- The document reports on the synthesis and characterization of Schiff base ligands derived from L-valine and their Cu(II), Ni(II), and Co(II) complexes.
- Spectroscopic data revealed that the ligands bind as tridentate in the Cu(II) and Ni(II) complexes and bidentate in the Co(II) complex.
- Magnetic susceptibility measurements showed the complexes have a bimetallic structure with antiferromagnetic interactions.
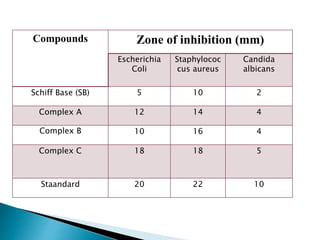
- Antimicrobial testing found the metal complexes to have greater activity than the free ligand, with the Co(II) complex exhibiting the strongest effects.




![ To a hot magnetically stirred ethanolic solution of Schiff base ligands
(0.5 mol), an aqueous solution of metal ions [Cu(II) nitrate (0.5 mol
20ml)] was added drop by drop at 60-65°c.
The mixture was continuously stirred for 30 min the intensity of the
color becomes translucent in nature.
To the translucent solution 0.5mol potassium hydroxide solution was
added to maintain the PH of the medium.
The mixture was further refluxed for 1h. The light green colored
precipitate was obtained, filtered out and washed repeatedly with
ethanol followed by diethyl ether and dried.](https://image.slidesharecdn.com/binuclearcuiiniiiandcoii-190212150937/85/Binuclear-cu-ii-ni-ii-and-co-ii-5-320.jpg)
![ To a hot magnetically stirred ethanolic solution of Schiff base ligand (0.5M),
an aqueous solution of metal ions [Ni(II) nitrate (0.5M,20ml)] was added drop
by drop at 60-65° C.
The mixture was continuously stirred for 30minutes, the intensity of the colour
becomes translucent in nature.
To the translucent solution 0.5M Potassium hydroxide solution was added to
maintain the pH of the solution.
The mixture was further refluxed for 1 hour. The green coloured precipitate
was obtained, filtered out and washed repeatedly with ethanol followed by
diethyl ether and dried.](https://image.slidesharecdn.com/binuclearcuiiniiiandcoii-190212150937/85/Binuclear-cu-ii-ni-ii-and-co-ii-6-320.jpg)
![ To a hot magnetically stirred ethanolic solution of Schiff base ligands (0.5M) an aqueous
solution of metal ions [Co(II) nitrate (0.5M,20ml)] was added drop by drop at 60-65ºC.
The mixture was continuously stirred for 30mins; the intensity of the colour becomes
translucent in nature.
To the translucent solution 0.5M.Potassium hydroxide solution was added to maintain
the pH of the solution.
The mixture was further refluxed for 1 hour. The dirty brown coloured precipitate was
obtained, filtered and washed repeatedly with ethanol followed by diethyl ether and
dried.](https://image.slidesharecdn.com/binuclearcuiiniiiandcoii-190212150937/85/Binuclear-cu-ii-ni-ii-and-co-ii-7-320.jpg)

![ The carboxylate moiety of Schiff base shows, the vasy(COO-) and v sym(COO-)
respectively at 1587 and 1384 cm-1and for complexes has been structurally
characterized by the intense and broad absorptions in the range 1660-1330 cm-
1, due to asymmetric and symmetric stretching modes. The difference between
asymmetric and symmetric O=C=O stretching vibrations (∆v) has been used to
determine the mode of coordination of carboxylate moiety with metal ions. The
∆v= vas(COO-) - vs(COO-) for the complexes (A) (B), and (C) is the following:
∆v= 140cm-1 [vas =1567 – 1427 cm-1 ],142 cm-1 [vas =1565 – 1423 cm-1 ], and
202 cm-1 [1570-1368 cm-1] respectively, and this evidence strongly suggests
that the bridging coordination of carboxylate moiety of Schiff base ligand with
Cu(II) (A) and Ni(II) (B).In contrast, for Co(II) complex(C) monodentate
coordination nature.](https://image.slidesharecdn.com/binuclearcuiiniiiandcoii-190212150937/85/Binuclear-cu-ii-ni-ii-and-co-ii-9-320.jpg)