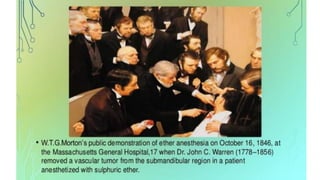
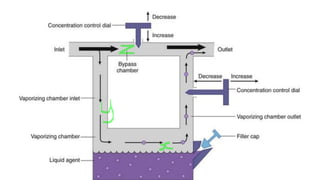
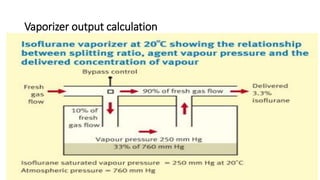
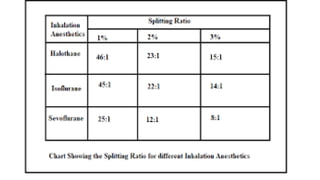
1) Inhalational agents are commonly used to maintain general anesthesia by providing hypnosis. They work by disrupting synaptic transmission in the CNS through various proposed mechanisms including lipid membrane interactions.
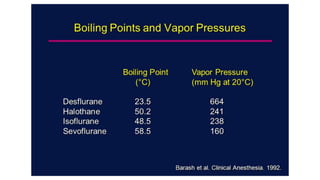
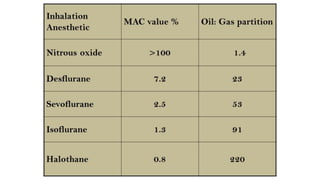
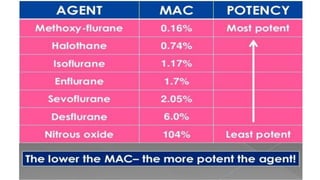
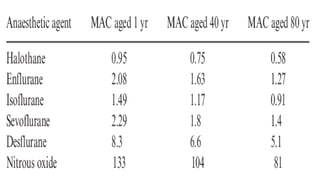
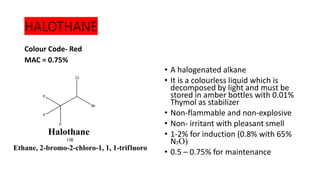
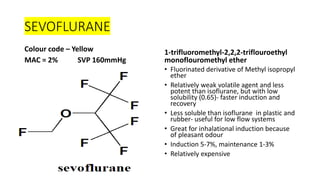
2) The minimum alveolar concentration (MAC) measures the potency of an agent and is defined as the alveolar concentration that prevents movement in response to surgical incision in 50% of subjects. Lower MAC values indicate greater potency.
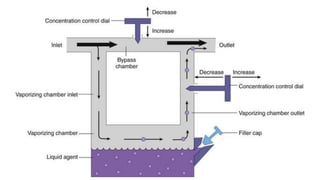
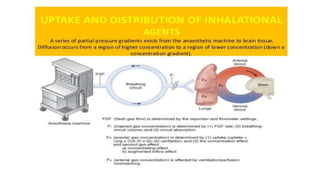
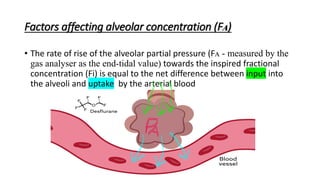
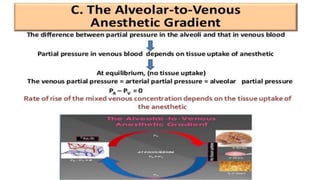
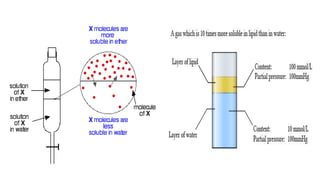
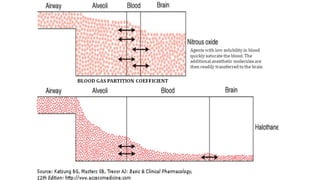
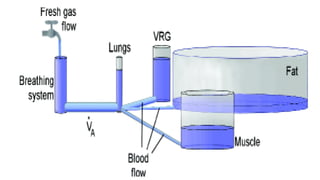
3) Uptake and distribution of inhalational agents in the body is determined by factors such as solubility, cardiac output, alveolar ventilation and fresh gas flow. Their elimination primarily occurs through exhalation from the lungs.