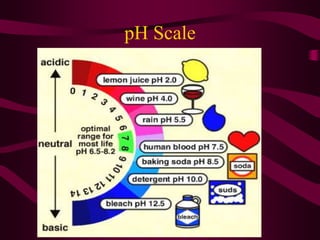
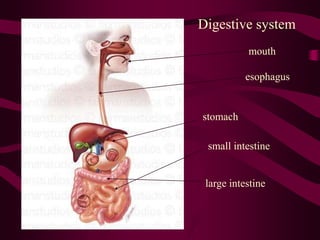
Acids have a pH below 7 and donate hydrogen ions in solution, forming H+ ions. They react with metals and bases. Bases have a pH above 7 and accept hydrogen ions, forming OH- ions. Common acids include HCl and H2SO4, while common bases include NaOH and Mg(OH)2. A salt is formed during a neutralization reaction between an acid and a base, producing water. The digestive system uses acids and bases to break down foods, with the stomach having a highly acidic pH around 2 to aid in digestion.