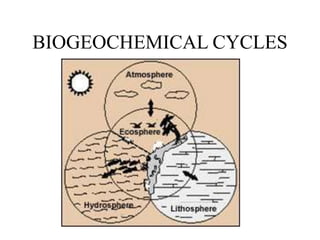
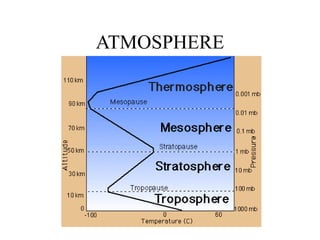
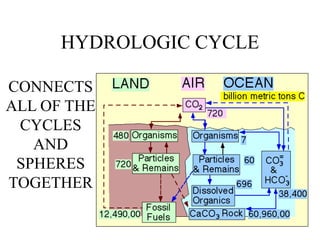
The document discusses several biogeochemical cycles including the hydrologic cycle, carbon cycle, nitrogen cycle, phosphorus cycle, and sulfur cycle. It notes how each cycle moves key elements through the biosphere, lithosphere, hydrosphere and atmosphere. It also describes some major human impacts on each cycle such as withdrawing fresh water, burning fossil fuels, using fertilizers, and emitting pollutants that can disrupt the natural cycling of elements. Understanding these biogeochemical cycles is important because they are integral to maintaining life on Earth.
















![NITROGEN CYCLE
MAJOR STEPS IN NITROGEN CYCLE
• 1. Nitrogen fixation by cyanobacteria or Rhizobium
bacter.
• 2. Ammonification by decomposers and/or nitrogen
fixing bacteria.
• 3. Nitrification by bacteria (NO2 [toxic to plants]to
NO3 [useable by plants]).
4. Assimilation (used by plants) OR Denitrification by
anaerobic bacteria in waterlogged soils or bottom of
lakes convert NH3 and NH4 back to NO2 and NO3
ions and then into N2 and NO2 gas to atmosphere.](https://image.slidesharecdn.com/biogeochemicalcycles-240125194239-836a7472/85/BIOGEOCHEMICAL_CYCLES-ppt-20-320.jpg)






















