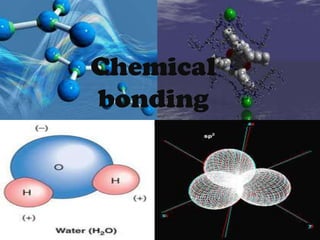
Chemical Bonding Types: Covalent & Ionic
•Download as PPTX, PDF•
0 likes•145 views
Covalent bonding occurs when pairs of electrons are shared between atoms to form a full outer electron shell. Ionic bonding involves the transfer of electrons from one atom to another, resulting in positively and negatively charged ions that are attracted to each other electrostatically to form an ionic compound. Examples of each type of bonding are provided.
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Types Of Chemical Bonds- Ionic Bond,Covalent Bonds,Coordinate Bonds, Basic In...

Types Of Chemical Bonds- Ionic Bond,Covalent Bonds,Coordinate Bonds, Basic In...
Viewers also liked
Viewers also liked (15)
Fin 571 guide 1 39) A firm cannot simply adopt the industry average debt rati...

Fin 571 guide 1 39) A firm cannot simply adopt the industry average debt rati...
Fin 571 guide 1 40) There can be a variety of motives for stock repurchases i...

Fin 571 guide 1 40) There can be a variety of motives for stock repurchases i...
Fin 571 guide 1 34) Net present value ( NPV) is the difference between ______...

Fin 571 guide 1 34) Net present value ( NPV) is the difference between ______...
Recently uploaded
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
Recently uploaded (20)
Introduction to ArtificiaI Intelligence in Higher Education

Introduction to ArtificiaI Intelligence in Higher Education
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Privatization and Disinvestment - Meaning, Objectives, Advantages and Disadva...

Privatization and Disinvestment - Meaning, Objectives, Advantages and Disadva...
18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf

18-04-UA_REPORT_MEDIALITERAСY_INDEX-DM_23-1-final-eng.pdf
Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...

Presentation by Andreas Schleicher Tackling the School Absenteeism Crisis 30 ...
Z Score,T Score, Percential Rank and Box Plot Graph

Z Score,T Score, Percential Rank and Box Plot Graph
TataKelola dan KamSiber Kecerdasan Buatan v022.pdf

TataKelola dan KamSiber Kecerdasan Buatan v022.pdf
Web & Social Media Analytics Previous Year Question Paper.pdf

Web & Social Media Analytics Previous Year Question Paper.pdf
Call Girls in Dwarka Mor Delhi Contact Us 9654467111

Call Girls in Dwarka Mor Delhi Contact Us 9654467111
Chemical Bonding Types: Covalent & Ionic
- 2. Covalent bond Covalent bonding occurs when pairs of electrons are shared by atoms. Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell.
- 3. Examples
- 4. Ionic bond bond Ionic In ionic bonding, electrons are In from electrons are transferred transferredionic bonding,one atomthe another to from one atom to another resulting in formation of positive and negative ions. The resulting in the formation of positive electrostatic attractions between the positive and negative ions.hold the compound and negative ions The electrostatic together. attractions between the positive and negative ions hold the compound together.
- 5. Examples
