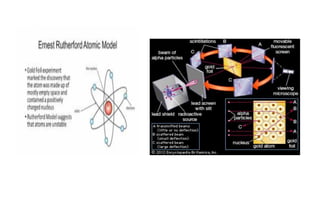
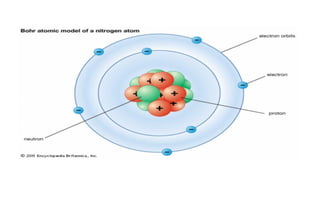
This document outlines the lesson plan for a chemistry class covering topics on atomic structure and chemical bonding. The week will focus on flame tests and electron configuration on Monday, the quantum mechanical model of the atom and predicting electron locations on Tuesday, and writing electron configurations on Wednesday. Thursday and Friday's lessons will cover ionic bonding, covalent bonding, and metallic bonding. Activities include flame testing, writing electron configurations, drawing atomic models, and explaining bonding types. Videos and online resources will supplement the lessons. The teacher will assess student understanding through activities and a short quiz each day.