More Related Content Similar to Chemistry-Introduction_150921_01e
Similar to Chemistry-Introduction_150921_01e (20) More from Art Traynor (20) 1. © Art Traynor 2011
Chemistry
Principles
Scientific Method
Section 1.2, (Pg. 5)
Zumdahl
?
? ?
?
Pro-Forma
(Secret Shopper)
Audit/Survey
Pro-Forma
(Secret Shopper)
Audit/Survey
Overture
Letter/Proposal
Overture
Letter/Proposal
Permissive Store
Visit or Mockup
Permissive Store
Visit or Mockup
Scientific
Method
?
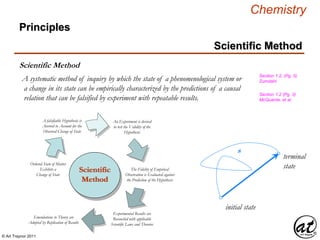
Ordered State of Matter
Exhibits a
Change of State
The Fidelity of Empirical
Observation is Evaluated against
the Prediction of the Hypothesis
Emendations to Theory are
Adopted by Replication of Results
s
initial state
terminal
state
A falsifiable Hypothesis is
Averred to Account for the
Observed Change of State
An Experiment is devised
to test the Validity of the
Hypothesis
Scientific Method
A systematic method of inquiry by which the state of a phenomenological system or
a change in its state can be empirically characterized by the predictions of a causal
relation that can be falsified by experiment with repeatable results.
Experimental Results are
Reconciled with applicable
Scientific Laws and Theories
Section 1.2 (Pg. 3)
McQuarrie, et al.
2. © Art Traynor 2011
Chemistry
Principles
Scientific Method
Scientific Method
A methodology for characterizing the behavior of a phenomenological system.
The system must admit
measurement
Section 1.2, (Pg. 4)
Phenomena Hypothesis
Empirical ( Measurable ) Theory ( Conjecture )
Falsifiable ( Refutable )
Replicable ( Reproducible )
Systematic
Experiment
Observation
Organized
Predictable
Qualitative
Quantitative
Procedure ( Operation)
Measurement
Number
Unit
Testing
Interpretive
Results
Model
Explanative
Refining
Consistent
System
Process ( Reaction )
Behavior
Logical Consequence
Population
Null – Alternative
Formulation
Analysis
Question
Simplicity
Principle of Parsimony
Occam’s Razor
Correlation Causation
Conflation
Selection Bias
Confirmation Bias
Hindsight Bias (Postdiction)
Control
Accuracy
Precision
Uncertainty ( Error )
Confirmation
Heuristic
Data
3. © Art Traynor 2011
Chemistry
Principles
Phenomenological System
Phenomenological System
An ordered state of matter possessed of attributes capable of being
characterized by a descriptive quantification
Section 1.2, (Pg. 5)
Zumdahl
4. © Art Traynor 2011
Chemistry
Principles
Scientific Law
Law of Science
It’s not so much a Law…
as a Good Idea
A durable declaration – premised on uncontroverted, aggregated observation,
such as to suggest universal validity –
inferring the operation of a causal relation
accounting for a particular change of state
within a phenomenological system.
A Scientific Law should not
be thought of a Theory that
has graduated status with
the accumulation of
additional evidence
Section 1.2, (Pg. 5)
Zumdahl
Section 1.2 (Pg. 3)
McQuarrie, et al.
Does not proffer Explanation !
Summarizes a Relationship
5. © Art Traynor 2011
Chemistry
Principles
Scientific Theory
A Theory is typically much
broader in scope than a
Scientific Law, which is much
more limited in the scope of
its explanatory compass
Section 1.2, (Pg. 5)Theory
A well-substantiated explanation –
premised on systematic observation and experiment
( typical of The Scientific Method ) –
inferring the existence of a causal relation
accounting for a particular change of state
within a phenomenological system.
A valid theory is characterized by the following:
Falsifiable Prediction with Consistent Accuracy
Empirically Premised on Observation and/or Experiment
Parsimonious as to Alternatives
Adaptive to Supplemental Data
Replicable Results
Explanation
explanandum
less
obvious
more
obvious
elucidation
A Theory can incorporate
several Scientific Laws
Represents a Unification of Ideas
( derivative of one or more laws )
Cannot be Proven Correct
6. © Art Traynor 2011
Chemistry
Principles
Scientific Theory
Section 1.2, (Pg. 5)Theory
A well-substantiated explanation –
premised on systematic observation and experiment
( typical of The Scientific Method ) –
inferring the existence of a causal relation
accounting for a particular change of state
within a phenomenological system.
Logical Positivistic
Explanation
explanandum
less
obvious
more
obvious
elucidationNumerous epistemological interpretations of the Scientific
Method have emerged to enrichen its vitality:
Theories are to be considered as analogous to mathematical
axioms – a propositional form
7. © Art Traynor 2011
Chemistry
Principles
Scientific Theory
Section 1.2, (Pg. 5)Theory
A well-substantiated explanation –
premised on systematic observation and experiment
( typical of The Scientific Method ) –
inferring the existence of a causal relation
accounting for a particular change of state
within a phenomenological system.
Explanation
explanandum
less
obvious
more
obvious
elucidationNumerous epistemological interpretations of the Scientific
Method have emerged to enrichen its vitality:
Models
Theories are to be considered as analogous to mathematical
axioms
8. © Art Traynor 2011
Chemistry
Principles
Hypothesis
Hypothesis
A conjecture arising from the formulation of an empirical (scientific) question
which supplies a plausible explanation
to account for a particular change of state observed to occur within a
phenomenological system.
Null Hypothesis
Alternative Hypothesis
The conjecture that experiment will falsify a relation posited to account for a
particular change of state observed to occur within a phenomenological system.
The preferred conjecture that experiment will verify a relation posited to account for
a particular change of state observed to occur within a phenomenological system.
Essentially synonymous with
Conjecture, however
Conjecture is more formally
defined in Mathematics as a
Proposition for which a Proof
has yet to be stated
9. © Art Traynor 2011
Chemistry
Principles
Hypothesis
Hypothesis
A conjecture arising from the formulation of an empirical (scientific) question
which supplies a plausible explanation
to account for a particular change of state observed to occur within a
phenomenological system.
A distillation of Question
Formulation Criteria as
proposed by Dietrich Dörner
with subsequent expansion
/refinement by Joachim
Funke
Transparency ( Intransparency )
Polytely ( Objective Multiplicity/Profusion , “ mission creep ” )
Complexity
Dynamics
10. © Art Traynor 2011
Chemistry
Principles
Experiment
Experiment
An ordered procedure by which to establish the validity of a Hypothesis purporting
to characterize a phenomenological system or to otherwise account for any observed
change of state within the system.
Observation
Qualitative – phenomena are characterized according to Class Equivalencies
Color
Odor
Reactivity
Quantitative – susceptible to measurement Section 1.2 (Pg. 5)
McQuarrie, et al.
11. © Art Traynor 2011
Chemistry
Principles
Chemistry
Chemistry
A discipline within the Physical Sciences dedicated to the explication of the
Composition
Structure
Properties
Changes of State
Of ( or within ) a phenomenological system.
Chemistry seeks to describe the properties of
Individual Atoms ( Elements )
Bonding
Intermolecular Forces
Reactions
Chemistry is regarded as a “ Central Science ” as its precepts are of equal
explanatory force within the related disciplines of Physics, Geology, and Biology
Wiki “Chemistry”
12. © Art Traynor 2011
Chemistry
Principles
Chemistry
Chemistry
The etymology of “ Chemistry ” can be traced through several alternatively
plausible origins
Alchemy
al-kīmīā ( Arabic )
χημεία or chemeia ( Greek – “ cast together ” )
Khem – ( ancient Greek name for Egypt, for their prowess in metalworking )
Wiki “Chemistry”
Kim Mi – ( Chinese for “ the secret of Gold ” )
13. © Art Traynor 2011
Chemistry
Principles
Measurement
Measurement
A quantity ascertained by observation characterized by:
Magnitude
Unit
The Modulus or Absolute Value, | m | assigned to
represent the observation.
The ordering Metric by which a Relation with the magnitude
of the observation might be expressed as a scaled ratio.
Section 1.3, (Pg. 7)
Uncertainty
A parameter associated with an observation relating the
probabilistic dispersion of the Magnitude.
Measurand – that quantifiable
aspect of a phenomenological
system by which class
equivalences can be metrically
determined
Measurand – the object of the
measurement
Measurement – the estimation
of the ratio (typically attribute
differences) between the
magnitude of a continuous
quantity and a unit magnitude
of the same class.
14. © Art Traynor 2011
Chemistry
Principles
Measurement – Qualities of Unit Quantities
Unit quantities can be classed as one of two types:
Extensive Quantities
Classes of phenomena admitting a Concatenation operation .
Section 1.3, (Pg. 7)
Also known as Base Quantities
Examples: Length, Mass, Time, Electrical Resistance,
Plane Angle, etc.
Intensive Quantities
Classes of phenomena not admitting a Concatenation operation .
Examples: Temperature, etc.
Measurement
15. © Art Traynor 2011
Chemistry
Measurement
SI Units
Systèm Internationale ( SI )
Fundamental SI Units
Mass
Length
Time
Temperature
Electric Current
Substance Cardinality
Luminous Intensity
kilogram
meter
second
Kelvin
ampere
mole
candela
kg
m
s
K
A
mol
cd
There are only seven
Fundamental SI Units
“Dimension” is rigorously
defined as the indexed
product of a fundamental
physical Metric ( of which
there are precisely five )
Dimension Name Abbreviation
Section 1.4, ( Pg. 11 )
Variable
m
l
t
T
I
n
The notion of “Dimension”
is hierarchically distinct
from the notion of a
“ Scale Unit ”
“Dimension” is conceived
to describe a distinct
phenomenological
equivalence class on
which a relation to a
measurand can be defined
by a scaled ratio
A “Fundamental” unit is
one from which every
other measure in its
phenomenological class
can be derived (akin to
vector Basis, or Linearly
Independent Set)
16. © Art Traynor 2011
Chemistry
Measurement
SI Units
Systèm Internationale ( SI )
Additional SI (?) Units
Physical Quantity Name Abbreviation Variable
Volume Liter L v
Inductance Henry H L
Units which can be “ factored ”
into other units, or are com-
positions of one or more
Base Units are not fundamental,
( e.g. a volume in Liters can be
equally stated in terms of length
as in 1mL = 1cm3 , or velocity
expressed in mi/hr )
Capacitance Farad F C
Resistance Ohm Ω R
Electro-Motive Force Volt V E
Electric Charge Coulomb C Q
17. © Art Traynor 2011
Chemistry
Measurement
SI Units
Systèm Internationale ( SI )
Additional Units – Compound Units
Physical Quantity Name Abbreviation Variable
Density
Kilogram
L
Compound Units
A composition of measures drawn from two or more of the fundamental units.
Liter
kg
⍴= kg · L – 1
v
m
=
18. © Art Traynor 2011
Chemistry
Measurement
SI Units
Systèm Internationale ( SI )
Fundamental SI Units
Mass
A measure characterizing the Inertia of a body, or its resistance to a change in its state
of motion ( i.e. the Force necessary to impart a certain acceleration ).
Weight
The response of a Mass to the Force of Gravity .
Temperature
A measure of the relative tendency of heat to escape a radiant body .
Section 1.2 (Pg. 5)
McQuarrie, et al.
19. © Art Traynor 2011
Chemistry
Measurement
SI Units
10 – 24
10 – 21
10 – 18
10 – 15
10 – 12
10 – 9
10 – 6
10 – 3
10 – 2
10 – 1
Yocto y 10 24YottoY
Zepto z 10 21ZettaZ
Atto a 10 18ExaE
Fempto f 10 15PetaP
Pico P 10 12TeraT
Nano n 10 9GigaG
Micro μ 10 6MegaM
Milli m 10 3Kilok
Centi c 10 2
Deci d 10 1Dekada
Hectoh
Systèm Internationale ( SI )
Unit Prefixes
20. © Art Traynor 2011
Chemistry
Measurement
SI Units
Systèm Internationale ( SI )
Additional SI Units
1m = 10dm
10dm = 1m
1m = 10dm
1 meter = 10 decimeters
Length Volume
1 m3 = 1,000 dm3
or 1,000 L
1dm3 = 1L
1cm3 = 1mL
1 decimeter = 10 centimeters 1 dm3 = 1,000 cm3
or 1 mL
1dm = 10 cm
1dm = 10 cm
1dm = 10 cm
21. © Art Traynor 2011
Chemistry
Measurement
Uncertainty
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
A parameter associated with an observation relating the
probabilistic dispersion of the Magnitude.
As previously noted (essential
elements of a Measurement )
Etiology of Error
Precision & Accuracy
Significant Figures ( Sig Figs )
22. © Art Traynor 2011
Chemistry
Measurement
Error
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
A parameter associated with an observation relating
the probabilistic dispersion of the Magnitude.
As previously noted (essential
elements of a Measurement )
Etiology of Error
n Systematic Error
Error which is introduced into a measurement
by a defect in the measurement device
n Sensitivity Error
Error which is introduced into a measurement
by an overspecification of device precision
Characterized by a posteriori
“ predictable ” error
23. © Art Traynor 2011
Chemistry
Measurement
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
A parameter associated with an observation relating the
probabilistic dispersion of the Magnitude.
As previously noted (essential
elements of a Measurement )
Etiology of Error
n Random Error
Non-systematic error ( typically ) introduced into
the measurement by factors other than those
inhering to the measurement device ( e.g. human
factors )
Characterized by a posteriori
“ unpredictable ” error
This species of error can be systematically
characterized by a Probability Density
Function ( PDF )
o
Error
24. © Art Traynor 2011
Chemistry
The discrepancy between an exact value and some approximation
( measurement ) of it
Measurement
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Error
Etiology of Error
n Approximation Error
Swok, Section 11.5, Pg. 560o If “ E” represents error in an approximation, the approximation
can be considered accurate to “ k” decimal places if the modulus
of “ E” is less than the product of five-tenths and ten indexed
to the minus “ k”
1-decimal place Accuracy | E | < 0.5 x 10 – 1 = 0.0.5
①
Moving 1 position
in the “+” direction
k-decimal place Accuracy | E | < 0.5 x 10 – k
2-decimal place Accuracy | E | < 0.5 x 10 – 2 = 0.0.0.5 Moving 2 positions
in the “+” direction
①②
3-decimal place Accuracy | E | < 0.5 x 10 – 3 = 0.0.0.0.5 Moving 3 positions
in the “+” direction
①②③
25. © Art Traynor 2011
Chemistry
The discrepancy between an exact value and some approximation
( measurement ) of it
The magnitude of the difference between the exact value
and the approximation ( e.g. ± 0.05m )
Magnitudes are always expressed
as absolute values and are thus
always positive numbers
Measurement
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Error
Etiology of Error
n Approximation Error
n Absolute Error ( Tolerance )
26. © Art Traynor 2011
Chemistry
Measurement
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Error
Etiology of Error
n Relative / Fractional / Percentage Error
The absolute error expressed
as a ratio of the exact value ( e.g. 56.47 ± 0.02mm )
0.02mm
56.47mm
=
Absolute Error
Exact Value
= 0.0004 → ( 0.0.0.04 ) ( 100% ) = 0.04%
Relative Error ➀ ➁ Percentage Error
27. © Art Traynor 2011
Chemistry
Measurement
Examples: 843.6m or 843.0m or 800.0m
implies a margin of error of 0.05m or ± 0.05m
843.55m ≤ x ≤ 843.65m (nominal 843.6m)
842.95m ≤ x ≤ 843.05m (nominal 843.0m)
800.95m ≤ x ≤ 800.05m (nominal 840.0m)
An interval composed such that its mean is situated
equidistant to a difference and sum of the absolute error
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Error
Etiology of Error
n Margin of Error
28. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
A parameter associated with an observation relating the
probabilistic dispersion of the Magnitude.
As previously noted (essential
elements of a Measurement )
Precision & Accuracy
Instruments of measurement feature a graduated scale
( enumerated intervals incremented in the ordering
metric appropriate to the measurand aspect to be
quantified/recorded, e.g. weight, volume, length ). 1 2 3
mm
Measurement Instrument Precision ( MIP )
A measurement instrument’s precision is given
by the magnitude of its least graduation
Measurement Reading Precision ( MRP )
The measure to be recorded will be 1/10 the MIP
MIP
10
= = 0.001 x 10 – 1
MIP = 0.001M = 1mm
MRP = 0.0.001 M = 0.1mm
➀
0.001M
10
MIP
MRP
n
n
29. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Precision & Accuracy
Certain Digits ( CD )
The digital cardinality ( i.e. count of digits ) of the MIP
constitute that part of the measurement which is certain
| MIP | = n = CD
Uncertain Digits ( UCD )
The additional digit supplied by the MRP is estimated
and thus considered Uncertain
| MRP | = n + 1 = UCD
Significant Figures of Measurement ( SFOM )
The count of all certain digits plus that of the first
uncertain digit
n
n
n
30. © Art Traynor 2011
Chemistry
Precision
The degree of closeness to which a quantitative
measurement approximates the true value of
the quantity measured
Measurement
Precision & Accuracy
The agreement of a particular
experimental result with its true
result
Section 1.4, ( Pg. 11 )
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Precision & Accuracy
Random or Indeterminate Error
Introduces an equal probability of
the measurement registering either
high or low from its true value
Ideal
Accurate & Precise
Neither Accurate
nor Precise
n
o
31. © Art Traynor 2011
Chemistry
Accuracy
The degree to which repeated measurements (under
unchanged conditions) yield the same results
Measurement
Precision & Accuracy
The agreement of a particular
experimental result with its true
result
Section 1.4, ( Pg. 11 )
Measurement
A quantity ascertained by observation characterized by:
Uncertainty
Precision & Accuracy
Systematic or Determinate Error
Introduces a bias error manifesting in a
consistent deviation in experimental
result from the measurand true value
Ideal
Accurate & Precise
Precise but
not Accurate
n
o
32. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Significant Figures of Measurement ( SFOM )
Uncertainty
Significant Figures of Computation ( SFOC )
Exponential ( Scientific ) Notation
Normalized Scientific Notation ( NSN )
Engineering Scientific Notation ( ESN )
n
n
n
n
n
33. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Significant Figures of Measurement ( SFOM )
The count of all certain digits plus that of the first
uncertain digit
Uncertainty
As previously noted (essential
elements of a Measurement )
Significant Figures of Computation ( SFOC )
All non-zero digits are considered significant
Zeros bounded by non-zeros are significant
Leading zeros are not significant
Trailing zeros following a decimal point are significant
Trailing zeros not accompanied by a decimal point are ambiguous
A decimal point may be placed after the number
to ratify the significance of the trailing zeros
Integers or Fractions are considered to be significant
“ Captive ” Zeros
Section 1.4, ( Pg. 13 )
“ Leading ” Zeros do nothing
more than locate a (string of)
sig figs relative to a decimal
numerical representation
n
n
o
o
o
o
o
o
34. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Significant Figures of Measurement ( SFOM )
The count of all certain digits plus that of the first
uncertain digit
Uncertainty
As previously noted (essential
elements of a Measurement )
Significant Figures of Computation ( SFOC )
Exact Numbers are considered significant
Sect 1.5, pg 9
Section 1.4, ( Pg. 13 )
“ Exact ” Numbers are those not
derived from measurement,
akin to discrete enumeration
derived from counting, or Ex
Vi Termini ( EVT – from
definition ), which have an
unlimited number of Sig Figs
Exponential ( Scientific ) Notation
whereby a number with a surfeit of zeros (either large or small in relative magnitude)
or otherwise populated by digits beyond those necessary for the desired precision ( sig figs )
A species of mathematic operation (exponentiation)
is alternatively expressed as the product of a coefficient ( reduced to only its sig figs )
and a multiplier-constant (ten) indexed by an integer.
a x 10b
n
n
n
o
35. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Exponential ( Scientific ) Notation
whereby a number with a surfeit of zeros (either large or small in relative magnitude)
or otherwise populated by digits beyond those necessary for the desired precision ( sig figs )
A species of mathematic operation (exponentiation)
is alternatively expressed as the product of a coefficient ( reduced to only its sig figs )
and a multiplier-constant (ten) indexed by an integer.
a x 10b
Example: “ 350 ”
350 = 3.5.0.0 = 3.5 x 102
Representing integers by
scientific notation (resultant)
entails movement of the
decimal in the “ + “ direction
① ②
Moving 2 positions
in the “+” direction
350 = 35.0.0 = 35.0 x 101
①
Moving 1 position
in the “+” direction
350 = 350.0 = 350.0 x 100
i
Moving 0 positions
in the “+” direction
n
36. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Exponential ( Scientific ) Notation
a x 10b
Normalized Scientific Notation ( NSN )
“ a ” is bounded between one and ten, 1 ≤ |a | < 10 , which allows
for easy comparison of two numbers so expressed as the exponent “ b ”
in this form represents the product’s order of magnitude
The exponent “ b ” is chosen so that the absolute value of the coefficient
For numbers with absolute value between zero and one, 0 < |a | < 1
the exponent b, is expressed as a negative index (e.g. – 5 x 10-1 )
Examples: – 0.5 = – 0.5.0 = – 5.0 x 10-1
➀
Moving 1 position
in the “–” direction
Representing a decimal by
scientific notation (resultant)
entails movement of the
decimal in the “ – “ direction
Wikipedia
n
n
o
37. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
In ESN the exponent “ b ” is restricted to multiples of three
so that the absolute value of the coefficient “ a ” lies between
one and one-thousand, 1 ≤ |a | < 1000 ,
which allows for easy comparison of two numbers so expressed
as the exponent “ b ” corresponds to specific SI prefixes
Example: “ 0.0000000125m ” →
12.5 x 10-9m “ twelve-point-five nanometers ”
1.25 x 10-8m “ one-point-two-five times ten-to-the-negative-eight meters ”
⑨
( 0.0.0.0.0.0.0.0.1.25 ) = 1.25 x 10-8 m
⑧① ②③ ④ ⑤⑥ ⑦
Moving 8 positions
in the “–” direction
( 0.0.0.0.0.0.0.0.1.2.5 ) = 12.5 x 10-9 m
⑧① ②③ ④ ⑤⑥ ⑦
Moving 9 positions
in the “–” direction
Engineering Scientific Notation ( ESN )
o
Wikipedian
o
38. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Significant Figures of Computation ( SFOC )
o Arithmetic: Addition/Subtraction
The number of significant figure decimal places
in the sum or difference of the operation
should equal the smallest number of decimal places
in any of the operands
Multiplicative: Multiplication/Division
The number of significant figures
in the product or quotient of the operation
is the same as the number of significant figures
in the least accurate of the operands
(having the lowest number of significant figures)
n
o
39. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Significant Figures of Computation ( SFOC )
o Arithmetic: Addition/Subtraction
The number of significant figure decimal places
in the sum or difference of the operation
should equal the smallest number of decimal places
in any of the operands
Example:
123
+ 5.35
≠ 128.35
= 128
3 Sig Figs, 0 Decimal Places
3 Sig Figs, 2 Decimal Places
5 Sig Figs > 3 Sig Figs (2 Decimals)
3 Sig Figs, 0 Decimals
n
40. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Significant Figures of Computation ( SFOC )
o Multiplicative: Multiplication/Division
L
WA = l x w
Example:
L : 16.3cm ± 0.1cm { 16.2cm – 16.4cm }
W : 4.5cm ± 0.1cm { 4.4cm – 4.6cm }
A : 16.3cm
x 4.5cm
≠ 73.35cm 2 { 71cm – 75cm }
= 73cm 2
3 Sig Figs
2 Sig Figs
4 Sig Figs > 2 Sig Figs
2 Sig Figs
Serway, pg 15
n
41. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Significant Figures of Computation ( SFOC )
o Rounding: Round to Integer ( RTI )
There is at least one (1) non-direct method to produce round-to-integer results
Round To Nearest – “ q” is the integer that is closest to “ y”
“ y” is the number to be rounded ( y ℝ )
“ q” is the integer result ( q ℤ ) of the rounding operation
n
42. © Art Traynor 2011
Chemistry
Measurement
Precision & Accuracy
Measurement
A quantity ascertained by observation characterized by:
Significant Figures ( Sig Figs )
Uncertainty
Significant Figures of Computation ( SFOC )
o Rounding: Round to Integer ( RTI )
There is at least one (1) non-direct method to produce round-to-integer results
Tie-Breaking Rule
for when “ y” is half-way between two integers, i.e. y = 0.5
Round Half-Up or round half towards +∞
q = : ⌊ y + 0.5⌋ = – ⌈ – y – 0.5⌉
Examples: ⌊ 23.5 + 0.5⌋ = 24 ;
– ⌈ – ( – 23.5 ) – 0.5⌉ = – 23
Rosen, pg 149
n
43. © Art Traynor 2011
Chemistry
Dimensional Analysis
Measurement – Qualities of Unit Quantities Section 1.6, (Pg. 17)
Unit Factors
Insofar as a principal salient of a phenomenological system
is its ability to be characterized by a descriptive quantification,
a well formed quantification ( i .e. measurement )
thereby consists of the following:
An ordered state of matter
possessed of attributes
capable of being
characterized by a
descriptive quantification
Phenomenological System
Unit of Measure ( UOM )
A fundamental or ordering Metric, characteristic
of the measurand and by which class equivalencies
with like phenomena can be gauged, by which a
Relation with the magnitude of an observation
might be expressed as a scaled ratio.
Magnitude
The Modulus or Absolute Value, | m | assigned
to represent an observation and by which a scaled
ratio relation to the UOM can be made .
Examples:
Length: 23.4 km ( Kilometers / Meters )
Mass: 38.5 lbs ( Pounds )
Time: 1:32 hrs ( Hour : Seconds )
Current: 0.32 mA ( Miliamps / Amperes )
| m | UOM
44. © Art Traynor 2011
Chemistry
Dimensional Analysis
Measurement – Qualities of Unit Quantities Section 1.6, (Pg. 17)
Unit Factors
Unit of Measure ( UOM )
Magnitude
Fundamental or Base Unit
A fundamental or ordering Metric, characteristic of
the measurand and by which class equivalencies with
like phenomena can be gauged, by which a Relation
with the magnitude of an observation might be
expressed as a scaled ratio.
A UOM from which every other
measure in its phenomenological class
can be derived.
Akin to a “Basis” or
Linearly Independent Set
Mass
Length
Time
Temperature
Electric Current
Substance Cardinality
Luminous Intensity
kilogram
meter
second
Kelvin
ampere
mole
candela
kg
m
s
K
A
mol
cd
The Seven Fundamental SI Units
Units which can be “ factored ” into other
units, or are compositions of one or more
Base Units are not fundamental, ( e.g. a
volume in Liters can equally stated in terms of length as in be
expressed as 1mL = 1cm3 , or velocity expressed in mi/hr )
n
n
45. © Art Traynor 2011
Chemistry
Dimensional Analysis
Measurement – Qualities of Unit Quantities Section 1.6, (Pg. 17)
Unit Factors
Unit of Measure ( UOM )
Magnitude
Fundamental or Base Unit
Conversion Factor ( CF )
A ratio ( constituted of disparate UOMs evaluating to a unity ) which
has the multiplicative effect of converting one UOM into another without
affecting the magnitude of the quantity as between the operands and (
resultant ) product.
Dimensional Invariance
The operation of a physical law will hold proportionally irrespective of
the UOM employed by which to assay its effects (e.g. two buildings
will measure the same height whether measured in feet or meters).
46. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Dimensional Homogeneity
Only commensurable quantities ( those with identical dimension)
may be ( algebraically ) manipulated to permit:
The veracity of a computational result can frequently be
gauged by the agreement of the solution UOMs with that of
those expected to be ascertained.
Dimensional Fidelity
Comparison
Equality
Addition
Difference
Incommensurable quantities, expressed as a ratio however, may be
employed as Conversion Factors ( CF’s ) indirectly enabling the
relations of commensurable quantities to be evaluated.
Quantities expressed in formulae
are said to be either “ Like
Dimensioned ” (in which case
operations of their Abelian Group
can be directly performed) or
“ Unlike Dimensioned ” in which
case those operations might still
be performed if those UD
quantities are expressed as ratios
47. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
UOMi
1
①
UOMf
1
O’Leary, Section 8.3,
Pg. 346
Akin to a Cantor Set
forming iteration
UOMi
1
→
State both the Initial and Final terms as rationals of unity
indicating the progression of the calculation LHS → RHS
An equation is constructed, with the Initial term occupying the LHS and the
Final term occupying the RHS of the equality. At least one CF will be
necessary ( presuming a CF rational featuring UOMf units as dividend and
with UOMi units for a divisor can be furnished for the calculation) in which
case the Final term is replicated on the RHS as the singular CF = CFf
UOMf
1
UOMf
1
=
Initial FinalFinal
Initial CFf Final
UOMi
2a
48. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
UOMi
1
Should a single CF rational with UOMf numerator and UOMi unit denominator
not be available for the calculation, at least one additional CF will need to be
introduced.
UOMf
1
UOMf
1
=
Initial CFf Final
CF’s necessary to
complete the
transformation will
be inserted here
UOMi
1
Lacking for a singular CF, the additional CF , denoted CFk , will be interpolated,
and juxtpositioned between the Initial term and CFf
UOMi
1
UOMf
1
UOMf
1
Initial CFk CFf Final
③
=
2b
49. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
The interpolated CF rational must then be constructed such that the
denominator of the CFk term bears UOMi units
UOMi
1 1
UOMf
1
UOMf
1
Initial CFk CFf Final
=
UOMi
④
The CF selected must further satisfy the condition that the UOM of
the CFk term numerator will transitively match a corresponding CFf
denominator to complete the desired quantity unit conversion
UOMi
1 1
UOMf
1
UOMf
1
=
UOMi
⑤
50. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
UOMi
1
If a CF term satisfying the condition that the UOM of the CFk term numerator
transitively match a corresponding CFf denominator term cannot quite be
had, yet another CF term will need to be introduced.
UOMf
1
UOMf
1
=
Initial CFf Final
UOMi
1
This added term – as was desired of the two-term CF case – denoted CFn,
will best be selected to form a transitive chain with the Initial and Final terms
via apposite alteration of UOMs in the numerators and denominators of the
chained terms respectively.
1 1
UOMf
1
UOMf
1
Initial CFk CFn CFf Final
=
⑥
1
CFk
UOMi
Additional
CF’s
will be
inserted here
UOMi
⑦
51. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
Thus constructed, the transitive chain of CF terms can be extended by further
interpolation of CFs to the chain ( denoted by either a CFk + 1 or CFn – 1
incrementation ) to accommodate any length of expression needed to effect
the desired UOM transformation
Initial CFk CFn CFf Final
UOMi
1 1 1
UOMf
1
UOMf
1UOMi
=
⑧
52. © Art Traynor 2011
Chemistry
Dimensional Analysis
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
AT Problem Solving Technique ( ATPST )
Example:
UOMi
1
①
UOMf
1
→
We begin by populating the UFM-PST table
with the data provided
Initial Final
A student has entered a 10.0-km run. How long is the run in miles?
Section 1.6 (Pg. 19)
10.0-km
1
Lf -mi
1
→
Problem Data & Formulae
Li = 10.0-km
UOMf = mi
CFf =
mi
km
53. © Art Traynor 2011
Chemistry
Dimensional Analysis
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
AT Problem Solving Technique ( ATPST )
Example:
An equation is constructed, with the Initial term occupying the
LHS and the Final term occupying the RHS of the equality.
At least one CF will be necessary ( presuming a CF rational
featuring UOMf units as dividend and with UOMi units for a
divisor can be furnished for the calculation) in which case the
Final term is replicated on the RHS as the singular CF = CFf
A student has entered a 10.0-km run. How long is the run in miles?
Section 1.6 (Pg. 19)
Problem Data & Formulae
Li = 10.0-km
UOMf = mi
CFf = ?
mi
km
UOMi
1
UOMf
1
UOMf
1
=
Initial CFf Final
UOMi
10.0-km
1
Lf -mi
1
Lf -mi
1
=
UOMi
CF1 =
1km
1000m
CF1 is not composed of the
necessary UOMs so additional
CF rationals will need to be
introduced, but it does suggest
the reciprocal will advance us
toward the solution.
mi
km
≠
≠
②
54. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
UOMi
1
Lacking for a singular CF, the additional CF , denoted CFk , will be
interpolated, and juxtpositioned between the Initial term and CFf
UOMi
1
UOMf
1
UOMf
1
Initial CFk CFf Final
③
=
Example:
A student has entered a 10.0-km run. How long is the run in miles?
Section 1.6 (Pg. 19)
Problem Data & Formulae
Li = 10.0-km
UOMf = mi
CFf = ?
mi
km
CF1 =
1km
1,000m mi
km
10.0-km
1
UOMi
1
Lf -mi
1
Lf -mi
1
=
UOMi -km
CF2 =
1m
1.094yd
mi
km
≠
=
≠
≠
CF2 fails to supply a CF
matching the UOMs needed.
This suggests an additional
term will need to be interpolated
to form a transitive chain (of the
form yards-per-mile).
10.0-km
1
UOMi
1
Lf -mi
1
Lf -mi
1
=
10.0-km
1,000-m
55. © Art Traynor 2011
Chemistry
Dimensional Analysis
Unit Factors
Method of Dimensional Analysis ( MODA )
Factor Label Method ( FLM ) or Unit Factor Method ( UFM )
Problem Solving Technique
This added term – as was desired of the two-term CF case –
denoted CFn, will best be selected to form a transitive chain with
the Initial and Final terms via apposite alteration of UOMs in the
numerators and denominators of the chained terms respectively.
Example:
A student has entered a 10.0-km run. How long is the run in miles?
Problem Data & Formulae
Li = 10.0-km
UOMf = mi
CFf = ?
mi
km
CF1 =
1km
1,000m mi
km
CF2 =
1m
1.094yd
mi
km
≠
=
≠
≠
CF3 supplies the missing UOM (in
reciprocal) equivalency we need to
complete the transitive chain,
which also matches up with the
UOM terms in the CFn term
10.0-km
1 1 1
Lf -mi
1
Lf -mi
1
Initial CFk CFn CFf Final
=
10.0-km
1,000-m
CF3 =
1,760 yd
1mi
mi
km
≠
≠
10.0-km
1 1 1
1-mi
1
Lf -mi
1
=
10.0-km
1,000-m
④
1-m
1.094-yd
1,760-yd
56. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Mathematical Implications
Mathematical Implications of DA
Dimension
The indexed product of a fundamental physical Metric
( e.g. Length, Mass, Time, Charge, Temperature )
by a rational argument is said to constitute the Dimension of that
phenomenological equivalence class ( i.e. physical quantity )
Scale Unit
The notion of Dimension is hierarchically distinct
from the notion of Scale Unit
Mass
Scale Unit
Pound ( lb )Gram ( g )
Dimension
( of Mass )
Scale Units
( in the Mass Dimension )
Any Legth has a Dimension of “L”
irrespective of what units of
“Length” are selected by which to
make a measure ( see
Dimensional Invariance )
57. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Mathematical Implications
Mathematical Implications of DA
Dimension
The fundamental physical Metrics constituting physical dimensions are
further characterized by the following properties:
Abelian Group over ℚ
Vector Space over ℚ
The fundamental physical Metrics are understood to
constitute a Basis for this Vector Space
n
A Change of Basis is similarly effected, as in any other
vector space, yielding alternate systems of units ( e.g.
whether the unit for Charge is derived from the unit for
Current or vice versa )
n
58. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Constitute Linear Combinations of the
Dimensional ( Fundamental ) Scale Units
The validity of the Factor Label Method ( FLM ) or Unit Factor Method
( UFM ) is limited to well-formed Conversion Factors ( CF’s ) which:
Feature homogenous solution sets ( i.e. with intercepts
intersecting the vector space at its coordinate origin ).
and
Temperature CFs ( in particular ) are problematic because:
Celsius ↔ FahrenheitCelsius ↔ Kelvin
Constant Difference∆
∝ In-Constant Ratio
In-Constant Difference∆
∝ In-Constant Ratio
Temperature can summarily be
defined as a measure of the
internal energy present in an
phenomenological system
59. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
The validity of the Factor Label Method ( FLM ) or Unit Factor Method
( UFM ) is limited to well-formed Conversion Factors ( CF’s ) which:
Temperature CFs ( in particular ) are problematic because:
Celsius ↔ FahrenheitCelsius ↔ Kelvin
Constant Difference∆
∝ In-Constant Ratio
In-Constant Difference∆
∝ In-Constant Ratio
Conversion Factors ( CF’s )
100 °C
212 °F – 32 °F
°F =
A ratio of the scale
interval over which
water H2O maintains
a liquid state
100 °C
180 °F
°F = = °C + 32
5
9
°C = °K – 273.15
Conversion Factors ( CF’s )
– 40° C = – 40° F
60. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
The validity of the Factor Label Method ( FLM ) or Unit Factor Method
( UFM ) is limited to well-formed Conversion Factors ( CF’s ) which:
Temperature CFs ( in particular ) are problematic because:
Celsius ↔ FahrenheitCelsius ↔ Kelvin
Constant Difference∆
∝ In-Constant Ratio
In-Constant Difference∆
∝ In-Constant Ratio
Conversion Factors ( CF’s )
100 °C
212 °F – 32 °F
°C =
A ratio of the scale
interval over which
water H2O maintains
a liquid state
100 °C
180 °F
°C = = ( °F – 32 )
9
5
°K = °C + 273.15
Conversion Factors ( CF’s )
– 40° C = – 40° F
61. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Fahrenheit °F
The “scaling” of the various standards of Temperature measure accounts for
the CF difficulty in applying FLM / UFM
After Daniel Gabriel Fahrenheit ( 1686 – 1736 ) ,
German Physicist
Tripartite Reference Interval ( defining unit scale )
Ambient Body Temperature ranks 96°Fn
Frigorific Equilibrium of water-ice H2O
slurry ranks 32°F
n
Frigorific Equilibrium of Ammonium Chloride
NH4CL ranks 0°F
n Danzig
Polish-Lithuanian
Commonwealth
62. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Celsius °C
The “scaling” of the various standards of Temperature measure
accounts for the CF difficulty in applying FLM / UFM
After Anders Celsius ( 1701 – 1744 ) ,
Swedish Astronomer
Original Bipartite Reference Interval ( defining unit scale )
Boiling point of water H2O ranks 100°Cn
Freezing point of water H2O ranks 0°Cn
Grand Principality of
Transylvania
Contemporary Bipartite Reference Interval
Triple Point of water H2O ranks 100°Cn
Absolute zero 0°K = – 273.15°Cn
o The Triple Point of a substance is that unique
temperature and pressure at which the three phases
coexist in thermodynamic equilibrium
63. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Celsius °C
The “scaling” of the various standards of Temperature measure
accounts for the CF difficulty in applying FLM / UFM
After Anders Celsius ( 1701 – 1744 ) ,
Swedish Astronomer
Grand Principality of
Transylvania
The Celsius and Kelvin temperature scales share
the same interval (defined by the partition of
the interval between absolute zero 0°K and the
Triple Point of water H2O at 273.15 °K ,
or one part in 273.15, or 0.003661).
n
Contemporary Bipartite Reference Interval
| Ttp – T0 |
1
= I
64. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Celsius °C
The “scaling” of the various standards of Temperature measure
accounts for the CF difficulty in applying FLM / UFM
After Anders Celsius ( 1701 – 1744 ) ,
Swedish Astronomer
Grand Principality of
Transylvania
Contemporary Bipartite Reference Interval
Interval System of Measure
The Celsius system employs a relative scale of
measure, as opposed to the absolute scales on
which other fundamental scale units are
premised
200-ft is twice as long as 100-ft, but 20°C water does
not is not twice as thermally energetic as 10°C water
n
65. © Art Traynor 2011
Chemistry
Dimensional Analysis
Wiki “Dimensional Analysis”
Limitations
Linear Homogeneity Restriction
Kelvin °K
The “scaling” of the various standards of Temperature measure
accounts for the CF difficulty in applying FLM / UFM
After William Thompson, Lord (Baron) Kelvin
( 1824 – 1907 ) , British Mathematical Physicist
Grand Principality of
Transylvania
United Kingdom
of Great Britain
& Ireland
Interval System of Measure
A unit of measure for Temperature premised on a
Thermodynamic, or absolute, scale and one of the
seven base units of the SI system
Coincident with the Celsius scale intervaln
o Preserving simplicity by a “ constant difference ”
in conversion between the two systems
66. © Art Traynor 2011
Chemistry
Properties of Substances
Density
Density : ⍴ (rho)
A fundamental property of any substances is its density.
Density is the mass per unit volume of any substance
There are several related notions
⍴ = →
m
V
mass
volume
Units are Kilograms per Cubic Meter
kg
m3
67. © Art Traynor 2011
Chemistry
Properties of Substances
Density
Density : ⍴ (rho)
A fundamental property of any substances is its density.
Density is the mass per unit volume of any substance
Specific or Unit Weight
The weight per unit volume of a material
⍴ = → in
m
V
mass
volumeThere are several related notions
Units are Newtons per Cubic Meter
N
m3
kg
m3
m
s2
G in
γ = ⍴ · G → · G → →
m
V
m · G
V
w
V
Can be affected by variations in Temperature and
Pressure ( via the Bulk Modulus of the material )
68. © Art Traynor 2011
Chemistry
Properties of Substances
Density
Density : ⍴ (rho)
A fundamental property of any substances is its density.
Density is the mass per unit volume of any substance
Specific or Unit Weight
The weight per unit volume of a material
⍴ = → in
m
V
mass
volumeThere are several related notions
kg
m3
γ = → in
w
V
weight
volume
N
m3
Relative Density
Density expressed as a ratio of one material to a
referent material
RD =
⍴s
⍴r
69. © Art Traynor 2011
Chemistry
Properties of Substances
Density
Density : ⍴ (rho)
A fundamental property of any substances is its density.
Density is the mass per unit volume of any substance
Specific or Unit Weight
The weight per unit volume of a material
⍴ = → in
m
V
mass
volumeThere are several related notions
kg
m3
γ = → in
w
V
weight
volume
N
m3
Relative Density
Density expressed as a ratio of one material to a
referent material
Specific Gravity
Relative Density expressed as a ratio of a material to
water ( the referent material )
RD =
⍴s
⍴r
70. © Art Traynor 2011
Chemistry
Properties of Substances
Density
Density : ⍴ (rho)
A fundamental property of any substances is its density.
Density is the mass per unit volume of any substance
Specific or Unit Weight
The weight per unit volume of a material
⍴ = → in
m
V
mass
volumeThere are several related notions
kg
m3
γ = → in
w
V
weight
volume
N
m3
RD =
⍴s
⍴r
Densities do not necessarily correlate to atomic masses
Atomic spacings and crystalline structure
affect elemental density
Avagadro’s Number
Density derived Metrics such as Molar
Volume encode Avagadro’s Number and
their dependence on atomic masses
n
71. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Matter”
Structure of Matter
Definition of Matter
Matter comprises the fundamental ontological constituents of which a
phenomenological system is composed, classically reckoned as that which
occupies space ( i.e. characterized by volume ) and expresses a mass
Properly excludes massless Particles ( e.g. Photon )
Not to be conflated with Mass
“Particles” do not
necessarily
constitute Matter!
Matter can assume a continuum of states of aggregation, determined by
intrinsic quantifiable parameters such as pressure, temperature, and
volume.
States / Phases of Matter
Solid
Liquid
Gas
Exotics ( Plasma, Super-states, Condensates )
72. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Substance”
Structure of Matter
Matter can assume a continuum of states of aggregation, determined by
intrinsic quantifiable parameters such as pressure, temperature, and
volume.
States / Phases of Matter
Solid : Characterized by Rigidity
Liquid : Characterized by Volumetric Fluidity
Gas : Characterized by Volumetric and Morphological Fluidity
Fixed Volume
Fixed Shape ( Morphology )
Fixed Volume
Indeterminate Shape ( Morphology )
Indeterminate Volume
Indeterminate Shape ( Morphology )
73. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Matter”
Structure of Matter
Definition of Matter
Matter comprises the fundamental ontological constituents of which a
phenomenological system is composed, classically reckoned as that which
occupies space ( i.e. characterized by volume ) and expresses a mass
Properly excludes massless Particles ( e.g. Photon )
Not to be conflated with Mass
“Particles” do not
necessarily
constitute Matter!
Definition of Substance
A configuration of Matter featuring:
Constant Chemical Composition
Characteristic Properties
cannot be separated into more
elemental components without
breaking chemical bonds
Not to be conflated
with a mixture
From which Law of
Constant Composition
arises
Suited to both an Element
and a Homogenous
“Compound”
74. © Art Traynor 2011
Chemistry
Principles
Structure of Matter
A species of Substance :
Composed of two or more Elements, characterized by :
Definition of Compound
Constant Composition / Unique Chemical Structure
Resolution into Elements via Chemical Processes
Substance criteria
Bonded ( Structure )
Fixed ratio of Atoms ( e.g. Formula )n
Representation by a Formula
75. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Definition of Mixture
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of which
obey a periodic verisimilitude ordering distinctive phenomenological classes
among its representatives.
A mixture is a combination of unbound substances, capable of
separation, the constituents of which remain chemically distinct and
irreducible.
Wiki: “ Mixture”
76. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of
which obey a periodic verisimilitude ordering distinctive
phenomenological classes among its representatives.
Properties of Elements
Atomic Number
Proton Cardinalityn
An element’s Atomic Number provides the cardinality
of the protons in its nucleus
Singularity of Atomic Numbern
Each element has a unique and singular atomic number
exclusively distinguishing it from any other element
Aluminium
2
13
26.9815386
GIIIA (13): Post-Transition Metal
P3
8
[Ne]3s23p1
3
Metal
Al
77. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of
which obey a periodic verisimilitude ordering distinctive
phenomenological classes among its representatives.
Properties of Elements
Aluminium
2
13
26.9815386
GIIIA (13): Post-Transition Metal
P3
8
[Ne]3s23p1
3
Metal
Al Periodicity
Chemical properties of the Elements were observed
to recur with regular recurrence, the patterns of
which are encoded in the Periodic Table
Al13
78. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of
which obey a periodic verisimilitude ordering distinctive
phenomenological classes among its representatives.
Properties of Elements
Aluminium
2
13
26.9815386
GIIIA (13): Post-Transition Metal
P3
8
[Ne]3s23p1
3
Metal
Al Periodicity
The table is superficially organized into
Groups ( columns ) and Periods ( rows )
Al13
13
3
79. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of
which obey a periodic verisimilitude ordering distinctive
phenomenological classes among its representatives.
Properties of Elements
Aluminium
2
13
26.9815386
GIIIA (13): Post-Transition Metal
P3
8
[Ne]3s23p1
3
Metal
Al Periodicity
Groups collect Elements with similar chemical
properties and exhibit trends in Atomic Radius
( Positively ) and in Electron Affinity and
Ionization Energy ( Inversely )
n
①
②
③
3d
④⑤ ⑥ ⑦⑧⑨ 10 11 12
13 14 16 17
18
15
Al13
80. © Art Traynor 2011
Chemistry
Principles
Wiki: “ Chemical Element”
Structure of Matter
Definition of Element
Elements are those fundamental constituents of matter uniquely
characterized by an atomic number, the chemical properties of
which obey a periodic verisimilitude ordering distinctive
phenomenological classes among its representatives.
Properties of Elements
Aluminium
2
13
26.9815386
GIIIA (13): Post-Transition Metal
P3
8
[Ne]3s23p1
3
Metal
Al Periodicity
Periods collect Elements with similar electronic
structure ( incremented by Atomic Number ) and
exhibit trends in Electron Affinity and Ionization
Energy ( positively ), and Atomic Radius ( Inversely )
n
1
2
4
5
6
7
Al133
81. © Art Traynor 2011
Chemistry
Principles
Structure of Matter
Definition of Mixture
A mixture is a combination of unbound substances, capable of
separation, the constituents of which remain chemically distinct and
irreducible.
Wiki: “ Mixture”
Properties of Mixtures
HeterogenousHomogenous
Visually Indistinguishable
Constituents
N N
Absence of Chemical Bonds
The main distinguishing characteristic of a mixture is the homogeneity of its composition
Visually Distinct Constituents
o Solution
( Gaseous /Aqueous /Concretion )
Variable Composition
82. © Art Traynor 2011
Chemistry
Principles
Fundamental Laws of Chemistry
Law of Conservation of Mass ( LOCOM )
For a phenomenological system,
closed to extraneous transfers of Matter & Energy,
the Mass of the system must remain constant over time,
implying:
Mass can neither be created or destroyed
Section 2.1 (Pg. 44)
Mass can only be spatially rearranged
Law of Definite Proportion ( LODP )
A chemical compound always contains
exactly the same proportion of Elements by Mass
Section 2.1 (Pg. 44)
83. © Art Traynor 2011
Chemistry
Principles
Fundamental Laws of Chemistry
Conservation of Mass
For a phenomenological system,
closed to extraneous transfers of Matter & Energy,
the Mass of the system must remain constant over time,
implying:
Mass can neither be created or destroyed
Section 2.1 (Pg. 44)
Mass can only be spatially rearranged
Definite Proportion
A chemical compound always contains
exactly the same proportion of Elements by Mass
Section 2.1 (Pg. 44)
Multiple Proportions
For two Elements
capable of forming at least two distinct Compounds
the ratios of the Masses of the second element combining with one gram of the first
will always reduce to a small whole number
Section 2.1 (Pg. 44)
84. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Dalton’s “ New System of Chemical Philosophy ”
Each element is composed of miniscule particles or Atoms
Section 2.3 (Pg. 46)
The Atoms of a Element are identical
United KingdomCounty of Cumbria
1766 – 1844
JohnDalton
Compounds are composed of disparate Elements and always
expresses the same relatives numbers and species of Atoms
Reactions entail the reorganization of Atoms ( in the
manner in which they are bound ) and remain
unaffected fundamentally by a reaction
85. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Avogadro’s Hypothesis
At constant/equal Temperature and Pressure:
Section 2.3 (Pg. 46)
Kingdom of SardiniaCity of Turin
1776 – 1856
AmadeoAvogadro
Refining the volumetric gas reactant results of Joseph Gay-Lussac ,
Avogadro postulated that gasses combine – irrespective of molecular
size – in equal volumes and thus equal particulate cardinality
Equal volumes of disparate ( reactant ) gasses contain the
same number of particles
Implying the sizes of the particles are negligible compared to
the distances separating constituent particles
86. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Thomson’s Cathode Ray Particles ( Electrons )
The particles exhibit directionality – they emanate at the negative
electrode ( i.e. Cathode ) and terminate at the positive ( i.e. Anode )
Section 2.3 (Pg. 50)
United KingdomCounty of Lincolnshire
1856 – 1940
Joseph John ( J.J. )
Thomson
High voltage applied to an evacuated tube produces a stream of particles
between electrodes observed to possess the following properties:
The particle stream ( i.e. Cathode Rays ) are repelled by the
negative pole of an applied electric field.
First observed in 1869 by German
physicist Johann Hittorf, named in
1876 by Eugen Goldstein
Kathodenstrahlen ( i.e. “ Cathode ” )
The Charge-to-Mass ratio of the constituent particles in the
stream can be determined by a measurement of deflection of the
stream subject to a magnetic field :
charge
mass = = 1.76 x 108 C/g
e
m
Electrodes of various metallic composition were each observed to
produce Cathode Rays , implying that all atoms must contain
the negative charge particles thus designated “ Electrons ”
87. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Thomson’s Plum Pudding Atomic Model
As the Atom exhibited electrical ( charge ) neutrality ,
the Atom must be composed of a particle with
opposite and equal charge as the Electron
Section 2.3 (Pg. 50)
United KingdomCounty of Lincolnshire
1856 – 1940
Joseph John ( J.J. )
Thomson
Thomson’s description of the Electron lead to a compositional
conjecture concerning the structure of the Atom
Thomson further speculated that this positive charge
would be randomly distributed throughout the Atom
with the negatively charged Electrons embedded within
the atomic composition ( i.e. a ‘Plum Pudding’ of
heterogenous particles )
The Plum Pudding analogy was first
proposed by Lord Baron Kelvin,
William Thomson ( unrelated )
88. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Millikan’s Derivation of the Electron Mass
The Oil Drop experiment allowed Millikan to arrive at a
precise value for the magnitude of the Electron Unit Charge
Section 2.3 (Pg. 51)
United StatesState of Illinois
1868 – 1953
Robert Andrews Millikan
Millikan contrived an experiment whereby the free-fall of charged
oil droplets within an atmospherically controlled chamber could be
halted ( equalized to the force of G ) by the application of a
voltage across two oppositely charged plates
The Plum Pudding analogy was first
proposed by Lord Baron Kelvin,
William Thomson ( unrelated )
Applying this Electron Unit Charge magnitude to J.J. Thomson’s
Charge-to-Mass Ratio allowed Millikan to posit a precise Mass
for the Electron at 9.11 x 10 – 31 kg
89. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Becquerel’s Discovery of Radiation
Becquerel’s results subsequently led to the tri-partite radiation
emission classifications :
Section 2.3 (Pg. 52)
Republic of France
Historical Province
of Brittany
1852 – 1908
Antoine Henri Becquerel
Becquerel serendipitously noted that a concealed Uranium mineral
was able to impart an image to an unexposed photographic plate
Region of Pays de la Loire
Department of Loire-Atlantique
αParticle
Carries a 2+ Charge ( opposite & twice the Electron )n
7300 times the Mass of the Electron !n
β Particle
A high-speed Electronn
γ Ray
A form of high-energy “ light ”n
90. © Art Traynor 2011
Chemistry
Atomic Theory
Historical Development
Rutherford’s Characterization of the Nucleus
A source utilizing Becquerel’s alpha particles
Section 2.3 (Pg. 52)
1871 – 1937
Earnest Rutherford
Rutherford devised an experiment to test Thomson’s Plum Pudding
model of the Atom whereby :
αParticle
Carries a 2+ Charge ( opposite & twice the Electron )n
7300 times the Mass of the Electron !n
Realm of New Zealand
Is trained on a gold foil target to probe the structure of the atom by
dispersion of the source in collision/deflection with the target
Occasional , significant deflection of the source – through large
angles – led to the conclusion that the Atom must be composed of
a concentrated , positive-charge massive “ nucleus ”
Minimal deflection of the majority of the source moreover led to the added
conjecture that the atom must be composed mostly of “ open space ”
91. © Art Traynor 2011
Chemistry
Atomic Structure
Modern Understanding
The Modern View of Atomic Structure
Nuclear Diameter : ~ 10 – 13 cm
Section 2.5 (Pg. 53)
Atomic Diameter : ~ 10 – 8 cm The margin at which the electrons
are observed to circulate the nucleus
Atomic Nucleus
Protons
Positive Chargen
Charge Equal in Magnitude to the Electron’s Negative Chargen
Neutrons
No Charge ( Neutral )n
Same ( essentially ) Mass as Protonn
Nuclear Size is minimal compared to Atomic Radii
Nuclear Density accounts for most all the Mass of the Atom
The Electrons constitute the vast
majority of the Atomic Volume
92. © Art Traynor 2011
Chemistry
Atomic Structure
Atomic Particles
Atomic Particles Section 2.5 (Pg. 53)
Electron 9.11 x 10 – 31 kg 1–
The Atomic Constituent Particles
Proton 1.67 x 10 – 27 kg 1+
Neutron 1.67 x 10 – 27 kg 0
( 1.60 x 10 – 19 C )
( 1.60 x 10 – 19 C )
Particle Mass Charge
93. © Art Traynor 2011
Chemistry
Atomic Structure
Atomic Variants
Isotope Section 2.5 (Pg. 54)
A Chemical Element variant in which the cardinality of its
Neutron constituents differs from its Atomic Number
( e. g. its Proton content which crucially imparts its unique
identity as a distinct Element).
All isotopes of an Element bear the same Atomic Number
All isotopes of an Element share the same number of Protons
All isotopes of an Element share the same number of Protons
but a differing number of Neutrons
From the Greek “ isos ” meaning
“ equal ” and “ topos ” meaning
“ place ” evoking the notion that
Isotopes occupy the “ same place”
in the Periodic Table hierarchy
A Nucleon is a constituent of the atomic nucleus of
which there are two classes: Protons and Neutrons
An atom’s Mass Number reflects the cardinality of its
Nucleon constituents
Wiki: “ Isotope”
94. © Art Traynor 2011
Chemistry
Atomic Structure
Atomic Variants
Isotope Section 2.5 (Pg. 54)
A Chemical Element variant in which the cardinality of its
Neutron constituents differs from its Atomic Number
All isotopes of an Element bear the same Atomic Number
From the Greek “ isos ” meaning
“ equal ” and “ topos ” meaning
“ place ” evoking the notion that
Isotopes occupy the “ same place”
in the Periodic Table hierarchy
An atom’s Mass Number ( integer value ) reflects the
cardinality of its Nucleon constituents
Al13
27
Al13
27
Mass
Number
Atomic
Number
→
The difference of the Mass Number and the Atomic
Number yields the cardinality of the Neutron constituent
of the Atom ( or Isotopic form of the Atom ) or the
Neutron Number
Wiki: “ Isotope”
95. © Art Traynor 2011
Chemistry
Atomic Combinations
Molecules
Molecules Section 2.5 (Pg. 55)
An Electrically Neutral composition of two or more Atoms
adhered by a Chemical Bond
Wiki: “ Molecule”
Distinguished from Ions by the absence of net Charge
By convention Polyatomic Ions are sometimes nevertheless
referred to as Molecules
Upper Level Equivalence Classifications
Homonuclear
A molecule composed of two or more Atoms of the same Element
( e.g. a Diatomic Molecule )
Heteronuclear
A molecule composed of two or more Atoms of the different Elements
96. © Art Traynor 2011
Chemistry
Molecular Representation Section 2.5 (Pg. 55)
Wiki: “ Chemical Formula”
Chemical Formula
There are several conventional models and representations by which
the Elemental constituents of a Molecule can be represented :
Atomic Combinations
Molecules
Structural Formula
Space-Filling Models
Ball & Stick Models
97. © Art Traynor 2011
Chemistry
Molecular Representation Section 2.5 (Pg. 55)
Wiki: “ Chemical Formula”
Chemical Formula
There are several conventional models and representations by which
the Elemental constituents of a Molecule can be represented :
Atomic Combinations
Molecules
A symbolic Molecular representation composed of the following:
Alpha characters indicating the constituent
Elements of the Molecule ( 1 – 3 characters )
Numeric subscripts indicate the cardinality of individual Elements in the
molecular composition ( after the fashion of a multiplicative factor )
Polyatomic Ionic constituents are further demarcated by parenthetical inclusion, with
like subscription indicating multiplicity ( as with Elemental molecular constituents )
n
Ionic constituents may be further denoted by an explicit ( superscripted )
Charge designation ( + / – ) , where a multiplicity of unit charge is
indicated by an integer coefficient ( if greater than unity )
98. © Art Traynor 2011
Chemistry
Molecular Representation Section 2.5 (Pg. 55)
Wiki: “ Chemical Formula”
Chemical Formula
There are several conventional models and representations by which
the Elemental constituents of a Molecule can be represented :
Atomic Combinations
Molecules
Structural Formula
A Molecular representation composed of the symbolism of the
Chemical Formula ( and multiplicity conventions ) supplemented
by Bond and Spatial Orientation representations :
Dashed lines portray 3D receding molecular constituents
whereas graduated wedges portray 3D orientation
projecting from a 2D representational surface
99. © Art Traynor 2011
Chemistry
Molecules
Bonding
Chemical Bond Section 2.5 (Pg. 55)
An attraction between Atoms enabling the formation of
Chemical Substances composed of two or more Atoms
The electrons and nuclei constituting the Substance formed
The Bond originates in an electrostatic force of attraction resulting from:
A Dipole moment among the Bonded Substance
Wiki: “ Chemical Bond”
Strong Bonds
Bonds can also be characterized by their relative strength
Covalent Bondingn
Ionic Bondingn
Weak Bonds
Dipole-Dipole Interactionsn
London Dispersion Forcen
Hydrogen Bondingn
100. © Art Traynor 2011
Chemistry
Molecules
Bonding
Chemical Bonds - Classified Section 2.5 (Pg. 55)
Wiki: “ Covalent Bond”
Strong Bonds
Bonds can also be characterized by their relative strength
Covalent Bondingn
A Bond fashioned by the sharing of Electrons as
between the constituent Elements of a Molecule
o Only a distinct number of Electrons participate in
the Bond and are thus designated the Shared Pairs
or Bonding Pairs
o Covalent Bonding can proceed to the extent that each
Atom in the Molecular composition is considered to
have thus attained a full outer shell corresponding to
a stable ( energetically minimized ) Electronic
configuration
101. © Art Traynor 2011
Chemistry
Chemical Bonds - Classified Section 2.5 (Pg. 55)
Wiki: “ Covalent Bond”
Strong Bonds
Bonds can also be characterized by their relative strength
Ionic Bonding ( Electrovalence )n
A Bond fashioned by the electrostatic attraction as between oppositely
charged Ions among the constituent Elements of a Molecule
o An Ion with electron cardinality less than its Elemental Atomic
Number – a Cation – will exhibit a positive charge, the magnitude
of which is an integer multiple of product of the unit Charge and the
difference between its Atomic Number and electron cardinality
Atomic Combinations
Molecules
o An Ion with electron cardinality greater than its Elemental Atomic
Number – a Anion – will exhibit a negative charge, the magnitude
of which is an integer multiple of product of the unit Charge and the
difference between its electron cardinality and Atomic Number
102. © Art Traynor 2011
Chemistry
Chemical Bonds - Classified Section 2.5 (Pg. 55)
Wiki: “ Covalent Bond”
Strong Bonds
Bonds can also be characterized by their relative strength
Ionic Bonding ( Electrovalence )n
A Bond fashioned by the electrostatic attraction as between oppositely
charged Ions among the constituent Elements of a Molecule
Typical interaction/product species include:
o A metal and non-metal wherein a net transfer of electrons proceeds to
such extent that both atoms achieve a state wherein their valence shells
can be considered to be simultaneously filled
Atomic Combinations
Molecules
o Reactants wherein a relatively large difference in Electronegativity
exists between constituent species
o Salts
103. © Art Traynor 2011
Chemistry
Chemical Bonds - Classified Section 2.5 (Pg. 55)
Wiki: “ Covalent Bond”
Strong Bonds
Bonds can also be characterized by their relative strength
Ionic Bonding ( Electrovalence )n
A Bond fashioned by the electrostatic attraction as between oppositely
charged Ions among the constituent Elements of a Molecule
Typical interaction species can be characterized by:
o Electrical conductivity ( in solution or molten state )
Atomic Combinations
Molecules
o Aqueous solubility
104. © Art Traynor 2011
Chemistry
Structure of Matter
Wiki: “ Chemical Element”
Periodicity
Definition of Element
Elements are those fundamental constituents of matter uniquely characterized by an
atomic number, the chemical properties of which obey a periodic verisimilitude
ordering distinctive phenomenological classes among its representatives.
Properties of Elements
Periodicity: Chemical properties of the Elements were observed to recur with
regular recurrence, the patterns of which are encoded in the Periodic Table
Upper Level Equivalence Classifications
Metals
Non-Metals
105. © Art Traynor 2011
Chemistry
Structure of Matter
Wiki: “ Chemical Element”
Periodicity
Properties of Metals
Ready Conductors of Heat & Electricity
Malleable ( can be formed into thin sheets )
Ductile ( can be formed into wires )
Lustrous appearance
Electron Donors forming Positive Ions ( typically )
Properties of Non-Metals
Tend to form Diatomic ( Homonucelar ) Molecules
Reactions with metals tend to produce Ionic Salts
Electron Receptors forming Negative Ions ( typically )
106. © Art Traynor 2011
Chemistry
Structure of Matter
Periodicity
Families of Elements ( Groups )
The Elements may be further classified according to representative “ families ”
exhibiting similar chemical properties. These class-similar elements are arrayed
within the Periodic Table vertically into Groups
Second Level Equivalence Classifications
Alkali Metals ( Group 1 )
Section 2.7 (Pg. 60)
Hydrogen, Lithium, Sodium, Potassium, Cesium, Francium
Tend to form 1+ Ions when reacted with non-metalsn
①
H1
Na11
Li3
K19
Rb37
Cs55
Fr87
107. © Art Traynor 2011
Chemistry
Structure of Matter
Periodicity
Families of Elements ( Groups )
The Elements may be further classified according to representative “ families ”
exhibiting similar chemical properties. These class-similar elements are arrayed
within the Periodic Table vertically into Groups
Second Level Equivalence Classifications
Alkaline Earth Metals ( Group 2 )
Section 2.7 (Pg. 60)
Beryllium, Magnesium, Calcium, Strontium, Barium, Radium
Tend to form 2+ Ions when reacted with non-metalsn
Mg12
Be4
Ca20
Sr38
Ba56
Ra88
②
108. © Art Traynor 2011
Chemistry
Structure of Matter
Periodicity
Families of Elements ( Groups )
The Elements may be further classified according to representative “ families ”
exhibiting similar chemical properties. These class-similar elements are arrayed
within the Periodic Table vertically into Groups
Section 2.7 (Pg. 60)
Second Level Equivalence Classifications
Halogens ( Group 17 )
Fluorine, Chlorine, Bromine, Iodine, Astatine, Ununseptium
Tend to form 1 – Ionic Salts when reacted with metalsn
17
F9
Uus117
I53
Br35
At85
Cl17
109. © Art Traynor 2011
Chemistry
Structure of Matter
Periodicity
Families of Elements ( Groups )
The Elements may be further classified according to representative “ families ”
exhibiting similar chemical properties. These class-similar elements are arrayed
within the Periodic Table vertically into Groups
Section 2.7 (Pg. 60)
Second Level Equivalence Classifications
Nobel Gases ( Group 18 )
Helium, Neon, Argon, Krypton, Xenon, Radon, Ununoctium
Tend to manifest as gases with little propensity for Chemical Reactionn
18
Uuo118
Ar18
Ne10
Kr36
Xe54
Rn86
He2