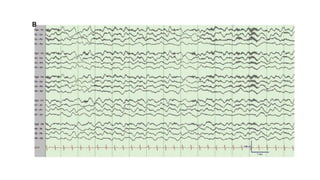
(1) Autoimmune encephalitis can present with acute symptomatic seizures or lead to autoimmune-associated epilepsy in some cases. Seizure types and characteristics can provide clues to the underlying antibody.
(2) Treatment of autoimmune-associated seizures involves immunotherapy targeting the underlying immune process in addition to anti-seizure medications. Immunotherapy is the primary treatment and typically involves initial induction therapy followed by maintenance treatment.
(3) Factors like antibody type, time to treatment, and response to initial immunotherapy can help determine prognosis for seizure remission or recurrence in autoimmune-associated epilepsy. An individualized approach is needed for medication management over time.