1) Conductivity measurements are useful for monitoring water quality in power plants, raw water treatment plants, and district heating systems.
2) Key applications include monitoring for cooling water leaks, controlling chemical dosing and water purification systems, and detecting impurities.
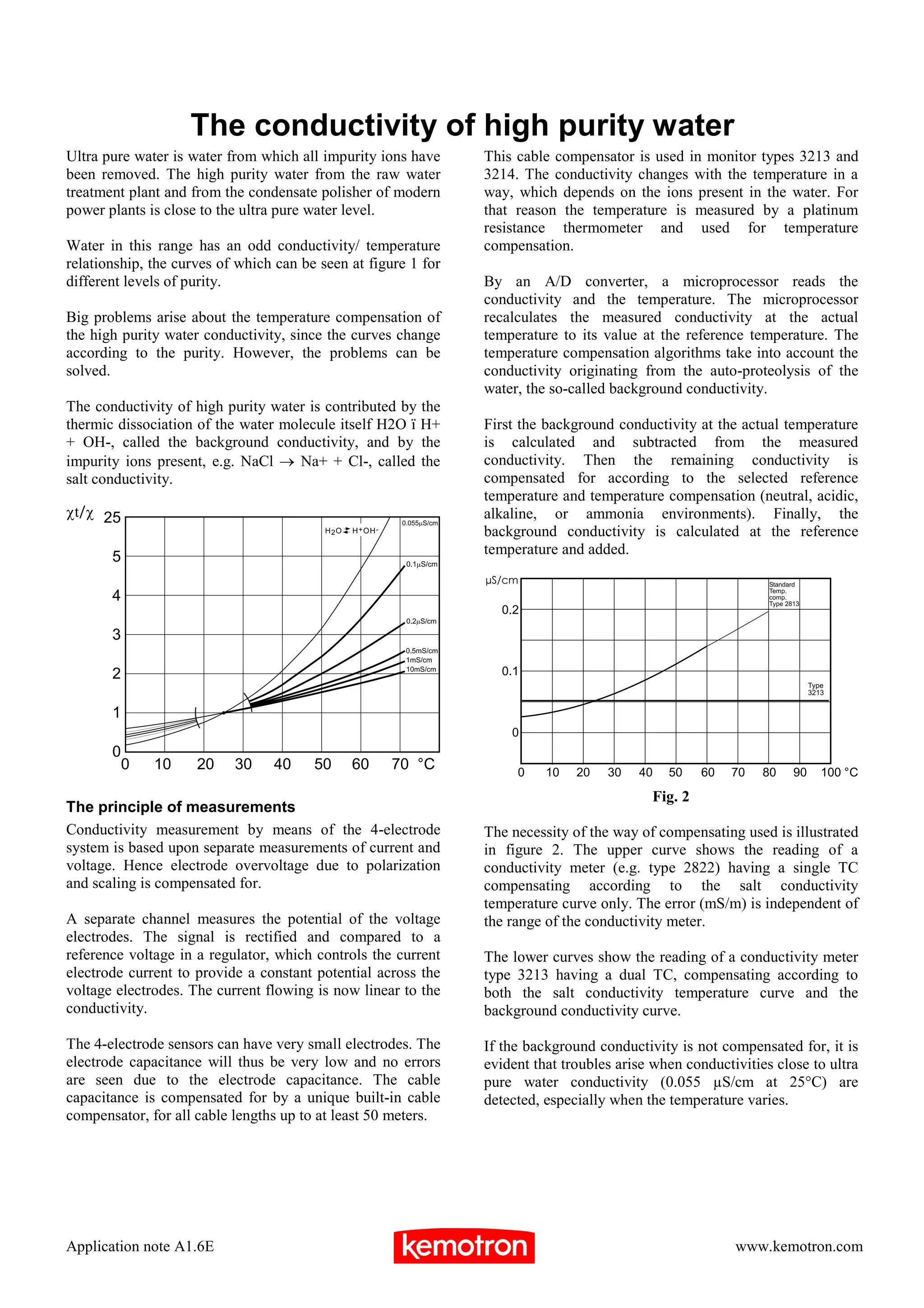
3) Proper temperature compensation is important for accurate conductivity readings, especially at very low conductivity levels close to that of ultra pure water.