ABCG5/ABCG8: A Structural View on Sterol Transport
- 1. ABCG5/ABCG8: A Structural View on Sterol Transport Jyh-Yeuan (Eric) Lee University of Texas Southwestern Medical Center at Dallas
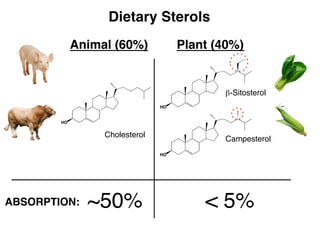
- 2. Dietary Sterols Cholesterol Animal (60%) Plant (40%) ABSORPTION: ~50% < 5% β-Sitosterol Campesterol
- 3. • Autosomal recessive genetic disease (50-120x plasma sitosterol) • Heterodimeric ATP-binding cassette (ABC) transporters • Amino acid sequence identity: ~28% Sitosterolemia v.s. ABCG5 & ABCG8 (Bhattacharyya & Conner, JCI, 1974) (Berge & Tian et al, Science, 2000) (Lee et al, Nat. Genet., 2001) (Lu et al, Am. J. Hum. Genet., 2001) (2p21) A: Walker A motif (GxxGxGKS/T) B: Walker B motif (ϕϕϕϕDE) S: ABC signature motif (ϕSGGQ/E) ϕ: hydrophobic amino acids Transmembrane domain Nucleotide-binding domain
- 4. Enterocyte NPC1L1 Chylomicron Small Intestine Cholesterol Plant Sterols G5G8 DIET Hepatocyte Liver G5G8 Bile Canaliculus Liver/Intestine-Specific Sterol Transporter
- 5. Enterocyte NPC1L1 Chylomicron Small Intestine Cholesterol Plant Sterols G5G8 DIET Hepatocyte Liver G5G8 Bile Canaliculus Inactivating Mutations ⇒ Sitosterolemia
- 6. Animal Model & in vivo Reconstitution (Yu et al, PNAS, 2002) +/+-/- G5G8-/- G5 & G8 restore biliary sterol secretion: (obligate heterodimers) mG5: mG8: Calnexin: Precursor Mature (Graf et al, JBC, 2003) bile extraction G5G8-/- mice develop sitosterolemia:
- 7. Asymmetric Nucleotide-Binding Sites (NBS) A: Walker A S: ABC Signature GSSGSGKT LSGGE GSSGCGRA ISTGE B: Walker B NBS1 NBS2 (Zhang et al, JBC, 2006) (Wang et al, JBC, 2011) Sterol Transport (mouse model) (inactivating mutations) VMLFDE ILILDE
- 8. G5G8-Mediated Sterol Transport (Functional Asymmetry Model)
- 9. TMD NBD Questions: • How is the ATPase activity coupled to sterol transport? • How does TMD move sterols across the membranes? Molecular Mechanism of G5G8 ? ? ABCB exporters nucleotide-free Nucleotide-bound ATP Sterols ?
- 10. 0 10 20 30 40 0 2000 4000 6000 Large-scale Purification of Human G5G8 Tandem Affinity Chromatography: (Pichia pastoris yeast) Ni-NTA CBP Gel filtration CBP: calmodulin-binding peptide (2-3 mg/L) (1-2 mg/L) 100 75 SDS-PAGE: 3C: 3C cleavage site ATPase activity: (Bile acid stimulation assay) Liver polar lipids 1% cholate (Wang et al, Biochemistry, 2006) (Johnson et al, Biochemistry, 2010) (WT/GD: G5WT/G8-G216D) 50 (WT/WT) nmolPi/mgproteins
- 11. Optimization of Protein Preparation 1° Ni-NTA 1° CBP Gel filtration Shaker culture (4-5 days) Membrane Prepara:on Solubiliza:on (β-DDM) DDM MNG + Endo H + 3C protease Methyla:on (CH3-Lys) Relipida:on Synthe:c phospholipids (Avan:) ± ATPase inhibitor Crystalliza:on (manual/robot set-up) (cholesterol) Crystal growth (2 wks – 6 mths) Ligands ± Mg/ATP 2° CBP2° Ni-NTA DDM MNG Detergent Exchange PD-10 (desalting) Alkyla:on (Cys capping) (DoDecyl Maltoside) (Maltose Neopentyl Glycol)
- 12. 100 nm Analytical gel filtration: Negative-stained TEM single particles: (FEI Tecnai G2) ABCB1 Void G5G8 G5G8 (Lee et al, J Biol Chem, 2002) Stable and Monodisperse G5G8
- 13. Bicelle Crystallization (Lipid Bilayers) G5G8 bicelle preparation Bilayer (DMPC + cholesterol) Micelle (CHAPSO) (DHPC) Bicelle G5G8 G5G8 (MNG) Crystal growth & X-ray diffraction 100 µm • Long exposure 2-5 sec @ APS 30 sec @ ALS • Radiation damage 3-5 frames (< 5°) • Signal (I/σ = 1-1.5 at 3.9-4Å) 3.9 Å 3.6 Å
- 14. Model Building & Refinement (Rwork/Rfree = 0.242/0.328) W-SAD (tungsten single anomalous dispersion)
- 15. (Hollenstein et al, Curr Opin Struct Biol, 2007) (Rees et al, Nature Rev Mol Cell Biol, 2009) New TMD Fold for ABC Transporters (Lee et al, Nature, 2016)
- 16. Domain Features in G5G8 Heterodimer RMSD ~ 2Å (~28% sequence identity) TMD: transmembrane domain NBD: nucleotide-binding domain ECD: extracellular domain CnH: connecting helix CpH: coupling helix (Lee et al, Nature, 2016)
- 17. CpH/CnH/E-helix Triple Helical Bundles (Connecting TMD with NBS) CnH: connecting helix CpH: coupling helix ★: conserved polar residues
- 18. TMD Polar Relays (Conserved Polar residues Proximal to NBS) (Lee et al, Nature, 2016)
- 19. : conserved; : not conservedFo - Fc (3.0 σ)Vestibules Vestibules at TMD-Membrane Interface: Sterol Binding/Entry?
- 20. A540 F540 * * Precursor Mature Vestibules at TMD-Membrane Interface: Structure-based Mutagenesis (Lee et al, Nature, 2016)
- 21. ER-escape mutations Non-ER-escape mutations E146Q R419H/P R543S G574R R389H N437K R184H P231T R263Q L501P G574E L596R G5 G5G8 G8 Missense Mutations in Sitosterolemia (Graf et al, JBC, 2004) : conserved (multiple sequence alignment (MSA) value ≥ 7) : less/non-conserved (MSA < 7)
- 22. E146Q R419H/P R543S G574R R389H N437K R184H P231T R263Q L501P G574E L596R G5 G5G8 G8 Missense Mutations in Sitosterolemia (Graf et al, JBC, 2004) : conserved (multiple sequence alignment (MSA) value ≥ 7) : less/non-conserved (MSA < 7) Polar relay Polar relay E-helix Sterol exit? Vestibule
- 23. G5G8-Mediated Sterol Transport Inward movement (CpH/CnH/E-helix bundle) G5 G8 CnH CpH E-helix CnH CpH E-helix Upward movement (TM helices) G5 G8 4 3 1 6 4 3 Molecular Dynamics Simulation
- 25. (Lee et al, Nature, 2016)
- 26. G5G8-Mediated Sterol Transport (Updated Model) Inward Upward
- 27. Summary • G5G8 bicelle crystal structure at 3.9 Å (lipid bilayer-bound) • New TMD fold for ABC transporters • Cpn/CnH/E-helix bundle connects NBS to TMD • Asymmetric interaction between CnH & CpH • Conserved polar residues proximal to NBS (TMD polar relays) • Updated model for G5G8-mediated sterol transport • Structure-based mutagenesis at sterol-interacting site • Structure-based interpretation for disease-causing mutations
- 28. Vertebrates Eukaryotic ABCG Exporters (TMD-based CLANS Network) (Lee et al, Nature, 2016)
- 29. ABC2 Exporter Superfamily CLANS Network (Lee et al, Nature, 2016)
- 30. Acknowledgement • UT Southwestern: Helen Hobbs Jonathan Cohen Daniel Rosenbaum Zbyszek Otwinowski Dominika Borek Nick Grishin Lisa Kinch Xiao-Song Xie Jin Wang Junmei Wang Structural Biology Lab (SBL) • Texas Tech HSC: Ina Urbatsch • Beamlines: APS 19ID, 23ID (Argonne) ALS 821, 822 (Berkeley) • Funding: National Institute of Health American Heart Association Howard Hughes Medical Institute
