ASMS Fall 2018 Metabolomics Informatics Workshop Peak Picking
•Download as PPTX, PDF•
3 likes•768 views
Principles of Peak Picking and Alignment in Pictures and further "doing". ASMS Fall Metabolomics Informatics Workshop 2018. https://www.asms.org/conferences/fall-workshop/program
Report
Share
Report
Share
Recommended
ASMS Fall Metabolomics Informatics Workshop 2018 Identifying Unknown Metabolites

Characterising unknown metabolites talk from the ASMS Fall Metabolomics Informatics Workshop 2018 in San Francisco, California.
https://www.asms.org/conferences/fall-workshop/program
Slides with active hyperlinks accessible via tinyurl on the front page.
Environmental Cheminformatics for Unknown ID UC Davis Nov 2018

Environmental Cheminformatics to Identify Unknown Chemicals and their Effects
Assoc. Prof. Dr. Emma L. Schymanski
FNR ATTRACT Fellow and PI: Environmental Cheminformatics, Luxembourg Centre for Systems Biomedicine (LCSB), University of Luxembourg, 6 avenue du Swing, L-4367 Belvaux, Luxembourg.
The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond.
NOTE: some slides causing errors have been removed but can be accessed through the tinyurl on the front page.
Active hyperlinks can be retrieved using the tinyurl on the front page. Please cite this work if you use any of the contents.
RSC Environmental Cheminformatics to Identify Unknowns Feb 2019

The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond.
Small Molecules in Big Data fTALES Ghent

Finding small molecules in big data, f-TALES meeting in Ghent (https://ftales.be/).
FSUJena Environmental Cheminformatics to Identify Unknowns April 2019

The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond. Kindly hosted by Prof. Christoph Steinbeck
VIB Metabolite ID with MS March 2019

Identifying Metabolites and Small Molecules with Mass Spectrometry - in effect a walk through structure elucidation with MetFrag.
Presentation given for the VIB training event ‘Metabolomics Data Interpretation’
https://training.vib.be/metabolomics-data-interpretation
DMCM2018 Community Resources Connecting Chemistry and Toxicity Knowledge

Community Resources Connecting Chemistry and Toxicity Knowledge to Environmental Observations presented at the Disease Map Community Meeting 2018 in Paris
Small Molecules in Big Data - Analytica Munich

Finding small molecules in big data
E.L. Schymanski, Belvaux/LU, A.J. Williams, North Carolina/USA
Dr. Emma L. Schymanski, Luxembourg Centre for Systems Biomedicine (LCSB), University of Luxembourg, 6 avenue du Swing, L-4367 Belvaux, Luxembourg
Metabolomics and exposomics are amongst the youngest and most dynamic of the omics disciplines. While the molecules involved are smaller than proteomics and the other, larger “omics”, the challenges are in many ways greater. Elements are less constrained, there are no given “puzzle pieces” and there is a resulting explosion in terms of potential chemical space. It is impossible to even enumerate all chemically possible small molecules. The challenges and complexity of identifying small molecules even using the most advanced analytical technologies available today is immense. Current “big data” methods for small molecules rely heavily on chemical databases, the largest of which presently available contain ~100 million chemicals. Despite this large number, high resolution mass spectrometry (HR-MS) measurements contain tens of thousands of features, of which only a few percent can be annotated as “known” and confirmed as metabolites or chemicals of interest using these chemical databases. How can we find relevant small molecules in the ever increasing data loads? How can we annotate more of the unknown features in HR-MS experiments? This talk will present European, US and worldwide initiatives to help find small molecules in big data - from chemical databases to spectral libraries, real-time monitoring to retrospective screening. It will touch on the challenges of standardized structure representations, data curation and deposition. Finally, it will show how interdisciplinary communication, data sharing and pushing the boundaries of current capabilities can facilitate research efforts in metabolomics, exposomics and beyond. This abstract does not necessarily represent U.S. EPA policy.
Recommended
ASMS Fall Metabolomics Informatics Workshop 2018 Identifying Unknown Metabolites

Characterising unknown metabolites talk from the ASMS Fall Metabolomics Informatics Workshop 2018 in San Francisco, California.
https://www.asms.org/conferences/fall-workshop/program
Slides with active hyperlinks accessible via tinyurl on the front page.
Environmental Cheminformatics for Unknown ID UC Davis Nov 2018

Environmental Cheminformatics to Identify Unknown Chemicals and their Effects
Assoc. Prof. Dr. Emma L. Schymanski
FNR ATTRACT Fellow and PI: Environmental Cheminformatics, Luxembourg Centre for Systems Biomedicine (LCSB), University of Luxembourg, 6 avenue du Swing, L-4367 Belvaux, Luxembourg.
The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond.
NOTE: some slides causing errors have been removed but can be accessed through the tinyurl on the front page.
Active hyperlinks can be retrieved using the tinyurl on the front page. Please cite this work if you use any of the contents.
RSC Environmental Cheminformatics to Identify Unknowns Feb 2019

The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond.
Small Molecules in Big Data fTALES Ghent

Finding small molecules in big data, f-TALES meeting in Ghent (https://ftales.be/).
FSUJena Environmental Cheminformatics to Identify Unknowns April 2019

The Environmental Cheminformatics group at the Luxembourg Centre for Systems Biomedicine focuses on the comprehensive identification of known and unknown chemicals in our environment to investigate their effects on health and disease. The environment and the chemicals to which we are exposed is incredibly complex, with over 125 million chemicals registered in the largest chemical registry and over 70,000 in household use alone. Detectable molecules in complex samples can now be captured using high resolution mass spectrometry (HRMS), which provides a “snapshot” of all chemicals present in a sample and allows for retrospective data analysis through digital archiving. However, scientists cannot yet identify the vast majority of the tens of thousands of features in each sample, leading to critical bottlenecks in identification and data interpretation. For instance, recent studies indicate a strong connection between the gut microbiome and Parkinson’s disease, yet over 60 % of significant metabolites in microbiome experiments are unknown. Unknown identification remains extremely time consuming and, in many cases, a matter of luck. Prioritizing efforts to find significant metabolites or potentially toxic substances responsible for observed effects is the key, which involves reconciling highly complex samples with expert knowledge and careful validation. This talk will cover European, US and worldwide community initiatives to help connect knowledge on chemistry and toxicity with environmental observations - from compound databases to spectral libraries and retrospective screening. It will touch on the challenges of standardized structure representations, data curation, deposition and communication between resources. Finally, it will show how interdisciplinary efforts and data sharing can facilitate research in metabolomics, exposomics and beyond. Kindly hosted by Prof. Christoph Steinbeck
VIB Metabolite ID with MS March 2019

Identifying Metabolites and Small Molecules with Mass Spectrometry - in effect a walk through structure elucidation with MetFrag.
Presentation given for the VIB training event ‘Metabolomics Data Interpretation’
https://training.vib.be/metabolomics-data-interpretation
DMCM2018 Community Resources Connecting Chemistry and Toxicity Knowledge

Community Resources Connecting Chemistry and Toxicity Knowledge to Environmental Observations presented at the Disease Map Community Meeting 2018 in Paris
Small Molecules in Big Data - Analytica Munich

Finding small molecules in big data
E.L. Schymanski, Belvaux/LU, A.J. Williams, North Carolina/USA
Dr. Emma L. Schymanski, Luxembourg Centre for Systems Biomedicine (LCSB), University of Luxembourg, 6 avenue du Swing, L-4367 Belvaux, Luxembourg
Metabolomics and exposomics are amongst the youngest and most dynamic of the omics disciplines. While the molecules involved are smaller than proteomics and the other, larger “omics”, the challenges are in many ways greater. Elements are less constrained, there are no given “puzzle pieces” and there is a resulting explosion in terms of potential chemical space. It is impossible to even enumerate all chemically possible small molecules. The challenges and complexity of identifying small molecules even using the most advanced analytical technologies available today is immense. Current “big data” methods for small molecules rely heavily on chemical databases, the largest of which presently available contain ~100 million chemicals. Despite this large number, high resolution mass spectrometry (HR-MS) measurements contain tens of thousands of features, of which only a few percent can be annotated as “known” and confirmed as metabolites or chemicals of interest using these chemical databases. How can we find relevant small molecules in the ever increasing data loads? How can we annotate more of the unknown features in HR-MS experiments? This talk will present European, US and worldwide initiatives to help find small molecules in big data - from chemical databases to spectral libraries, real-time monitoring to retrospective screening. It will touch on the challenges of standardized structure representations, data curation and deposition. Finally, it will show how interdisciplinary communication, data sharing and pushing the boundaries of current capabilities can facilitate research efforts in metabolomics, exposomics and beyond. This abstract does not necessarily represent U.S. EPA policy.
Adding complex expert knowledge into chemical database and transforming surfa...

Adding complex expert knowledge into chemical database and transforming surfa...US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
The increasing popularity of high mass accuracy non-target mass spectrometry methods has yielded extensive identification efforts based on chemical compound databases. Candidate structures are often retrieved with either exact mass or molecular formula from large resources such as PubChem, ChemSpider or the EPA CompTox Chemistry Dashboard. Additional data (e.g. fragmentation, physicochemical properties, reference and data source information) is then used to select potential candidates, depending on the experimental context. However, these strategies require the presence of substances of interest in these compound databases, which is often not the case as no database can be fully inclusive. A prominent example with clear data gaps are surfactants, used in many products in our daily lives, yet often absent as discrete structures in compound databases. Linear alkylbenzene sulfonates (LAS) are a common, high use and high priority surfactant class that have highly complex transformation behaviour in wastewater. Despite extensive reports in the environmental literature, few of the LAS and none of the related transformation products were reported in any compound databases during an investigation into Swiss wastewater effluents, despite these forming the most intense signals. The LAS surfactant class will be used to demonstrate how the coupling of environmental observations with high resolution mass spectrometry and detailed literature data (expert knowledge) on the transformation of these species can be used to progressively “fill the gaps” in compound databases. The LAS and their transformation products have been added to the CompTox Chemistry Dashboard (https://comptox.epa.gov/) using a combination of “representative structures” and “related structures” starting from the structural information contained in the literature. By adding this information into a centralized open resource, future environmental investigations can now profit from the expert knowledge previously scattered throughout the literature. Note: This abstract does not reflect US EPA policy.High throughput mining of the scholarly literature; talk at NIH

The scientific and medical literature contains huge amounts of valuable unused information. This talk shows how to discover it, extract, re-use and interpret it. Wikidata is presented as a key new tool and infrastructure. Everyone can become involved. However some of the barriers to use are sociopolitical and these are identified and discussed.
A Global Commons for Scientific Data: Molecules and Wikidata

Methods for extracting facts from the scientific literature, and linking them to Wikidata IDs. Wikidata is introduced by an architectural example and bioscience. Then we explore how data can be extracted from text and from images
Printout webinar r ax costanza 05 05-2020

Read-across (RAx) is probably the most used strategy to waive new demanding in vivo tests for toxicological assessment of chemicals. It is based on the possibility to translate available information from well-characterized chemicals (source) to the substance for which there is a toxicological data gap (target). In spite of the widespread use, regulatory acceptance is still limited,
New Approach Methods (NAM) may be used to confirm chemical and toxicological similarities and to contribute to the reduction of the uncertainty.
ContentMining for France and Europe; Lessons from 2 years in UK

I have spend 2 years carrying out Content Mining (aka Text and Data Mining) in the UK under the 2014 "Hargreaves" exception. This talk was given in Paris, to ADBU , after France had passed the law of the numeric Republique. I illustrate what worked in what did not and why and offer ideas to France and Europe
High throughput mining of the scholarly literature: journals and theses

Overview of content mining to tech-LING 2016.
Linking the silos. Data and predictive models integration in toxicology.

Silo storage system are typically designed to store one single type of grain. An information silo is characterized by a rigid design, not allowing easy exchange of information and integration with other systems. Life sciences software systems and those applied in toxicology in particular, are often developed independently and compatibility is rarely perceived as a primary design goal. The traditional approach to the integration challenge is to build data warehouses and custom applications managing all the information under a central authority and storing data in a single database schema. The majority of the existing chemical databases rely on a centralized design, offering data access via unique and incompatible interfaces. Loosely coupled systems, allowing end users to share and integrate data on the fly are increasingly considered more appropriate and actively developed [1], but not yet the mainstream in cheminformatics. Our experience in several past and on-going projects, dealing with data integration of chemical structures and toxicity databases, has been reflected in the architecture of the systems that we have designed (e.g. OpenTox, a set of distributed web services, with semantic representation of resources, including data [2], [3], [4]). The core concepts of the information integration are the web service oriented architecture and a data representation design that includes semantic and provenance information uncoupled from the software code and any proprietary metadata.
Towards Responsible Content Mining: A Cambridge perspective

ContentMining (Text and Data Mining) is now legal in the UK for non-commercial research. Cambridge UK is a natural centre, with several components:
* a world-class University and Library
* many publishers, both Open Access and conventional
* a digital culture
* ContentMine - a leading proponent and practitioner of mining
Cambridge University Press welcomes content mining and invited PMR to give a talk there. He showed the technology and protocols and proposed a practical way forward in 2017
Building linked data large-scale chemistry platform - challenges, lessons and...

Chemical databases have been around for decades, but in recent years we have observed a qualitative change from rather small in-house built proprietary databases to large-scale, open and increasingly complex chemistry knowledgebases. This tectonic shift has imposed new requirements for database design and system architecture as well as the implementation of completely new components and workflows which did not exist in chemical databases before. Probably the most profound change is being caused by the linked nature of modern resources - individual databases are becoming nodes and hubs of a huge and truly distributed web of knowledge. This change has important aspects such as data and format standards, interoperability, provenance, security, quality control and metainformation standards.
ChemSpider at the Royal Society of Chemistry was first public chemical database which incorporated rigorous quality control by introducing both community curation and automated quality checks at the scale of tens of millions of records. Yet we have come to realize that this approach may now be incomplete in a quickly changing world of linked data. In this presentation we will talk about challenges associated with building modern public and private chemical databases as well as lessons that we have learned from our past and present experience. We will also talk about solutions for some common problems.
Implementing chemistry platform for OpenPHACTS

The Open PHACTS project delivers an online platform integrating a wide variety of data from across chemistry and the life sciences and an ecosystem of tools and services to query this data in support of pharmacological research, turning the semantic web from a research project into something that can be used by practising medicinal chemists in both academia and industry. In the summer of 2015 it was the first winner of the European Linked Data Award. At the Royal Society of Chemistry we have provided the chemical underpinnings to this system and in this talk we review its development over the past five years. We cover both our early work on semantic modelling of chemistry data for the Open PHACTS triplestore and more recent work building an all-purpose data platform, for which the Open PHACTS data has been an important test case, what has worked well, what's missing and where this is is likely to go in future.
ISMB Workshop 2014

This talk explores how principles derived from experimental design practice, data and computational models can greatly enhance data quality, data generation, data reporting, data publication and data review.
The beauty of workflows and models

Carole Goble at RDMFMeeting, Westminster, 2014-06-20.
Workflows for research. Reproducible research.
NETTAB 2013

Presentation at NETTAB 2013, Venice Lido, Italy. October 2013. http://nettab.org/2013/progr.php
Overview of open resources to support automated structure verification and e...

Overview of open resources to support automated structure verification and e...US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
Cheminformatics methods form an essential basis for providing analytical scientists with access to data, algorithms and workflows. There are an increasing number of free online databases (compound databases, spectral libraries, data repositories) and a rich collection of software approaches that can be used to support automated structure verification and elucidation, specifically for Nuclear Magnetic Resonance (NMR) and Mass Spectrometry (MS). This presentation will provide an overview of freely available data, tools, databases and approaches available to support chemical structure verification and elucidation and highlight some of the known issues regarding data quality and suggest approaches for resolving some of the issues. The importance of structure and spectral standards for data exchange will be discussed, especially with regard to how spectral data can be made openly available to the community via online tools and through scientific publishing. This work does not necessarily reflect U.S. EPA policy.Cheminformatics and the Structure Elucidation of Natural Products

Cheminformatics and the Structure Elucidation of Natural ProductsUS Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
The structure elucidation of natural product structures from analytical data, specifically NMR and MS, remains a major challenge. With an enormous palette of NMR experiments to choose from, and supported by breakthrough technologies in hardware, the generation of high quality data to enable even the most complex of natural product structures to be determined is no longer the major hurdle. The challenge is in the analysis of the data. We are in a new era in terms of approaches to structure elucidation: one where computers, databases, and a synergy between scientists and algorithms can offer an accelerated path forward. Software tools are capable of digesting spectroscopic data to elucidate extremely complex natural products. Scientists can now elucidate chemical structures utilizing multinuclear chemical shift data, correlation data from an array of 2D NMR experiments and utilize existing data sets for the purpose of dereplication and computer-assisted structure elucidation. With the explosion of online data especially, in public databases such as PubChem and ChemSpider, many tens of millions of chemical structures are available to seed fragment databases to include in the elucidation process. This presentation will provide an overview of how cheminformatics and chemical databases have been brought together to assist in the identification of natural products. It will include an examination of the state-of-the-art developments in Computer-Assisted Structure Elucidation. Opening up pharmacological space, the OPEN PHACTs api

Description of the Open PHACTs API presented by at the technology track of the ISMB 20123 meeting in Berlin: 13-07-2013.
Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks

Keynote presentation at the iConference 2015, Newport Beach, Los Angeles, 26 March 2015.
Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks
http://ischools.org/the-iconference/
BEWARE: presentation includes hidden slides AND in situ build animations - best viewed by downloading.
Dr. gerald pfister challenges, solutions and innovations in modern flowcyto...

Dr. gerald pfister challenges, solutions and innovations in modern flowcytometry lab
More Related Content
What's hot
Adding complex expert knowledge into chemical database and transforming surfa...

Adding complex expert knowledge into chemical database and transforming surfa...US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
The increasing popularity of high mass accuracy non-target mass spectrometry methods has yielded extensive identification efforts based on chemical compound databases. Candidate structures are often retrieved with either exact mass or molecular formula from large resources such as PubChem, ChemSpider or the EPA CompTox Chemistry Dashboard. Additional data (e.g. fragmentation, physicochemical properties, reference and data source information) is then used to select potential candidates, depending on the experimental context. However, these strategies require the presence of substances of interest in these compound databases, which is often not the case as no database can be fully inclusive. A prominent example with clear data gaps are surfactants, used in many products in our daily lives, yet often absent as discrete structures in compound databases. Linear alkylbenzene sulfonates (LAS) are a common, high use and high priority surfactant class that have highly complex transformation behaviour in wastewater. Despite extensive reports in the environmental literature, few of the LAS and none of the related transformation products were reported in any compound databases during an investigation into Swiss wastewater effluents, despite these forming the most intense signals. The LAS surfactant class will be used to demonstrate how the coupling of environmental observations with high resolution mass spectrometry and detailed literature data (expert knowledge) on the transformation of these species can be used to progressively “fill the gaps” in compound databases. The LAS and their transformation products have been added to the CompTox Chemistry Dashboard (https://comptox.epa.gov/) using a combination of “representative structures” and “related structures” starting from the structural information contained in the literature. By adding this information into a centralized open resource, future environmental investigations can now profit from the expert knowledge previously scattered throughout the literature. Note: This abstract does not reflect US EPA policy.High throughput mining of the scholarly literature; talk at NIH

The scientific and medical literature contains huge amounts of valuable unused information. This talk shows how to discover it, extract, re-use and interpret it. Wikidata is presented as a key new tool and infrastructure. Everyone can become involved. However some of the barriers to use are sociopolitical and these are identified and discussed.
A Global Commons for Scientific Data: Molecules and Wikidata

Methods for extracting facts from the scientific literature, and linking them to Wikidata IDs. Wikidata is introduced by an architectural example and bioscience. Then we explore how data can be extracted from text and from images
Printout webinar r ax costanza 05 05-2020

Read-across (RAx) is probably the most used strategy to waive new demanding in vivo tests for toxicological assessment of chemicals. It is based on the possibility to translate available information from well-characterized chemicals (source) to the substance for which there is a toxicological data gap (target). In spite of the widespread use, regulatory acceptance is still limited,
New Approach Methods (NAM) may be used to confirm chemical and toxicological similarities and to contribute to the reduction of the uncertainty.
ContentMining for France and Europe; Lessons from 2 years in UK

I have spend 2 years carrying out Content Mining (aka Text and Data Mining) in the UK under the 2014 "Hargreaves" exception. This talk was given in Paris, to ADBU , after France had passed the law of the numeric Republique. I illustrate what worked in what did not and why and offer ideas to France and Europe
High throughput mining of the scholarly literature: journals and theses

Overview of content mining to tech-LING 2016.
Linking the silos. Data and predictive models integration in toxicology.

Silo storage system are typically designed to store one single type of grain. An information silo is characterized by a rigid design, not allowing easy exchange of information and integration with other systems. Life sciences software systems and those applied in toxicology in particular, are often developed independently and compatibility is rarely perceived as a primary design goal. The traditional approach to the integration challenge is to build data warehouses and custom applications managing all the information under a central authority and storing data in a single database schema. The majority of the existing chemical databases rely on a centralized design, offering data access via unique and incompatible interfaces. Loosely coupled systems, allowing end users to share and integrate data on the fly are increasingly considered more appropriate and actively developed [1], but not yet the mainstream in cheminformatics. Our experience in several past and on-going projects, dealing with data integration of chemical structures and toxicity databases, has been reflected in the architecture of the systems that we have designed (e.g. OpenTox, a set of distributed web services, with semantic representation of resources, including data [2], [3], [4]). The core concepts of the information integration are the web service oriented architecture and a data representation design that includes semantic and provenance information uncoupled from the software code and any proprietary metadata.
Towards Responsible Content Mining: A Cambridge perspective

ContentMining (Text and Data Mining) is now legal in the UK for non-commercial research. Cambridge UK is a natural centre, with several components:
* a world-class University and Library
* many publishers, both Open Access and conventional
* a digital culture
* ContentMine - a leading proponent and practitioner of mining
Cambridge University Press welcomes content mining and invited PMR to give a talk there. He showed the technology and protocols and proposed a practical way forward in 2017
Building linked data large-scale chemistry platform - challenges, lessons and...

Chemical databases have been around for decades, but in recent years we have observed a qualitative change from rather small in-house built proprietary databases to large-scale, open and increasingly complex chemistry knowledgebases. This tectonic shift has imposed new requirements for database design and system architecture as well as the implementation of completely new components and workflows which did not exist in chemical databases before. Probably the most profound change is being caused by the linked nature of modern resources - individual databases are becoming nodes and hubs of a huge and truly distributed web of knowledge. This change has important aspects such as data and format standards, interoperability, provenance, security, quality control and metainformation standards.
ChemSpider at the Royal Society of Chemistry was first public chemical database which incorporated rigorous quality control by introducing both community curation and automated quality checks at the scale of tens of millions of records. Yet we have come to realize that this approach may now be incomplete in a quickly changing world of linked data. In this presentation we will talk about challenges associated with building modern public and private chemical databases as well as lessons that we have learned from our past and present experience. We will also talk about solutions for some common problems.
Implementing chemistry platform for OpenPHACTS

The Open PHACTS project delivers an online platform integrating a wide variety of data from across chemistry and the life sciences and an ecosystem of tools and services to query this data in support of pharmacological research, turning the semantic web from a research project into something that can be used by practising medicinal chemists in both academia and industry. In the summer of 2015 it was the first winner of the European Linked Data Award. At the Royal Society of Chemistry we have provided the chemical underpinnings to this system and in this talk we review its development over the past five years. We cover both our early work on semantic modelling of chemistry data for the Open PHACTS triplestore and more recent work building an all-purpose data platform, for which the Open PHACTS data has been an important test case, what has worked well, what's missing and where this is is likely to go in future.
ISMB Workshop 2014

This talk explores how principles derived from experimental design practice, data and computational models can greatly enhance data quality, data generation, data reporting, data publication and data review.
The beauty of workflows and models

Carole Goble at RDMFMeeting, Westminster, 2014-06-20.
Workflows for research. Reproducible research.
NETTAB 2013

Presentation at NETTAB 2013, Venice Lido, Italy. October 2013. http://nettab.org/2013/progr.php
Overview of open resources to support automated structure verification and e...

Overview of open resources to support automated structure verification and e...US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
Cheminformatics methods form an essential basis for providing analytical scientists with access to data, algorithms and workflows. There are an increasing number of free online databases (compound databases, spectral libraries, data repositories) and a rich collection of software approaches that can be used to support automated structure verification and elucidation, specifically for Nuclear Magnetic Resonance (NMR) and Mass Spectrometry (MS). This presentation will provide an overview of freely available data, tools, databases and approaches available to support chemical structure verification and elucidation and highlight some of the known issues regarding data quality and suggest approaches for resolving some of the issues. The importance of structure and spectral standards for data exchange will be discussed, especially with regard to how spectral data can be made openly available to the community via online tools and through scientific publishing. This work does not necessarily reflect U.S. EPA policy.Cheminformatics and the Structure Elucidation of Natural Products

Cheminformatics and the Structure Elucidation of Natural ProductsUS Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
The structure elucidation of natural product structures from analytical data, specifically NMR and MS, remains a major challenge. With an enormous palette of NMR experiments to choose from, and supported by breakthrough technologies in hardware, the generation of high quality data to enable even the most complex of natural product structures to be determined is no longer the major hurdle. The challenge is in the analysis of the data. We are in a new era in terms of approaches to structure elucidation: one where computers, databases, and a synergy between scientists and algorithms can offer an accelerated path forward. Software tools are capable of digesting spectroscopic data to elucidate extremely complex natural products. Scientists can now elucidate chemical structures utilizing multinuclear chemical shift data, correlation data from an array of 2D NMR experiments and utilize existing data sets for the purpose of dereplication and computer-assisted structure elucidation. With the explosion of online data especially, in public databases such as PubChem and ChemSpider, many tens of millions of chemical structures are available to seed fragment databases to include in the elucidation process. This presentation will provide an overview of how cheminformatics and chemical databases have been brought together to assist in the identification of natural products. It will include an examination of the state-of-the-art developments in Computer-Assisted Structure Elucidation. Opening up pharmacological space, the OPEN PHACTs api

Description of the Open PHACTs API presented by at the technology track of the ISMB 20123 meeting in Berlin: 13-07-2013.
Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks

Keynote presentation at the iConference 2015, Newport Beach, Los Angeles, 26 March 2015.
Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks
http://ischools.org/the-iconference/
BEWARE: presentation includes hidden slides AND in situ build animations - best viewed by downloading.
What's hot (20)
Adding complex expert knowledge into chemical database and transforming surfa...

Adding complex expert knowledge into chemical database and transforming surfa...
High throughput mining of the scholarly literature; talk at NIH

High throughput mining of the scholarly literature; talk at NIH
A Global Commons for Scientific Data: Molecules and Wikidata

A Global Commons for Scientific Data: Molecules and Wikidata
ContentMining for France and Europe; Lessons from 2 years in UK

ContentMining for France and Europe; Lessons from 2 years in UK
High throughput mining of the scholarly literature: journals and theses

High throughput mining of the scholarly literature: journals and theses
Linking the silos. Data and predictive models integration in toxicology.

Linking the silos. Data and predictive models integration in toxicology.
Towards Responsible Content Mining: A Cambridge perspective

Towards Responsible Content Mining: A Cambridge perspective
Building linked data large-scale chemistry platform - challenges, lessons and...

Building linked data large-scale chemistry platform - challenges, lessons and...
Overview of open resources to support automated structure verification and e...

Overview of open resources to support automated structure verification and e...
Cheminformatics and the Structure Elucidation of Natural Products

Cheminformatics and the Structure Elucidation of Natural Products
Opening up pharmacological space, the OPEN PHACTs api

Opening up pharmacological space, the OPEN PHACTs api
Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks

Results Vary: The Pragmatics of Reproducibility and Research Object Frameworks
Similar to ASMS Fall 2018 Metabolomics Informatics Workshop Peak Picking
Dr. gerald pfister challenges, solutions and innovations in modern flowcyto...

Dr. gerald pfister challenges, solutions and innovations in modern flowcytometry lab
ChIP-seq - Data processing

This presentation gives an introduction to analysing ChIP-seq data and is part of a bioinformatics workshop. The accompanying websites are available at http://sschmeier.github.io/bioinf-workshop/#!galaxy-chipseq/
Museum impact: linking-up specimens with research published on them

An informal talk for the monthly #SciFri series of talks at the Natural History Museum, London. 2015-09-18
CMSY workshop - Gianpaolo Coro (ISTI-CNR)

This presentation introduces the CMSY model and the use of the BlueBRIDGE e-Infrastructure services to conduct experiments on data-limited stocks.
Lec6: Pre-Processing for Nuclear Medicine Images

2017 Spring, UCF Medical Image Computing
1. The use of PET/SPECT, PET/CT and MRI/PET Images
2. What to measure from Nuclear Medicine Images?
3. Denoising Nuclear MedicineI mages
4. PartialVolumeCorrection
ImageEnhancement • Filtering • Smoothing • Introduction to Medical Image Computing and Toolkits • Image Filtering, Enhancement, Noise Reduction, and Signal Processing • MedicalImageRegistration • MedicalImageSegmentation • MedicalImageVisualization • Machine Learning in Medical Imaging • Shape Modeling/Analysis of Medical Images Deep Learning in Radiology
Delivering The Benefits of Chemical-Biological Integration in Computational T...

Delivering The Benefits of Chemical-Biological Integration in Computational T...US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
Researchers at EPA’s National Center for Computational Toxicology integrate advances in biology, chemistry, and computer science to examine the toxicity of chemicals and help prioritize chemicals for further research based on potential human health risks. The goal of this research program is to quickly evaluate thousands of chemicals, but at a much reduced cost and shorter time frame relative to traditional approaches. The data generated by the Center includes characterization of thousands of chemicals across hundreds of high-throughput screening assays, consumer use and production information, pharmacokinetic properties, literature data, physical-chemical properties as well as the predictive computational modeling of toxicity and exposure. We have developed a number of databases and applications to deliver the data to the public, academic community, industry stakeholders, and regulators. This presentation will provide an overview of our work to develop an architecture that integrates diverse large-scale data from the chemical and biological domains, our approaches to disseminate these data, and the delivery of models supporting predictive computational toxicology. In particular, this presentation will review our new publicly-accessible CompTox Dashboard as the first application built on our newly developed architecture. This abstract does not reflect U.S. EPA policy. Otoacoustic Emissions : A comparison between simulation and lab measures.

Advanced Cochlear Simulator built in matlab allows researchers to run simulations of human's cochlea behavioir by analyzing otoacoustic response.
New Approach Methods - What is That?

New Approach Methods - What is That?US Environmental Protection Agency (EPA), Center for Computational Toxicology and Exposure
Presentation for Texas A&M Superfund Research Center virtual learning series, Big Data in Environmental Science and Toxicology. More details at https://superfund.tamu.edu/big-data-session-2-aug-18-2021/Using open bioactivity data for developing machine-learning prediction models...

Presented at the 256th American Chemical Society (ACS) National Meeting in Boston, MA (August 22, 2018).
==== Abstract ====
The retinoid X receptor (RXR) is a nuclear hormone receptor that functions as a transcription factor with roles in development, cell differentiation, metabolism, and cell death. Chemicals that interfere the RXR signaling pathway may cause adverse effects on human health. In this study, open bioactivity data available at PubChem (https://pubchem.ncbi.nlm.nih.gov) were used to develop prediction models for chemical modulators of RXR-alpha, which is a subtype of RXR that plays a role in metabolic signaling pathways, dermal cysts, cardiac development, insulin sensitization, etc. The models were constructed from quantitative high-throughput screening (qHTS) data from the Tox21 project, using various supervised machine learning methods (including support vector machine, random forest, neural network, k-nearest neighbors, decision tree, and naïve Bayes). The performance of the models was evaluated with an external data set containing bioactivity data submitted by ChEMBL and the NCATS Chemical Genomics Center (NCGC). This study showcases how open data in the public domain can be used to develop prediction models for chemical toxicity.
Managing & Processing Big Data for Cancer Genomics, an insight of Bioinformatics

Managing & Processing Big Data for Cancer Genomics, an insight of Bioinformatics
Genomic Cytometry: Using Multi-Omic Approaches to Increase Dimensionality in ...

Genomic Cytometry: Using Multi-Omic Approaches to Increase Dimensionality in ...Robert (Rob) Salomon
"Genomic Cytometry: Using Multi-Omic Approaches to Increase Dimensionality in Cytometry" was an Invited Tutorial given at the 2019 CYTO conference for the the International Society for the Advancement of Cytometry on the 22nd May 2019. This tutorial was recorded and we expect that it will be converted to a CYTOU webinar in the near future.
This tutorial will begin by explaining why the emerging field of Genomic Cytometry, i.e. the measurement of cells using genomic techniques (e.g. sequencing), in conjunction with more traditional cytometry techniques such as fluorescence, mass and imaging cytometry is becoming a standard tool for biologists looking to unravel complex cellular processes and to develop a deeper understanding of heterogeneity.
We will give a detailed overview of the various technologies that have allowed the emergence of Genomic Cytometry as well as those that continue to push the boundaries of cellular characterisation.
We will then provide a basic overview of the sequencing process such that both research cytometerists and the staff for the cytometry SRL are better equipped to understand the downstream genomic component of Genomic Cytometry.
Finally, we will wrap up the session with case studies that illustrate the power of the genomic cytometry approach and will give a brief outline of where we feel the field needs to go as it matures. We expect attendees will gain a better understanding of 1) the rapidly maturing field of Genomic Cytometry and 2) how Genomic Cytometry should be leveraged into more traditional cytometry workflows.
Similar to ASMS Fall 2018 Metabolomics Informatics Workshop Peak Picking (20)
Dr. gerald pfister challenges, solutions and innovations in modern flowcyto...

Dr. gerald pfister challenges, solutions and innovations in modern flowcyto...
Museum impact: linking-up specimens with research published on them

Museum impact: linking-up specimens with research published on them
Next-generation sequencing course, part 1: technologies

Next-generation sequencing course, part 1: technologies
Delivering The Benefits of Chemical-Biological Integration in Computational T...

Delivering The Benefits of Chemical-Biological Integration in Computational T...
Otoacoustic Emissions : A comparison between simulation and lab measures.

Otoacoustic Emissions : A comparison between simulation and lab measures.
Using open bioactivity data for developing machine-learning prediction models...

Using open bioactivity data for developing machine-learning prediction models...
Managing & Processing Big Data for Cancer Genomics, an insight of Bioinformatics

Managing & Processing Big Data for Cancer Genomics, an insight of Bioinformatics
Genomic Cytometry: Using Multi-Omic Approaches to Increase Dimensionality in ...

Genomic Cytometry: Using Multi-Omic Approaches to Increase Dimensionality in ...
Recently uploaded
Travis Hills' Endeavors in Minnesota: Fostering Environmental and Economic Pr...

Travis Hills of Minnesota developed a method to convert waste into high-value dry fertilizer, significantly enriching soil quality. By providing farmers with a valuable resource derived from waste, Travis Hills helps enhance farm profitability while promoting environmental stewardship. Travis Hills' sustainable practices lead to cost savings and increased revenue for farmers by improving resource efficiency and reducing waste.
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits: A Revolutionary
Breakthrough in Sustainable Energy Science
Nutraceutical market, scope and growth: Herbal drug technology

As consumer awareness of health and wellness rises, the nutraceutical market—which includes goods like functional meals, drinks, and dietary supplements that provide health advantages beyond basic nutrition—is growing significantly. As healthcare expenses rise, the population ages, and people want natural and preventative health solutions more and more, this industry is increasing quickly. Further driving market expansion are product formulation innovations and the use of cutting-edge technology for customized nutrition. With its worldwide reach, the nutraceutical industry is expected to keep growing and provide significant chances for research and investment in a number of categories, including vitamins, minerals, probiotics, and herbal supplements.
DERIVATION OF MODIFIED BERNOULLI EQUATION WITH VISCOUS EFFECTS AND TERMINAL V...

In this book, we use conservation of energy techniques on a fluid element to derive the Modified Bernoulli equation of flow with viscous or friction effects. We derive the general equation of flow/ velocity and then from this we derive the Pouiselle flow equation, the transition flow equation and the turbulent flow equation. In the situations where there are no viscous effects , the equation reduces to the Bernoulli equation. From experimental results, we are able to include other terms in the Bernoulli equation. We also look at cases where pressure gradients exist. We use the Modified Bernoulli equation to derive equations of flow rate for pipes of different cross sectional areas connected together. We also extend our techniques of energy conservation to a sphere falling in a viscous medium under the effect of gravity. We demonstrate Stokes equation of terminal velocity and turbulent flow equation. We look at a way of calculating the time taken for a body to fall in a viscous medium. We also look at the general equation of terminal velocity.
Richard's aventures in two entangled wonderlands

Since the loophole-free Bell experiments of 2020 and the Nobel prizes in physics of 2022, critics of Bell's work have retreated to the fortress of super-determinism. Now, super-determinism is a derogatory word - it just means "determinism". Palmer, Hance and Hossenfelder argue that quantum mechanics and determinism are not incompatible, using a sophisticated mathematical construction based on a subtle thinning of allowed states and measurements in quantum mechanics, such that what is left appears to make Bell's argument fail, without altering the empirical predictions of quantum mechanics. I think however that it is a smoke screen, and the slogan "lost in math" comes to my mind. I will discuss some other recent disproofs of Bell's theorem using the language of causality based on causal graphs. Causal thinking is also central to law and justice. I will mention surprising connections to my work on serial killer nurse cases, in particular the Dutch case of Lucia de Berk and the current UK case of Lucy Letby.
bordetella pertussis.................................ppt

Bordettela is a gram negative cocobacilli spread by air born drop let
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Since volcanic activity was first discovered on Io from Voyager images in 1979, changes
on Io’s surface have been monitored from both spacecraft and ground-based telescopes.
Here, we present the highest spatial resolution images of Io ever obtained from a groundbased telescope. These images, acquired by the SHARK-VIS instrument on the Large
Binocular Telescope, show evidence of a major resurfacing event on Io’s trailing hemisphere. When compared to the most recent spacecraft images, the SHARK-VIS images
show that a plume deposit from a powerful eruption at Pillan Patera has covered part
of the long-lived Pele plume deposit. Although this type of resurfacing event may be common on Io, few have been detected due to the rarity of spacecraft visits and the previously low spatial resolution available from Earth-based telescopes. The SHARK-VIS instrument ushers in a new era of high resolution imaging of Io’s surface using adaptive
optics at visible wavelengths.
What is greenhouse gasses and how many gasses are there to affect the Earth.

What are greenhouse gasses how they affect the earth and its environment what is the future of the environment and earth how the weather and the climate effects.
Salas, V. (2024) "John of St. Thomas (Poinsot) on the Science of Sacred Theol...

I Introduction
II Subalternation and Theology
III Theology and Dogmatic Declarations
IV The Mixed Principles of Theology
V Virtual Revelation: The Unity of Theology
VI Theology as a Natural Science
VII Theology’s Certitude
VIII Conclusion
Notes
Bibliography
All the contents are fully attributable to the author, Doctor Victor Salas. Should you wish to get this text republished, get in touch with the author or the editorial committee of the Studia Poinsotiana. Insofar as possible, we will be happy to broker your contact.
ANAMOLOUS SECONDARY GROWTH IN DICOT ROOTS.pptx

Abnormal or anomalous secondary growth in plants. It defines secondary growth as an increase in plant girth due to vascular cambium or cork cambium. Anomalous secondary growth does not follow the normal pattern of a single vascular cambium producing xylem internally and phloem externally.
20240520 Planning a Circuit Simulator in JavaScript.pptx

Evaporation step counter work. I have done a physical experiment.
(Work in progress.)
Shallowest Oil Discovery of Turkiye.pptx

The Petroleum System of the Çukurova Field - the Shallowest Oil Discovery of Türkiye, Adana
ESR spectroscopy in liquid food and beverages.pptx

With increasing population, people need to rely on packaged food stuffs. Packaging of food materials requires the preservation of food. There are various methods for the treatment of food to preserve them and irradiation treatment of food is one of them. It is the most common and the most harmless method for the food preservation as it does not alter the necessary micronutrients of food materials. Although irradiated food doesn’t cause any harm to the human health but still the quality assessment of food is required to provide consumers with necessary information about the food. ESR spectroscopy is the most sophisticated way to investigate the quality of the food and the free radicals induced during the processing of the food. ESR spin trapping technique is useful for the detection of highly unstable radicals in the food. The antioxidant capability of liquid food and beverages in mainly performed by spin trapping technique.
Recently uploaded (20)
Travis Hills' Endeavors in Minnesota: Fostering Environmental and Economic Pr...

Travis Hills' Endeavors in Minnesota: Fostering Environmental and Economic Pr...
Unveiling the Energy Potential of Marshmallow Deposits.pdf

Unveiling the Energy Potential of Marshmallow Deposits.pdf
Lateral Ventricles.pdf very easy good diagrams comprehensive

Lateral Ventricles.pdf very easy good diagrams comprehensive
Nutraceutical market, scope and growth: Herbal drug technology

Nutraceutical market, scope and growth: Herbal drug technology
DERIVATION OF MODIFIED BERNOULLI EQUATION WITH VISCOUS EFFECTS AND TERMINAL V...

DERIVATION OF MODIFIED BERNOULLI EQUATION WITH VISCOUS EFFECTS AND TERMINAL V...
bordetella pertussis.................................ppt

bordetella pertussis.................................ppt
Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...

Observation of Io’s Resurfacing via Plume Deposition Using Ground-based Adapt...
PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION

PRESENTATION ABOUT PRINCIPLE OF COSMATIC EVALUATION
What is greenhouse gasses and how many gasses are there to affect the Earth.

What is greenhouse gasses and how many gasses are there to affect the Earth.
Salas, V. (2024) "John of St. Thomas (Poinsot) on the Science of Sacred Theol...

Salas, V. (2024) "John of St. Thomas (Poinsot) on the Science of Sacred Theol...
20240520 Planning a Circuit Simulator in JavaScript.pptx

20240520 Planning a Circuit Simulator in JavaScript.pptx
Mudde & Rovira Kaltwasser. - Populism - a very short introduction [2017].pdf![Mudde & Rovira Kaltwasser. - Populism - a very short introduction [2017].pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Mudde & Rovira Kaltwasser. - Populism - a very short introduction [2017].pdf](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Mudde & Rovira Kaltwasser. - Populism - a very short introduction [2017].pdf
ESR spectroscopy in liquid food and beverages.pptx

ESR spectroscopy in liquid food and beverages.pptx
ASMS Fall 2018 Metabolomics Informatics Workshop Peak Picking
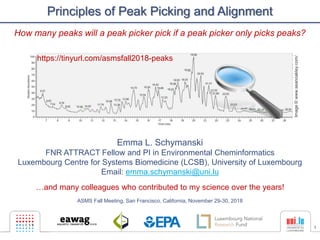
- 1. 1 Principles of Peak Picking and Alignment Emma L. Schymanski FNR ATTRACT Fellow and PI in Environmental Cheminformatics Luxembourg Centre for Systems Biomedicine (LCSB), University of Luxembourg Email: emma.schymanski@uni.lu …and many colleagues who contributed to my science over the years! ASMS Fall Meeting, San Francisco, California, November 29-30, 2018 Image©www.seanoakley.com/ https://tinyurl.com/asmsfall2018-peaks How many peaks will a peak picker pick if a peak picker only picks peaks?
- 2. 2 (nevertheless, I will do my best!) DISCLAIMER! MS1 MS2 Two very different worlds …
- 3. 3 Presenting Peak Picking: Plan o Why Peak Pick o Terminology • Peak Picking vs Centroid vs Profile … o Peak Picking & Peak Pickers • “best of” xcms and enviPick • Peak Picking in Pictures • Peak Picking Parameters • Alleviating Peak Picking Parameter Panic o Alignment ( / Profiling) • “best of” xcms and enviMass o Peak Picking Pointers o Don’t just listen to me … do it!
- 4. 4 Why Peak Pick (I) Example scheme of liquid chromatography - mass spectrometry Image © www.planetorbitrap.com/q-exactive Sampling Extraction (SPE) HPLC separation HR-MS/MS
- 5. 5 Why Peak Pick (II) This is what the output “really” looks like … Image © www.planetorbitrap.com/q-exactive
- 6. 6 Why Peak Pick (III) Identification = turning numbers into structures N N N S CH3 NHNH CH3 CH3 CH3 N N N S CH3 NHNHCH3 CH3 OH P O S SO CH3 CH3 CH3 P OHS S O CH3 CH3 OH CH3 S O O OH CH3 CH3 S N S O O OH S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 N N N S NHNH CH3 CH3 CH3 NH2 OH O massbank.eu
- 7. 7 TERMINOLOGY! o Peak picking can be multi-directional, i.e. • in mass… or time…
- 8. 8 Mass: Centroid vs Profile Data (enviPat) https://www.envipat.eawag.ch/index.php and Loos et al Anal. Chem. 87(11), 5738-5744. DOI: 10.1021/acs.analchem.5b00941
- 9. 9 Mass: Centroid vs Profile Data (enviPat) https://www.envipat.eawag.ch/index.php and Loos et al Anal. Chem. 87(11), 5738-5744. DOI: 10.1021/acs.analchem.5b00941
- 10. 10 TERMINOLOGY! http://proteowizard.sourceforge.net/ o Peak picking can be multi-directional (mass, time) • Peak picking in Proteowizard MSConvert is “centroiding” masses (turning profile mode data into centroided data for efficient processing)
- 11. 11 Peak Picking (in time) Source: R. Tautenhahn, C. Böttcher, S. Neumann, BMC Bioinformatics 2008, 9:504. DOI: 10.1186/1471-2105-9-504 o Peak picking along time axis (chromatographic peaks)
- 12. 12 Peak Picking Source: R. Tautenhahn, C. Böttcher, S. Neumann, BMC Bioinformatics 2008, 9:504. DOI: 10.1186/1471-2105-9-504 o Peak picking along time axis (chromatographic peaks)
- 13. 13 Peak Picking Source: Johannes Rainer; http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html o Peak picking along time axis (chromatographic peaks)
- 14. 14 Peak Picking Source: Johannes Rainer; http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html o Peak picking along time axis (chromatographic peaks) Several Samples Overlaid Red = KO Blue = wild type Rectangle = chromatographic peaks identified per sample
- 15. 15 Peak Picking o Several options for peak picking • XCMS and centWave • Tautenhahn et al 2008 DOI: 10.1186/1471-2105-9-504 • http://bioconductor.org/packages/xcms/ • MZmine 2 • Pluskal et al 2010 DOI: 10.1186/1471-2105-11-395 • http://mzmine.github.io/ • enviPick / enviMass • Loos 2018 DOI: 10.5281/zenodo.1213098 • http://www.looscomputing.ch/eng/enviMass/overview.htm • Plenty of other open, research and vendor options ...
- 16. 16 Peak Picking o Result is something like this (from Formulator output):
- 17. 17 Peak Picking – XCMS & XCMS Online o http://bioconductor.org/packages/xcms/
- 18. 18 Peak Picking – XCMS & XCMS Online o https://xcmsonline.scripps.edu/
- 19. 19 Peak Picking – enviMass and enviPick o http://www.looscomputing.ch/eng/enviMass/overview.htm o R packages …
- 20. 20 Peak Picking in Pictures http://www.looscomputing.ch/eng/enviMass/topics/peakpicking.htm Red = peaks Grey = noise
- 21. 21 Peak Picking .. Somewhat simpler picture http://www.looscomputing.ch/eng/enviMass/topics/peakpicking.htm
- 22. 22 centWave – Gaussian with “Mexican Hat” https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-9-504
- 23. 23 centWave – Gaussian with “Mexican Hat” https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-9-504
- 24. 24 centWave – Gaussian with “Mexican Hat” https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-9-504
- 25. 25 But … peaks are not perfect! http://www.looscomputing.ch/eng/enviMass/topics/peakpicking.htm o See enviMass website for explanation …
- 26. 26 Critical Point: Separating Peaks from Baseline
- 27. 27 Peak Picking Parameters https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-9-504 o There are a lot of options to tweak! • I will just run through (main) centWave parameters • enviPick is too complicated => further reading!
- 28. 28 Peak Picking Parameters: centWave ppm maximal tolerated m/z deviation in consecutive scans, in ppm (parts per million) NOTE: dependent on your mass spectrometer
- 29. 29 Peak Picking Parameters: centWave peakwidth Chromatographic peak width, given as range (min,max) in seconds NOTE: highly dependent on your chromatography!
- 30. 30 Peak Picking Parameters: centWave snthresh Signal to noise ratio cutoff
- 31. 31 Peak Picking Parameters: centWave prefilter prefilter=c(k,I). Prefilter step for the first phase. Mass traces are only retained if they contain at least k peaks with intensity >= I Only one “stick” so will fail recommended prefilter settings
- 32. 32 Too Many Peak Picking Parameters ??????? https://bioconductor.org/packages/ release/bioc/vignettes/IPO/inst/doc /IPO.html o IPO to the rescue! o Parameter optimization for xcms-based workflows … o Libiseller et al 2015, DOI: 10.1186/s12859-015-0562-8 IPO = Isotopologue Parameter Optimization
- 33. 33 Too Many Peak Picking Parameters ???????
- 34. 34 RECAP: Why Peak Pick? Identification = turning numbers into structures N N N S CH3 NHNH CH3 CH3 CH3 N N N S CH3 NHNHCH3 CH3 OH P O S SO CH3 CH3 CH3 P OHS S O CH3 CH3 OH CH3 S O O OH CH3 CH3 S N S O O OH S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 S O O OH CH3 CH3 N N N S NHNH CH3 CH3 CH3 NH2 OH O massbank.eu
- 35. 35 o Instruments change over time … o Before we can do fancy statistics, we need to make sure our samples are comparable!
- 36. 36 Alignment http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html#3_initial_data_inspection o Alignment / Profiling => which peaks belong together across large sample sets?
- 37. 37 Alignment http://www.looscomputing.ch/eng/enviMass/topics/profiling.htm o “Profiling” in enviMass
- 38. 38 Alignment ~= Retention Time Correction http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html#3_initial_data_inspection o Many algorithms and methods … o Before:
- 39. 39 Alignment ~= Retention Time Correction http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html#5_alignment o Many algorithms and methods … o After (Obiwarp algorithm in xcms)
- 41. 41 Changes over samples http://bioconductor.org/packages/release/bioc/vignettes/xcms/inst/doc/xcms.html#5_alignment o Difference between adjusted and raw retention times along the retention time axis
- 42. 42 Some advice … o Peak pickers are designed to pick the perfect peak • But life is never perfect and peaks are no different o Pick the peak picker that is best for your situation • Convenience, ease of use, designed for your data, … • The optimal choice is usually a compromise o Be sceptical (visualise your data, reality check it, etc.) • But don’t go overboard in evaluating peak pickers … remember your (real) goal …
- 43. 43 Peak Picking Overlap (centWave paper) https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-9-504
- 44. 44 Verify with EIC Extraction [these are NOT picked] https://github.com/schymane/ReSOLUTION/blob/master/R/RMB_EIC_prescreen.R No peak at all Nice peak, MSMS Peak, no MSMS Noise with MSMS (careful!) Isobars with MSMS (careful!)* Looking for chemicals known to be present in the sample
- 45. 45 Just because you find a peak … ENTACT Project: https://www.epa.gov/sites/production/files/2018-06/documents/comptox_cop_6-28-18.pdf o Mix 505: One candidate with this mass/formula • DTXSID9040001, C9H8O4 o One chemical… How many peaks?
- 46. 46 …doesn’t mean it’s your compound of interest!
- 47. 47 Beware of artefacts! o Your results also depend on the acquisition data!
- 48. 48 Further reading DOING! [Vendor independent] o Don’t just take my word for it … don’t just read about it … DO IT. There are so many ways to try it out … complete with sample data! [Open Science!] o http://bioconductor.org/packages/release/bioc/vignettes/x cms/inst/doc/xcms.html o http://www.looscomputing.ch/eng/enviMass/overview.htm o An interface that many enjoy, likely comes with example data but requires a login … o https://xcmsonline.scripps.edu/
- 49. 49 Further reading DOING! [Vendor independent] o http://mzmine.github.io/ o http://prime.psc.riken.jp/Metabolomics_Software/MS-DIAL/ o MS-DIAL
- 50. 50 Acknowledgements emma.schymanski@uni.lu Further Information: http://bioconductor.org/packages/xcms/ http://www.looscomputing.ch/eng/enviMass/overview.htm https://xcmsonline.scripps.edu/ http://mzmine.github.io/ EU Grant 603437 The CompMS Community (proxy photo)
- 51. 51 Extra Slides
- 52. 52 Quality Control of Data Slide c/o Michael Stravs o Always visualise results … never take anything for granted
- 53. 53 Homologues: Challenge Peak Pickers but are Present! Stravs et al. (2013), J. Mass Spectrom, 48(1):89-99. DOI: 10.1002/jms.3131 OHSO O CH3 O OH m n SPA-9C m+n=6 www.massbank.eu ACCESSIONS (LAS, SPACs): Literature MS/MS LIT00034, LIT00037 Std Mix., Sample ETS00012, ETS00018https://github.com/MassBank/RMassBank/ Tentatively Identified Spectra: http://goo.gl/0t7jGp
- 54. 54 Be wary of instrument specific phenomena! o R package nontarget: satellite peak removal
- 55. 55 Be wary of instrument specific phenomena II o Orbitrap-specific calibration issues (not observed in TOF)