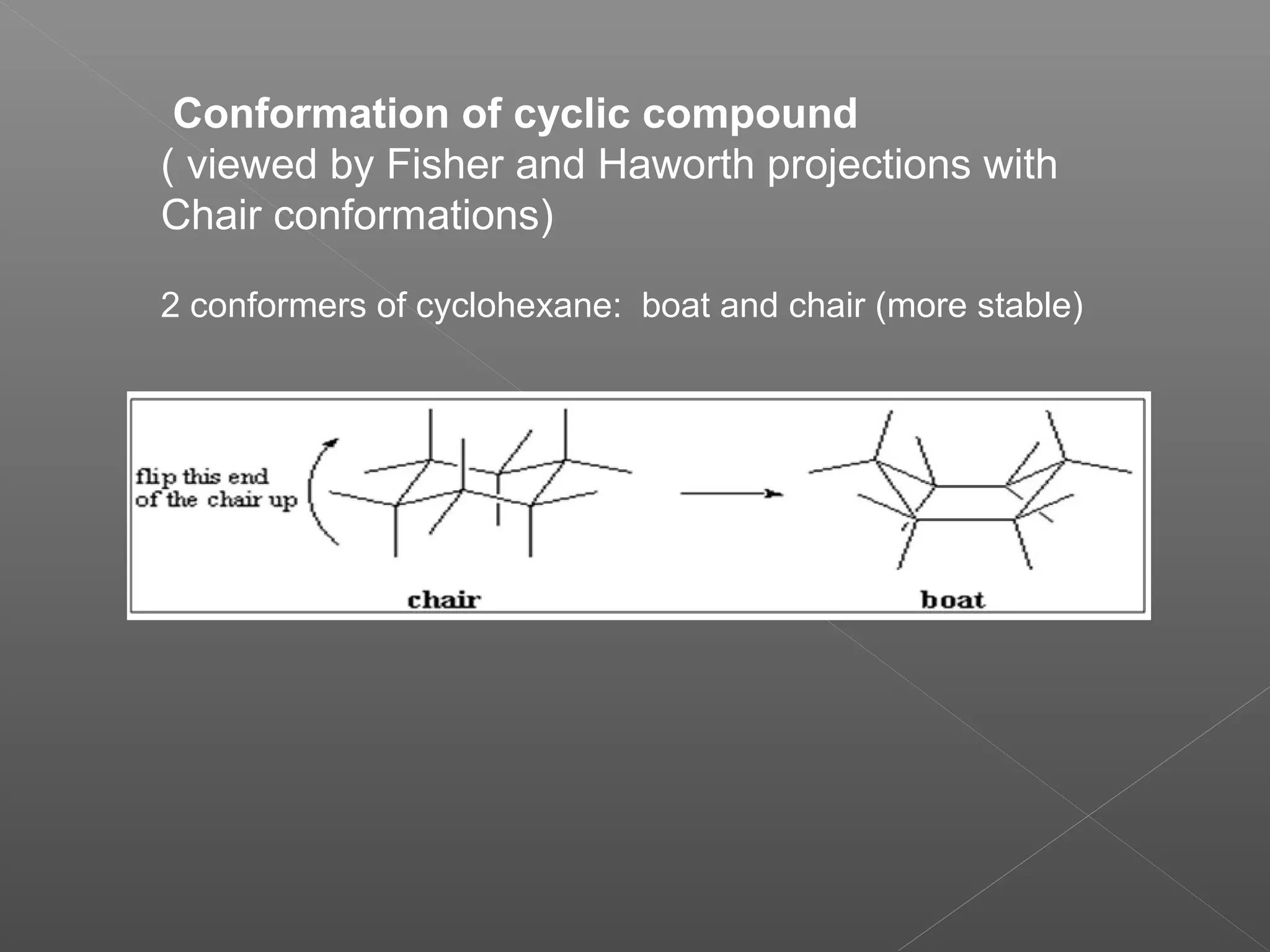
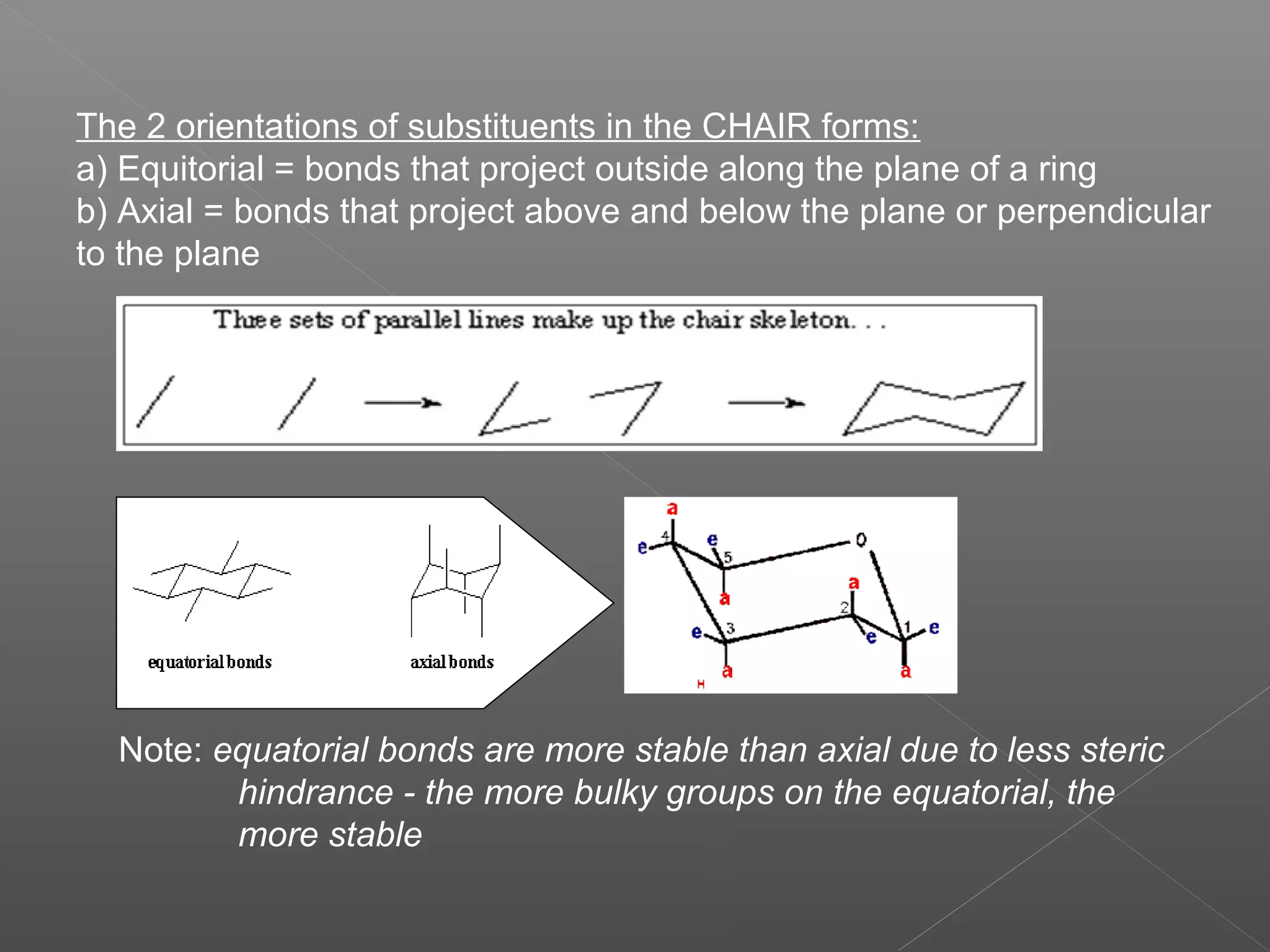
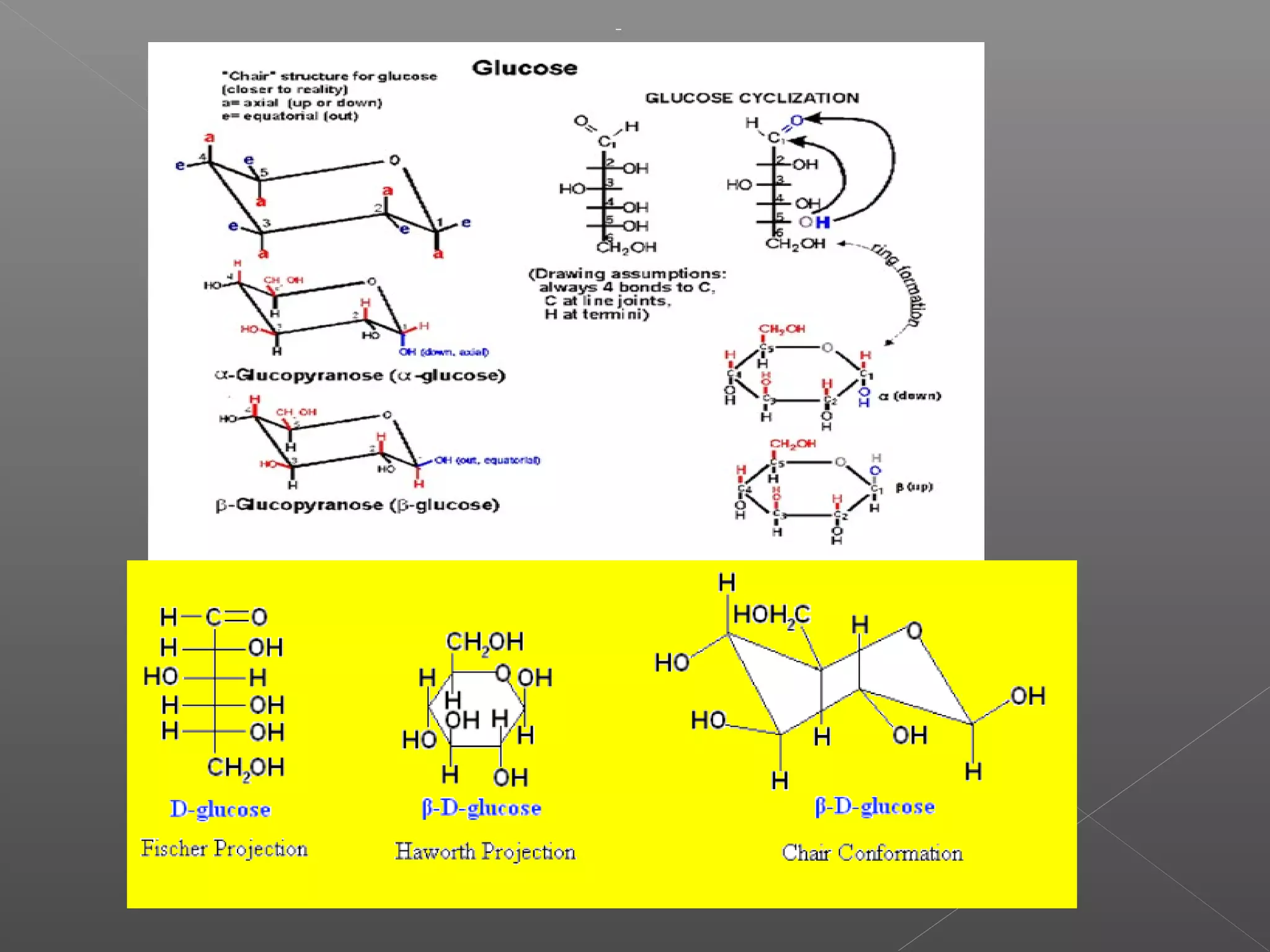
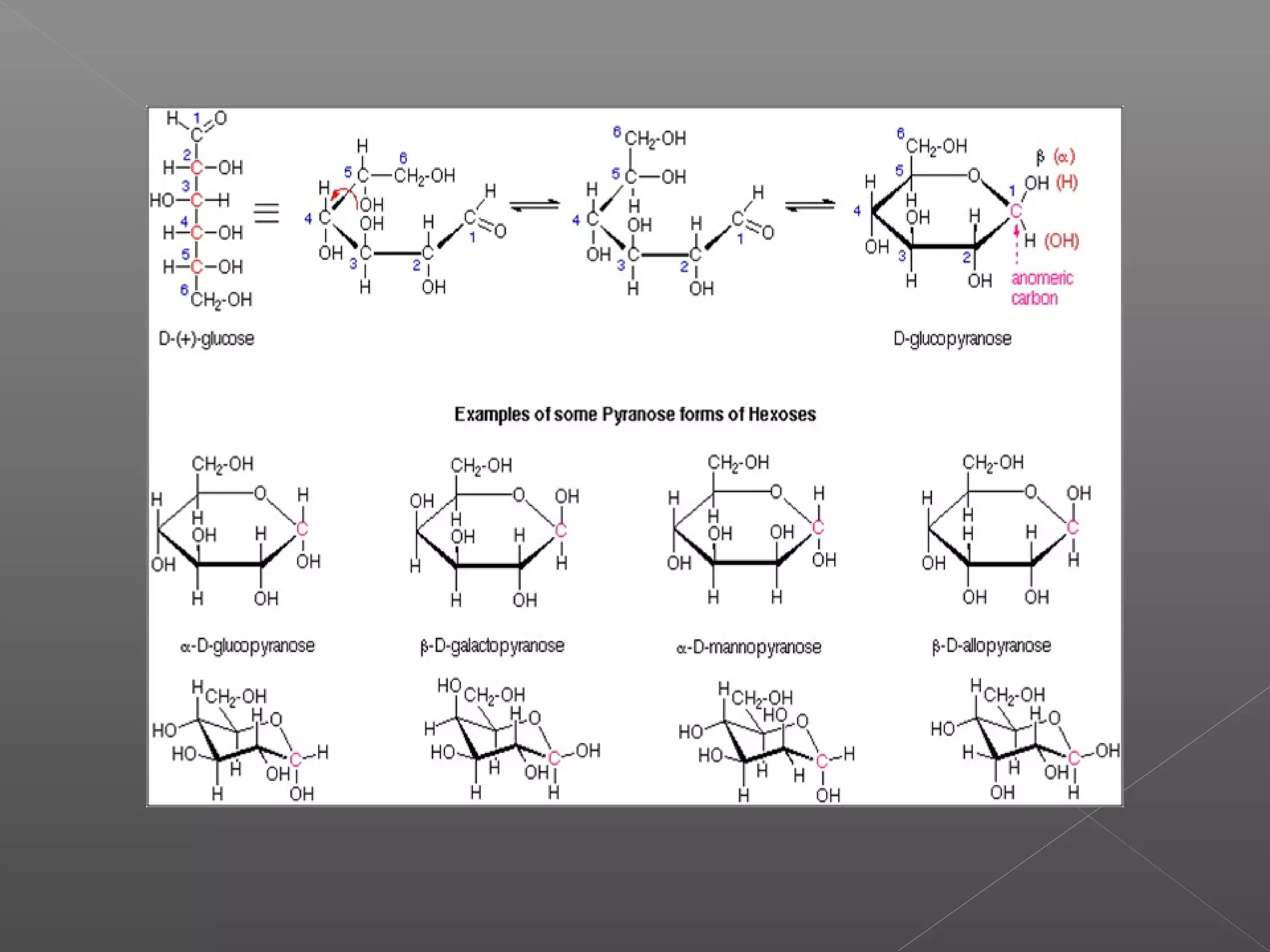
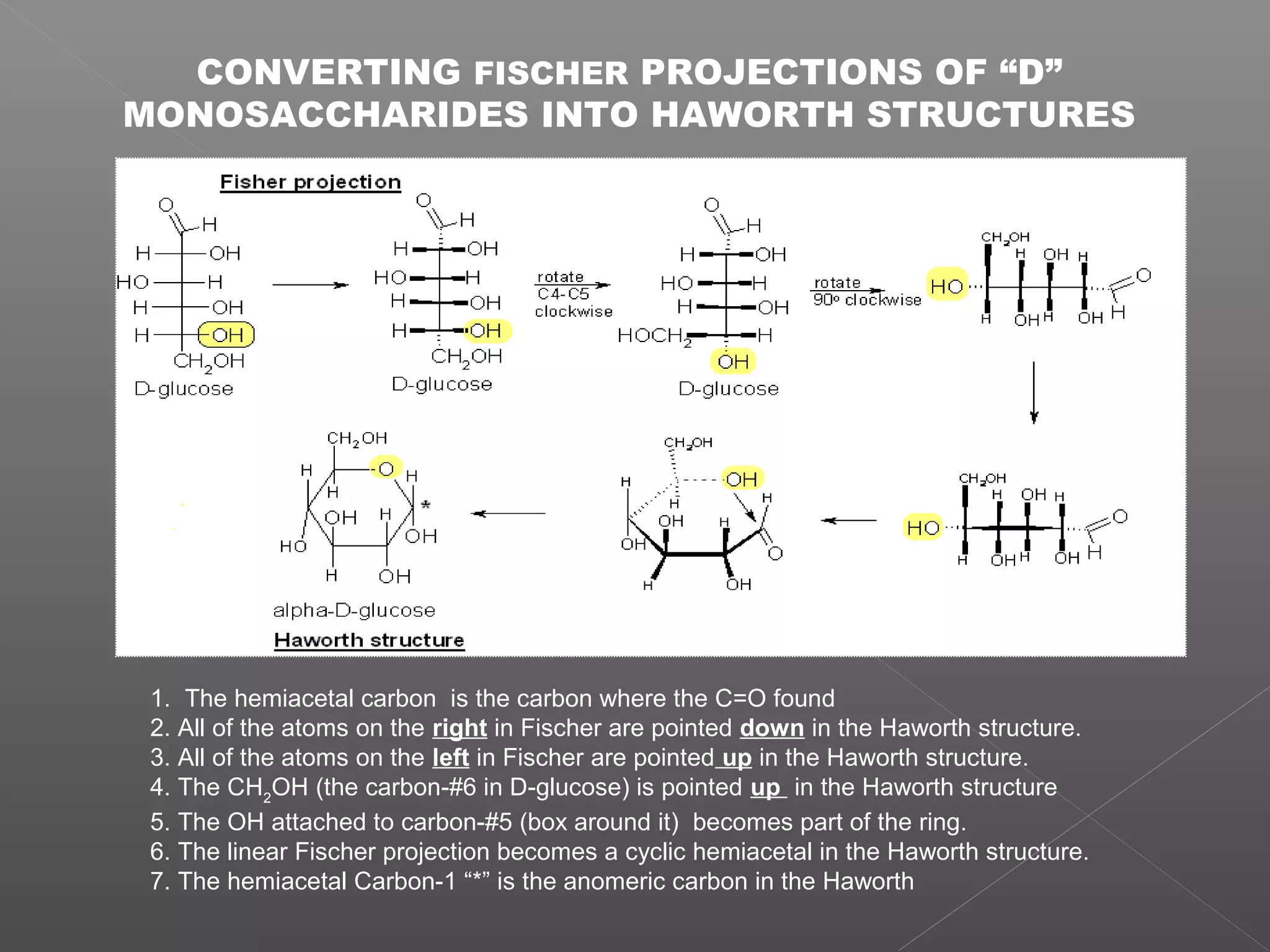
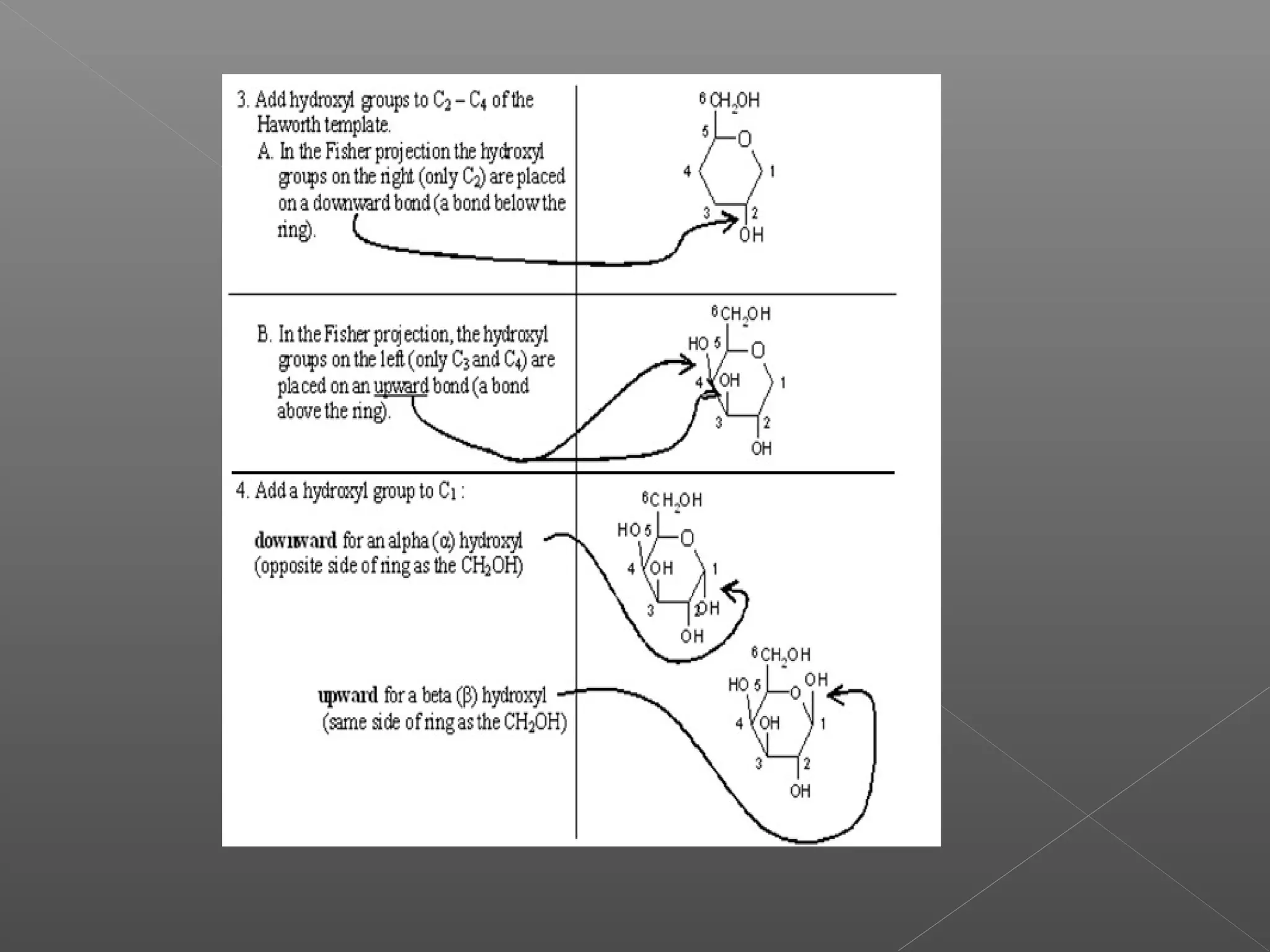
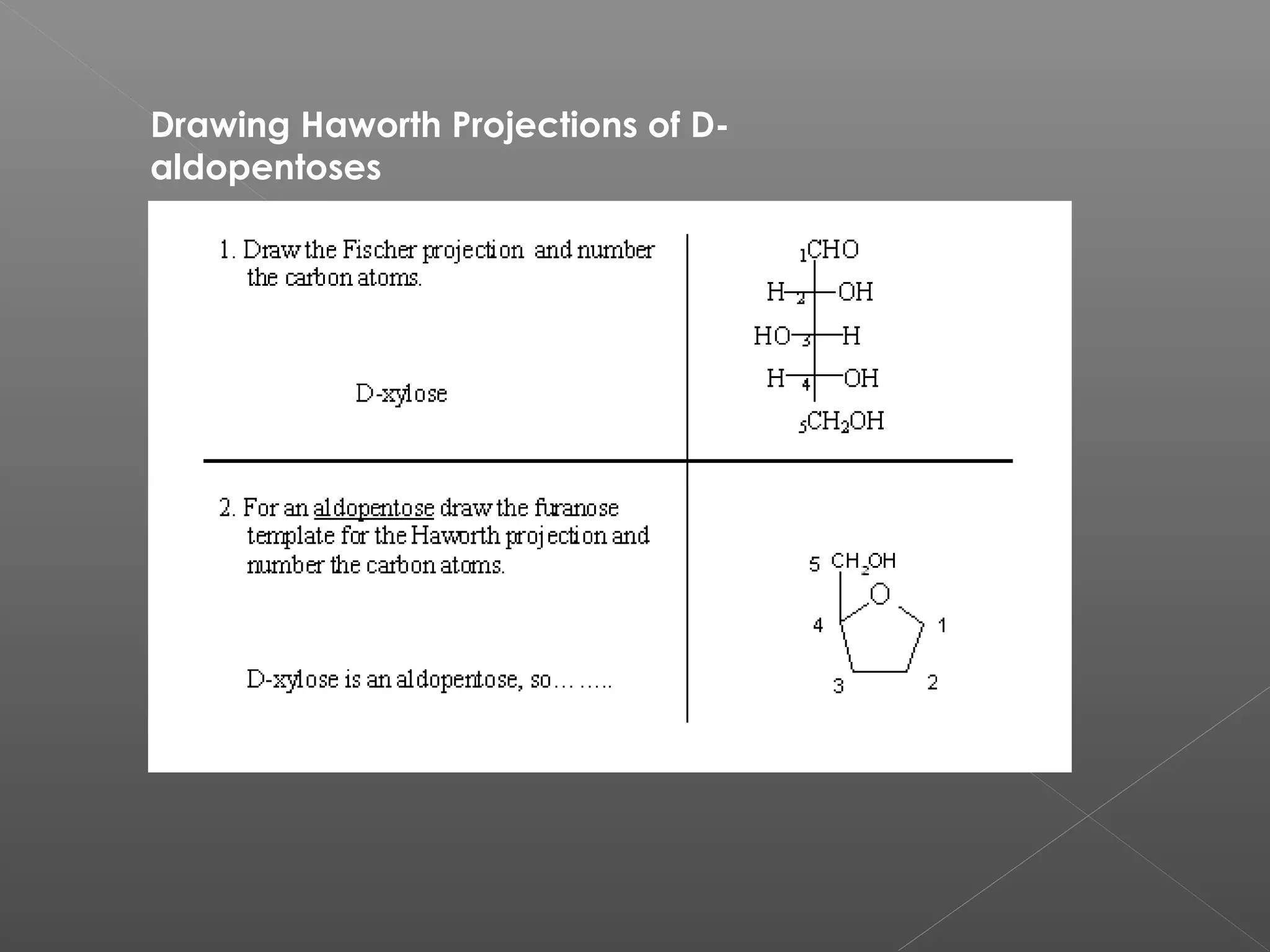
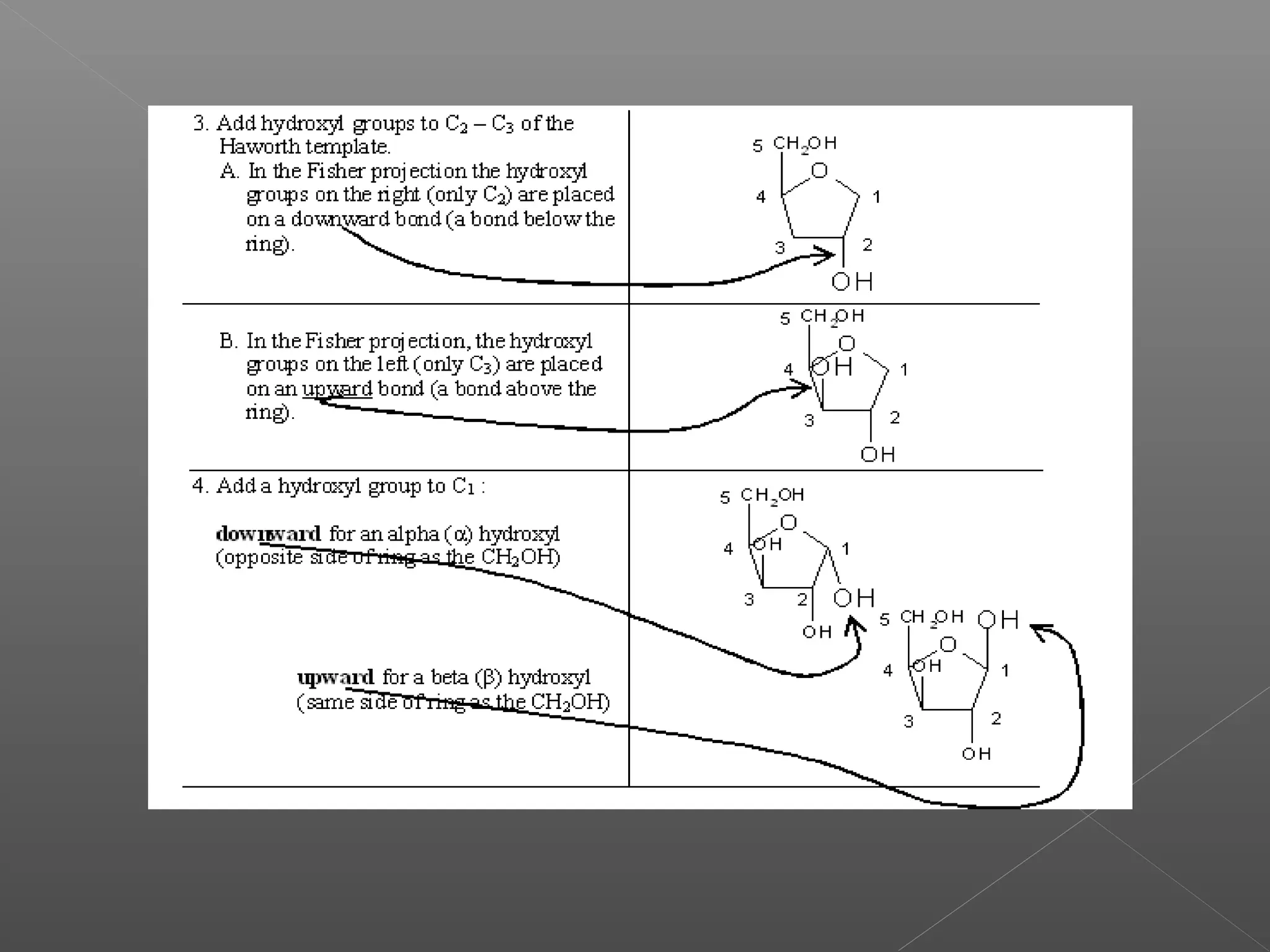
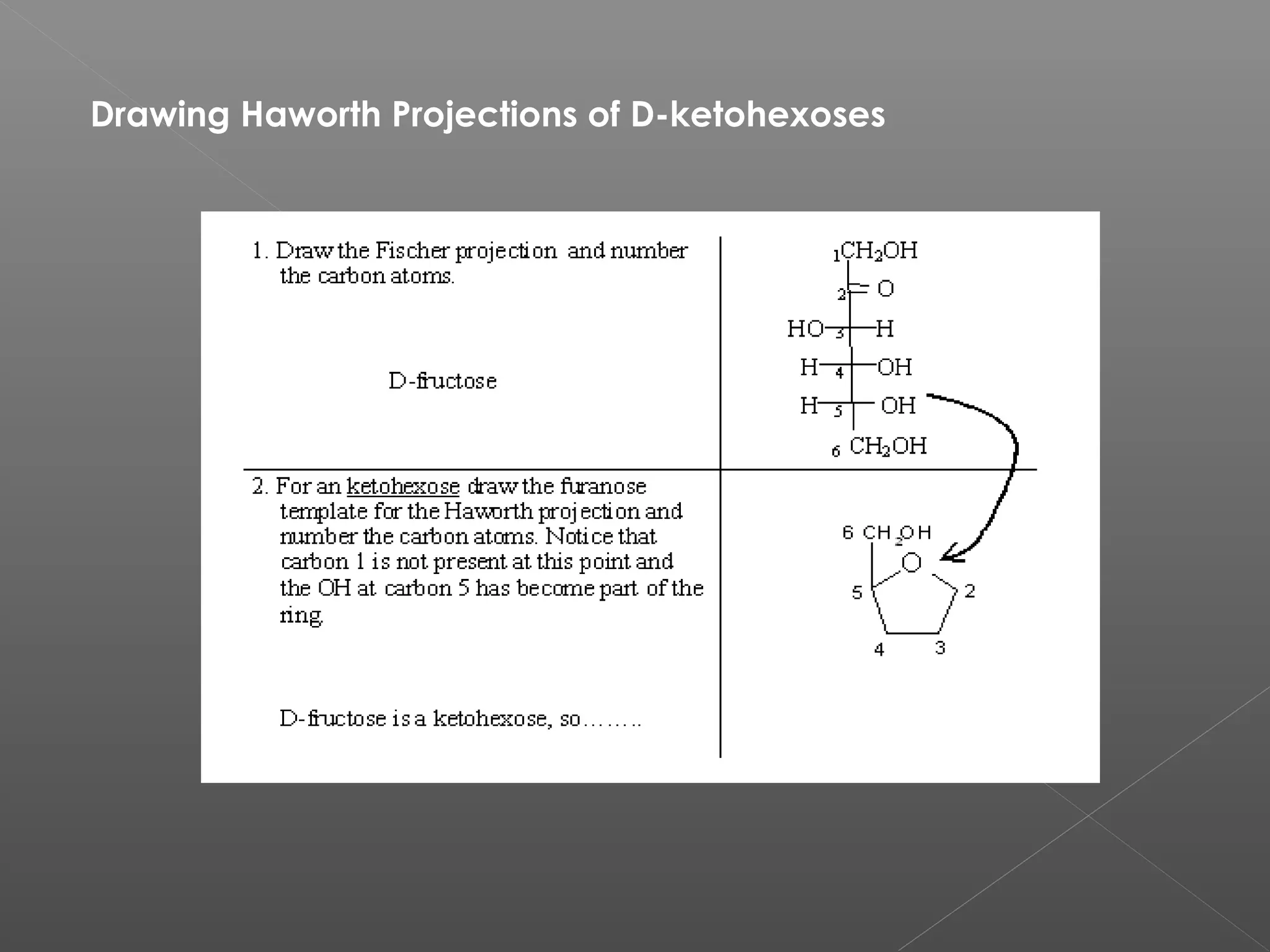
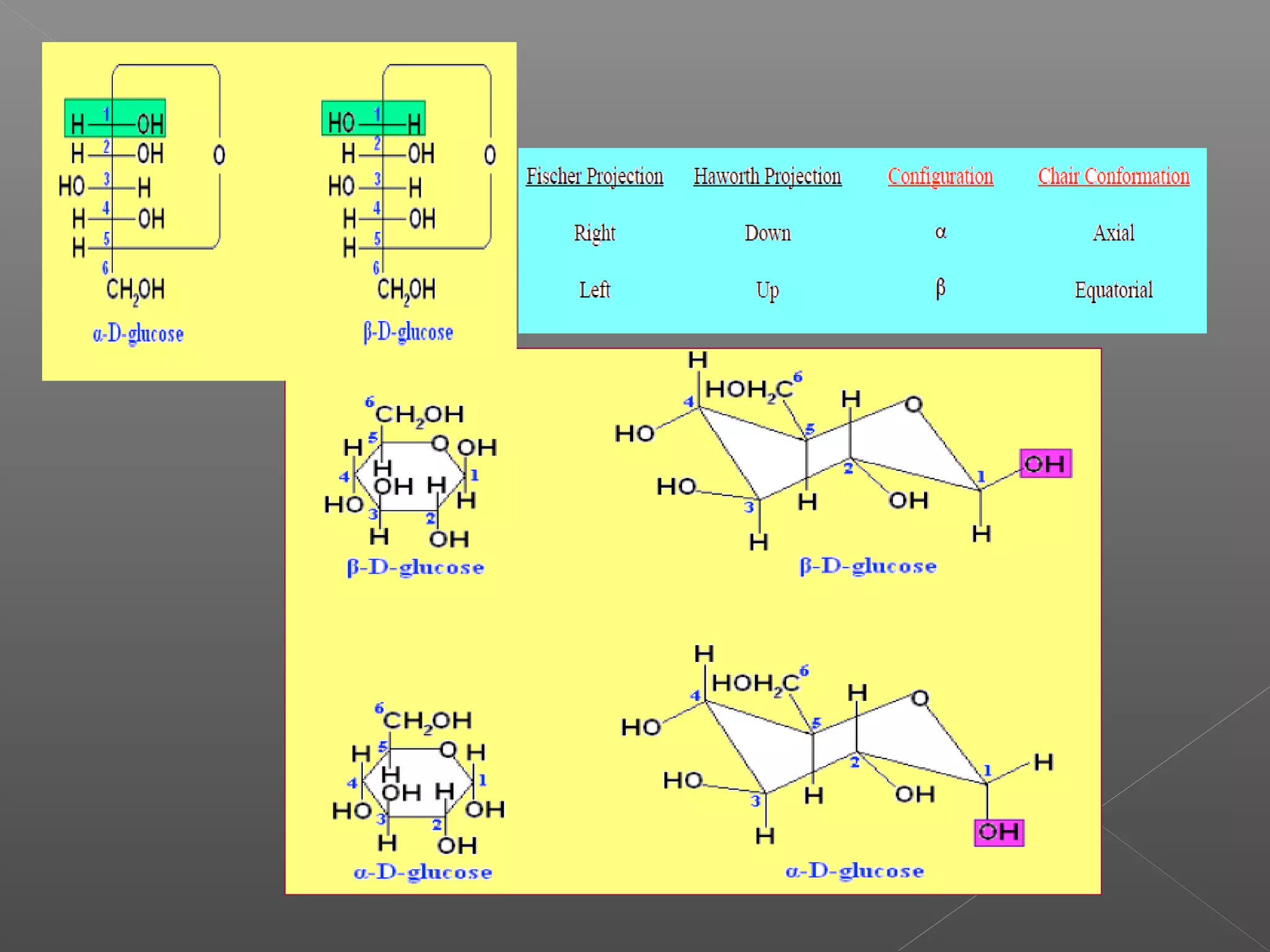
The document discusses conformations of cyclic compounds viewed through Fisher and Haworth projections. It notes that cyclohexane has two conformers, boat and chair, with chair being more stable. It also describes the orientations of substituents in the chair conformation as either equatorial or axial, with equatorial being more stable due to less steric hindrance. The document then provides instructions for converting Fischer projections of monosaccharides to Haworth structures.