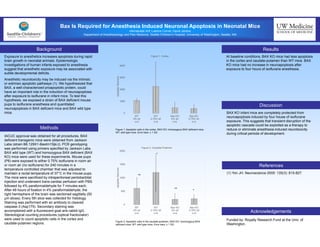
This study investigated the role of the BAX protein in isoflurane-induced neuronal apoptosis in neonatal mice. The researchers found that BAX knockout mice had less baseline apoptosis in the cortex and caudate-putamen compared to wild type mice. Exposure to isoflurane anesthesia for 4 hours did not increase neuroapoptosis in BAX knockout mice, but did increase it in wild type mice. This suggests that blocking the apoptotic cascade by inhibiting BAX may reduce or eliminate anesthesia-induced neurotoxicity in developing brains.