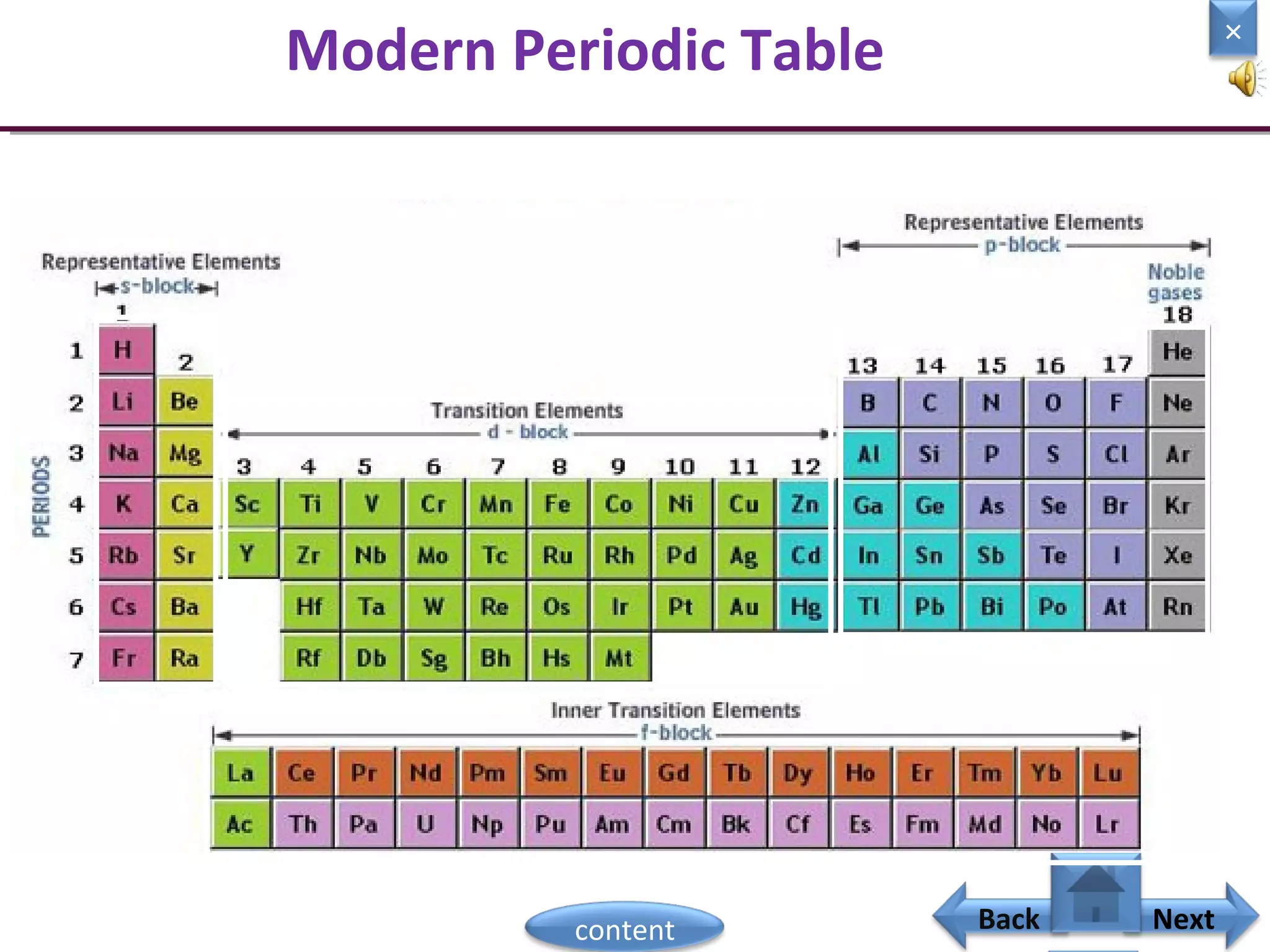
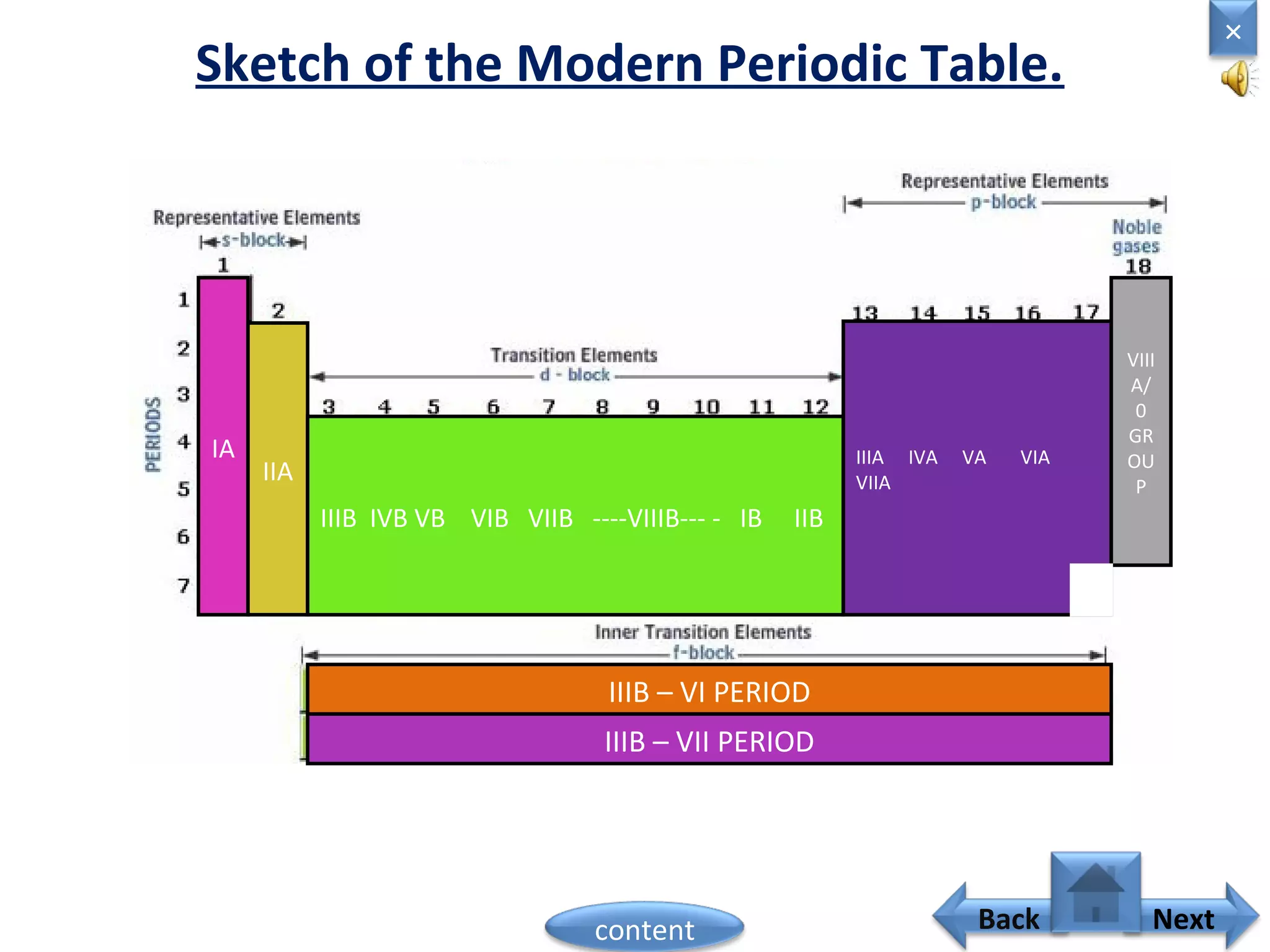
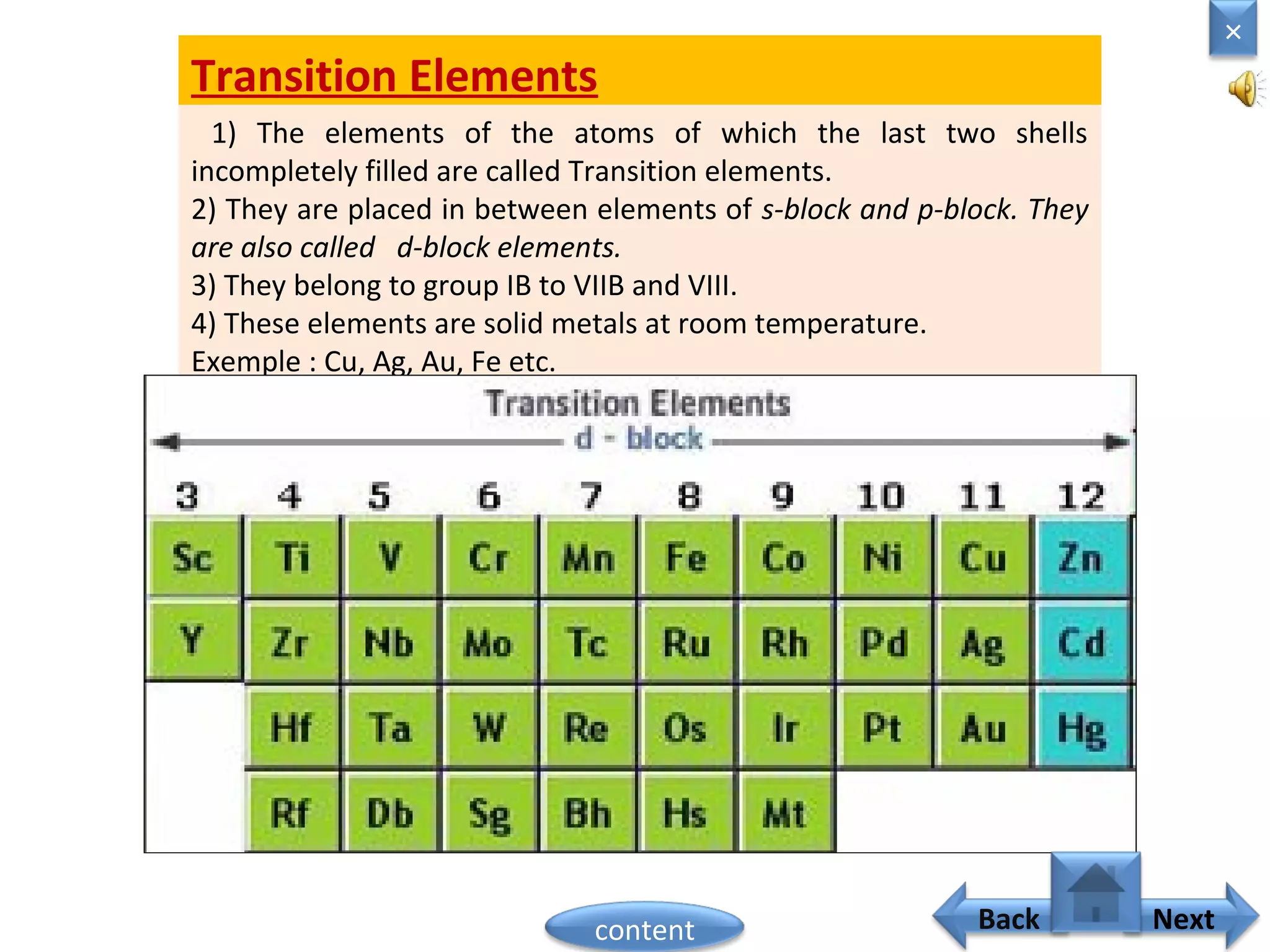
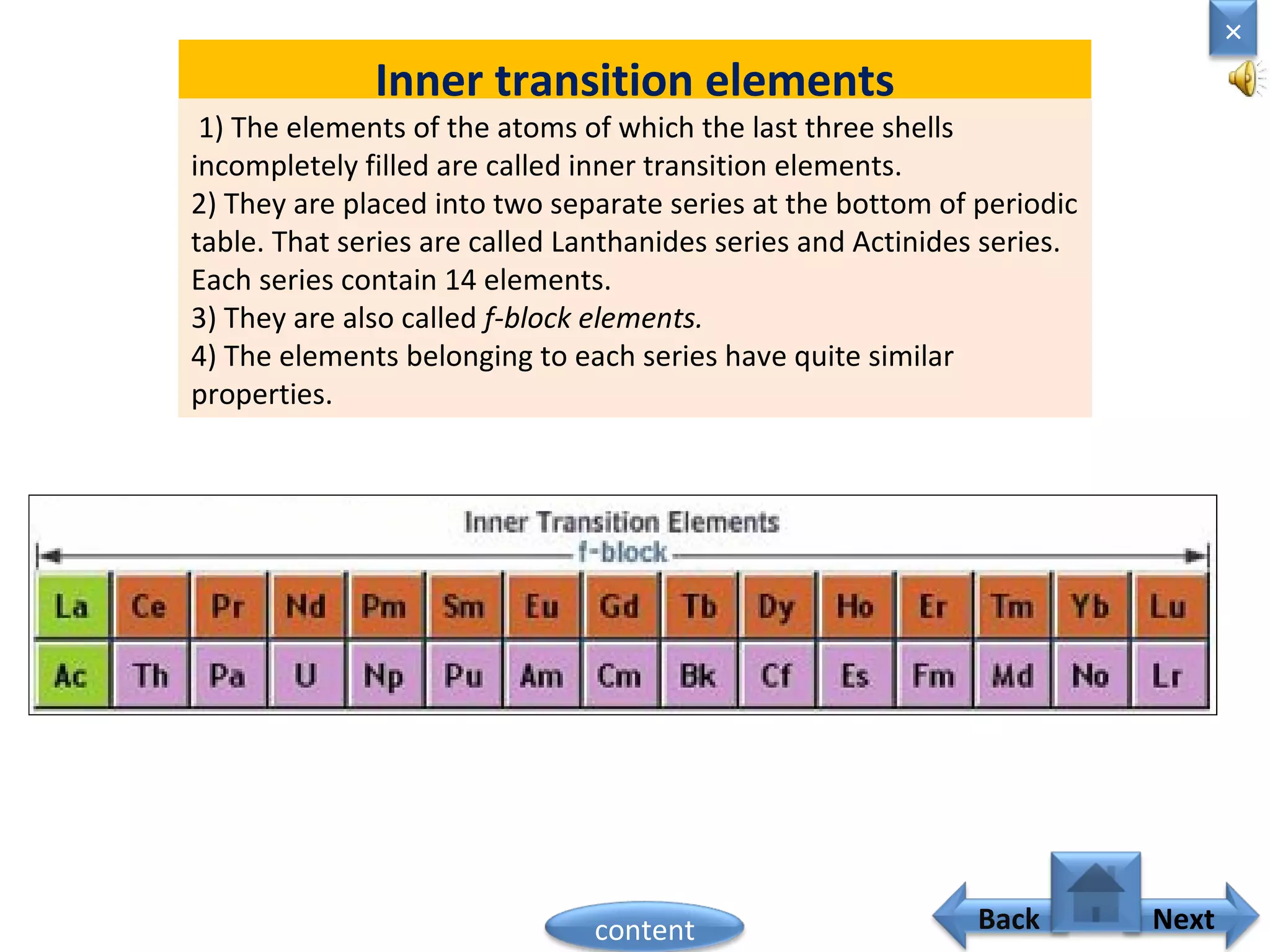
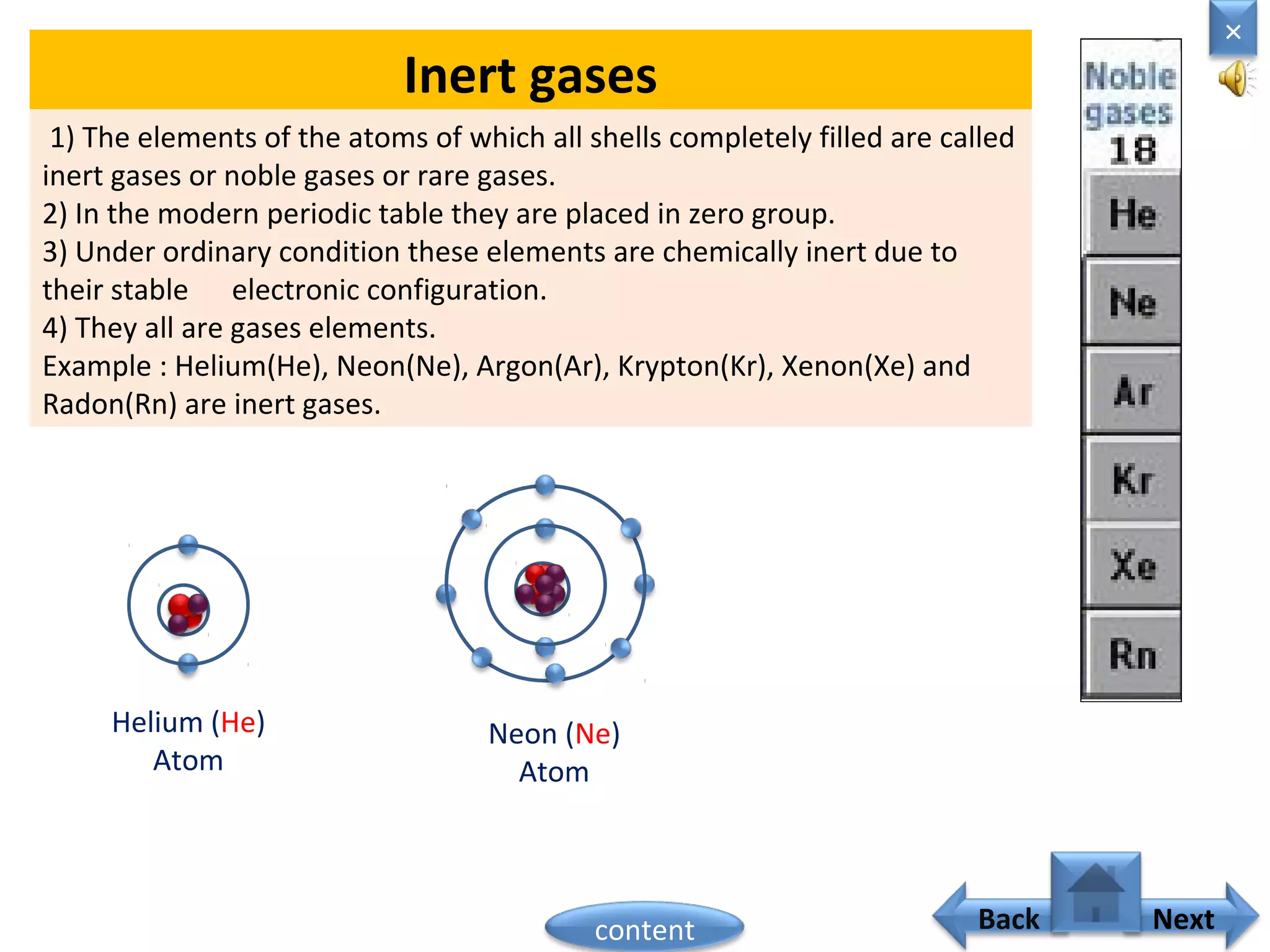
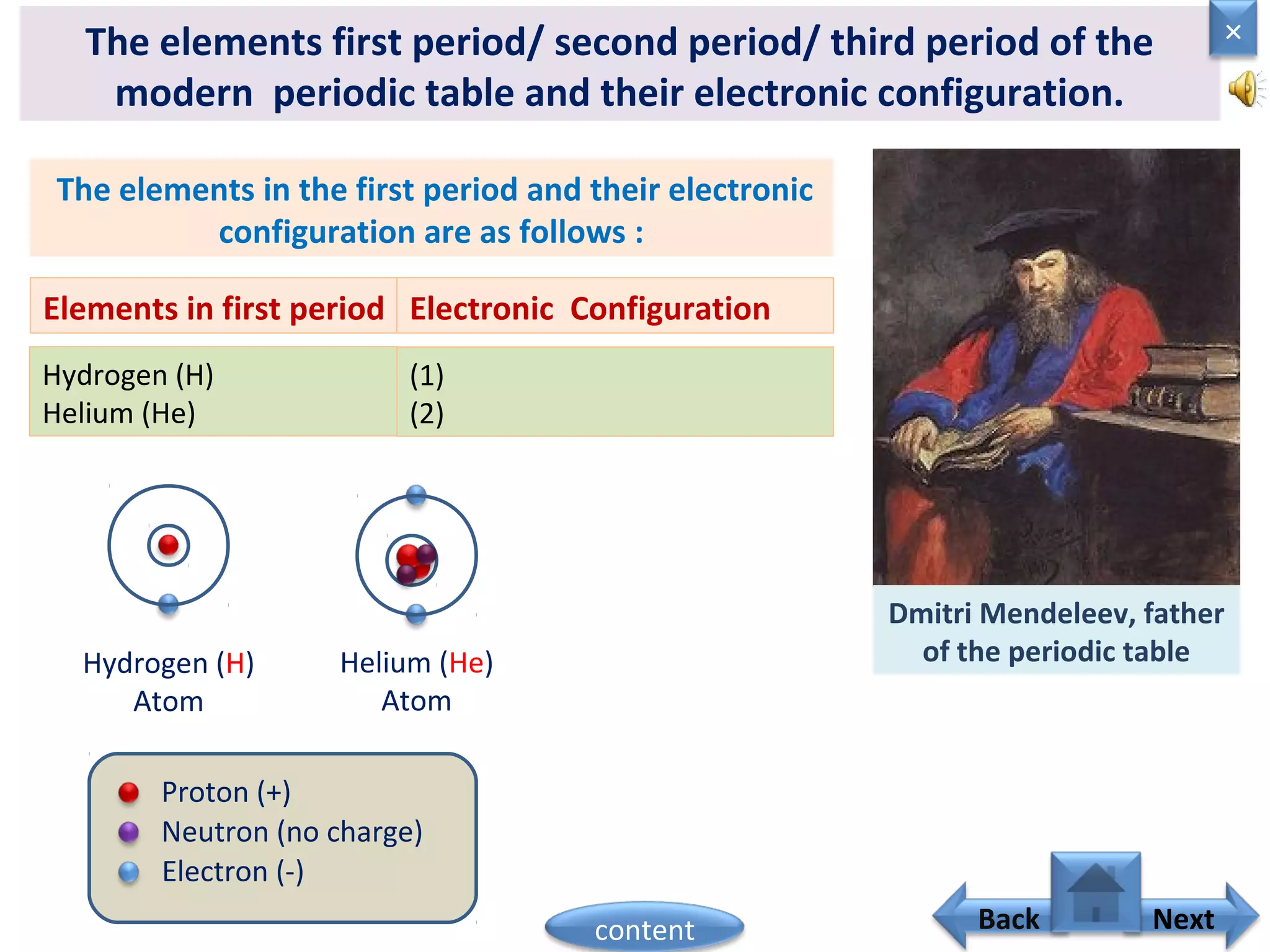
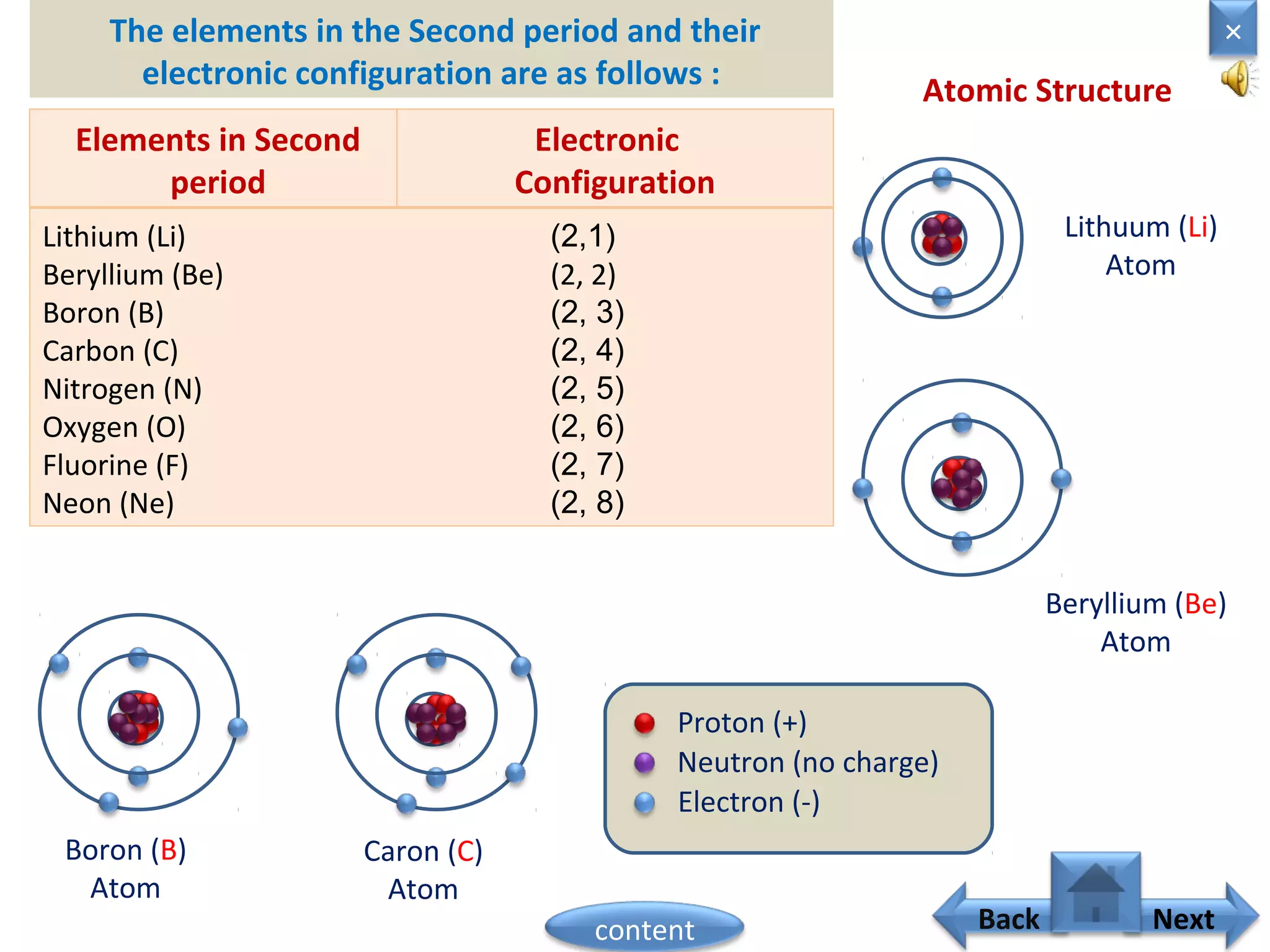
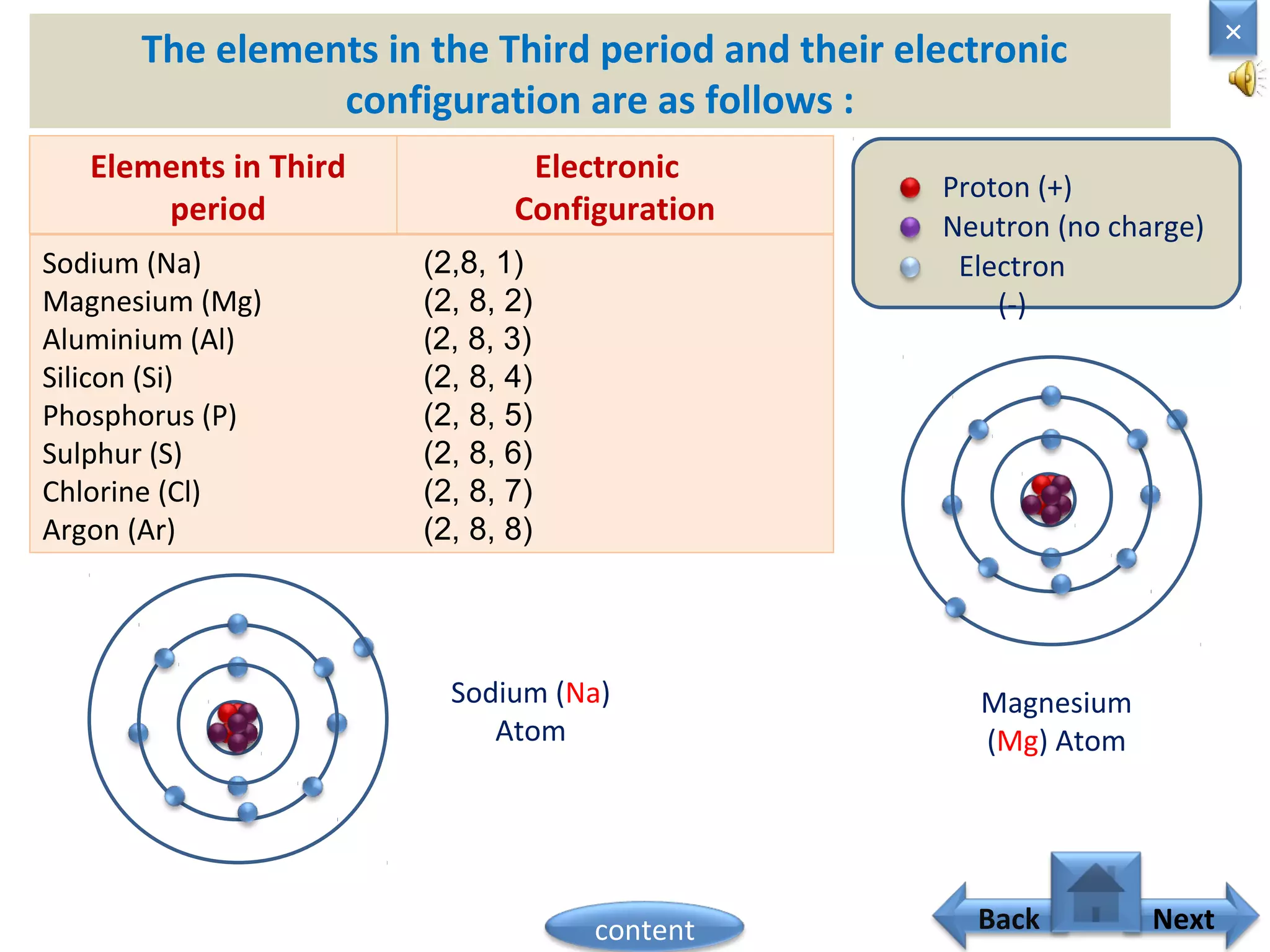
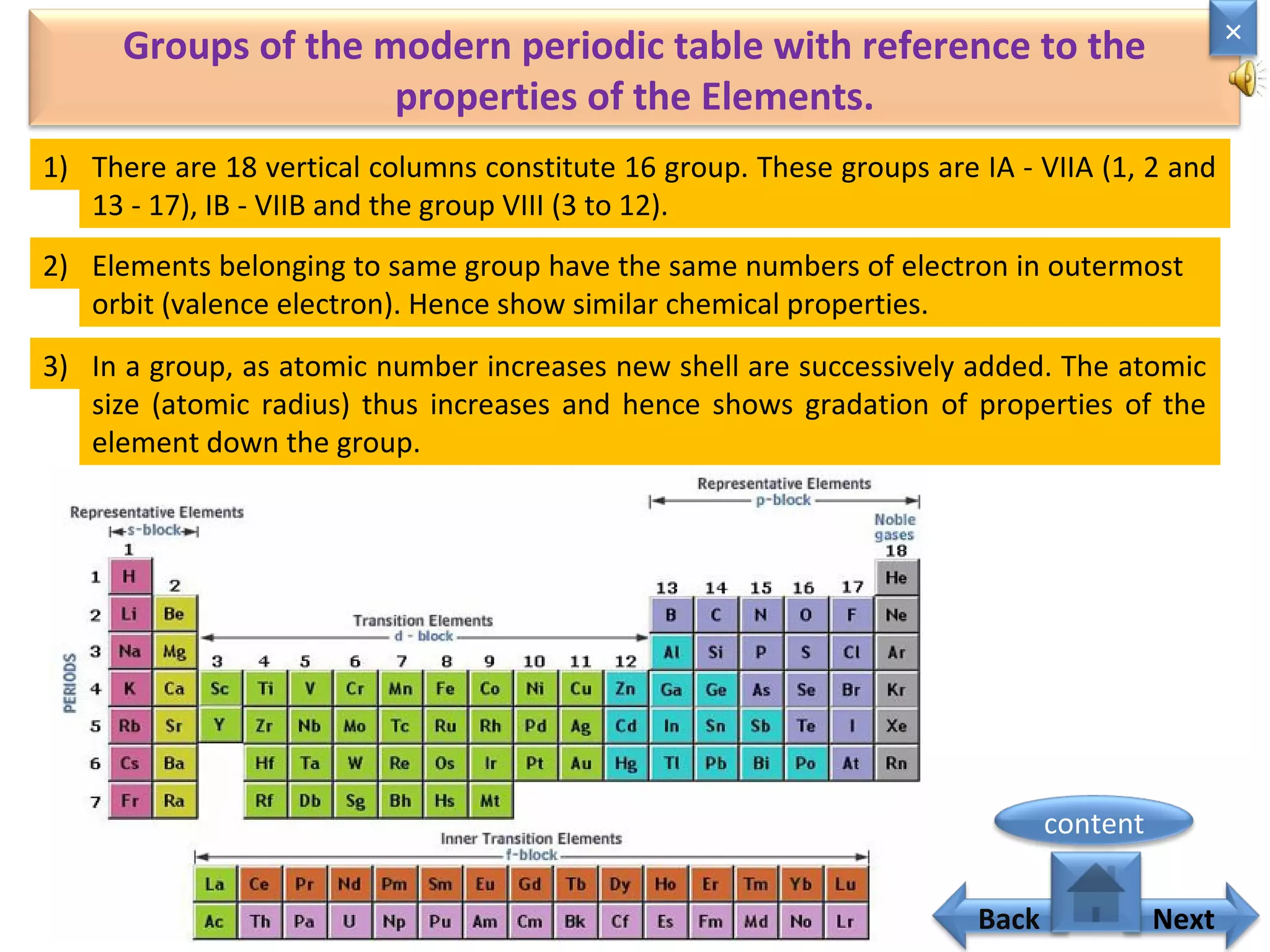
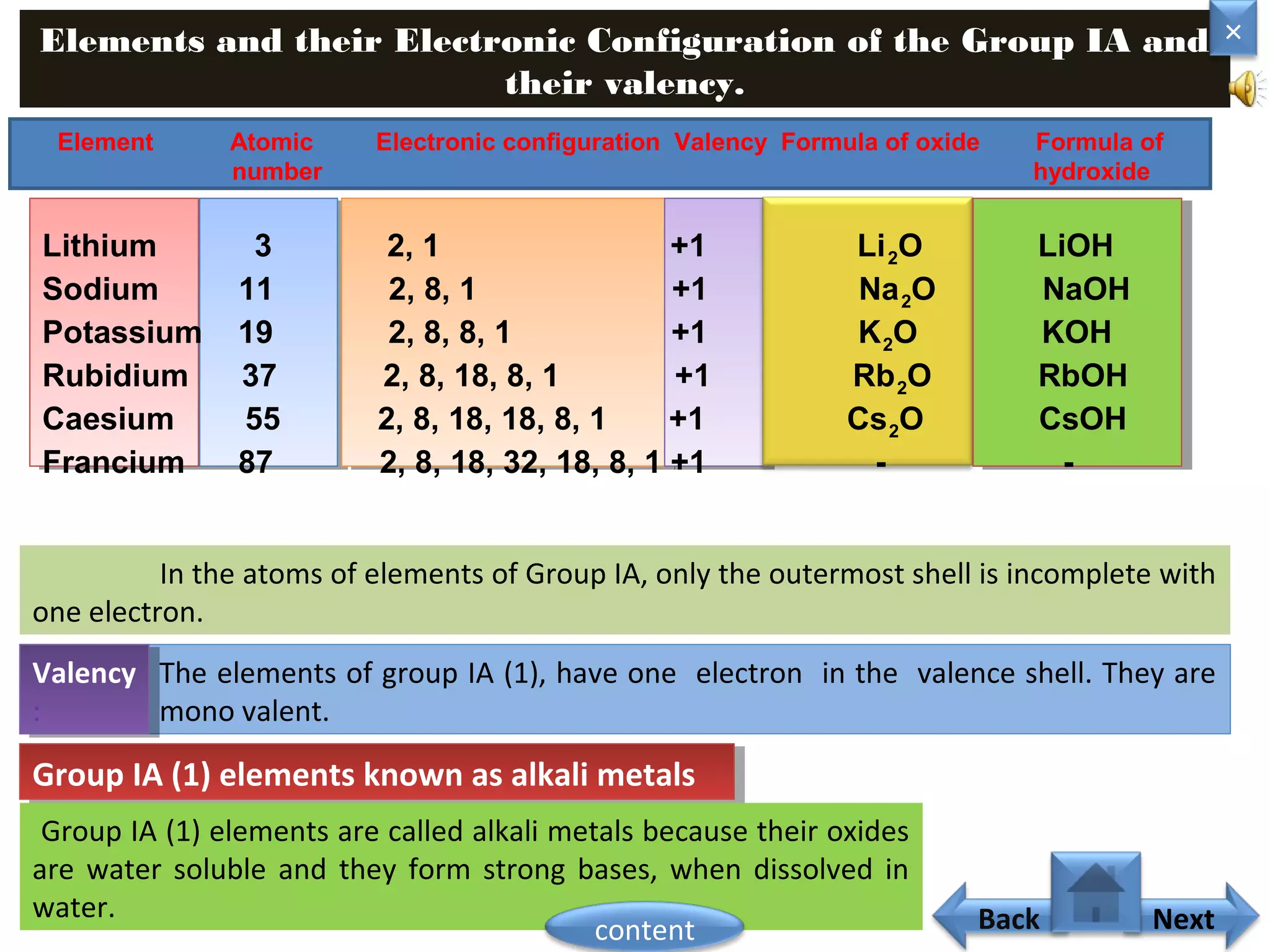
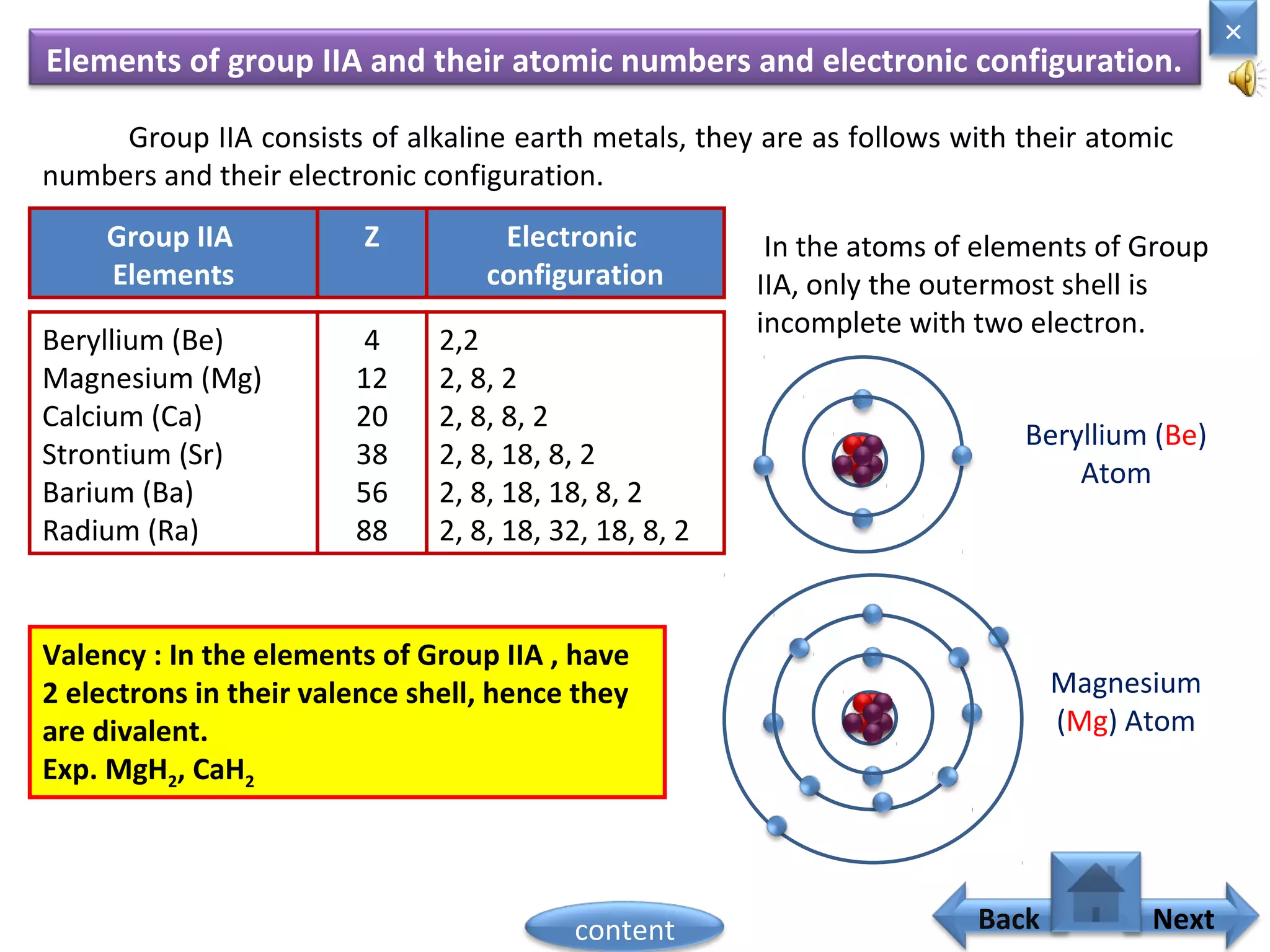
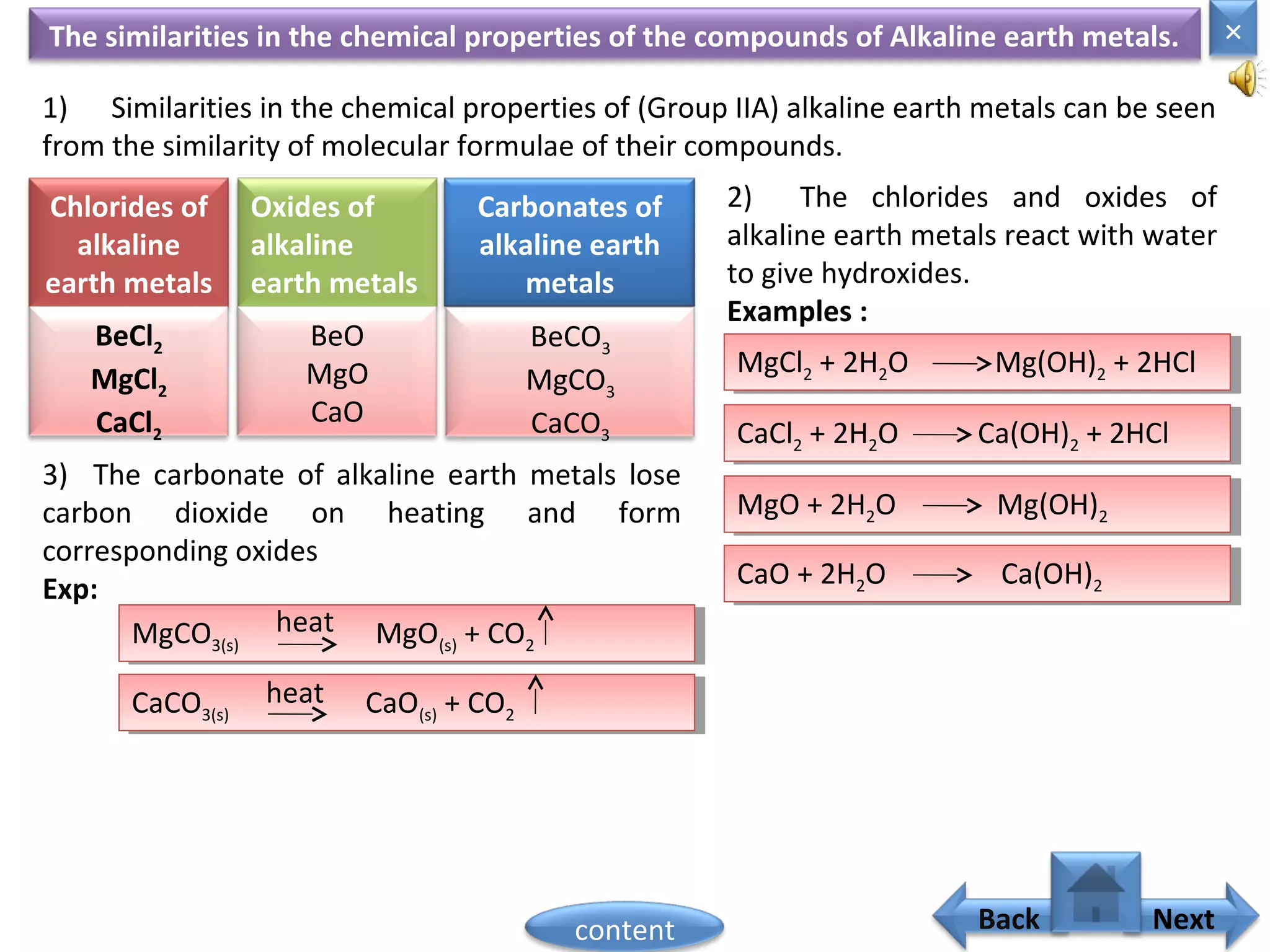
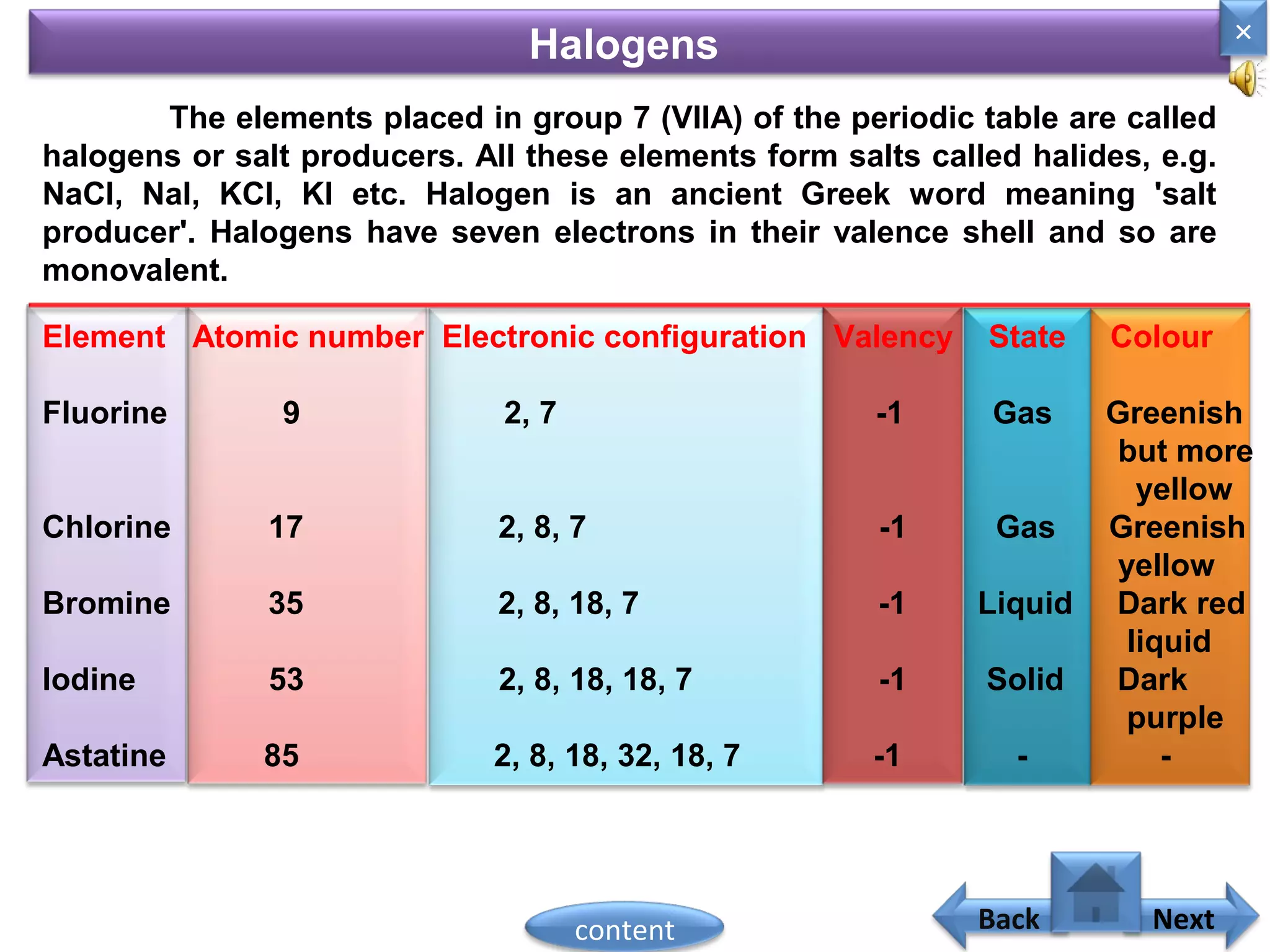
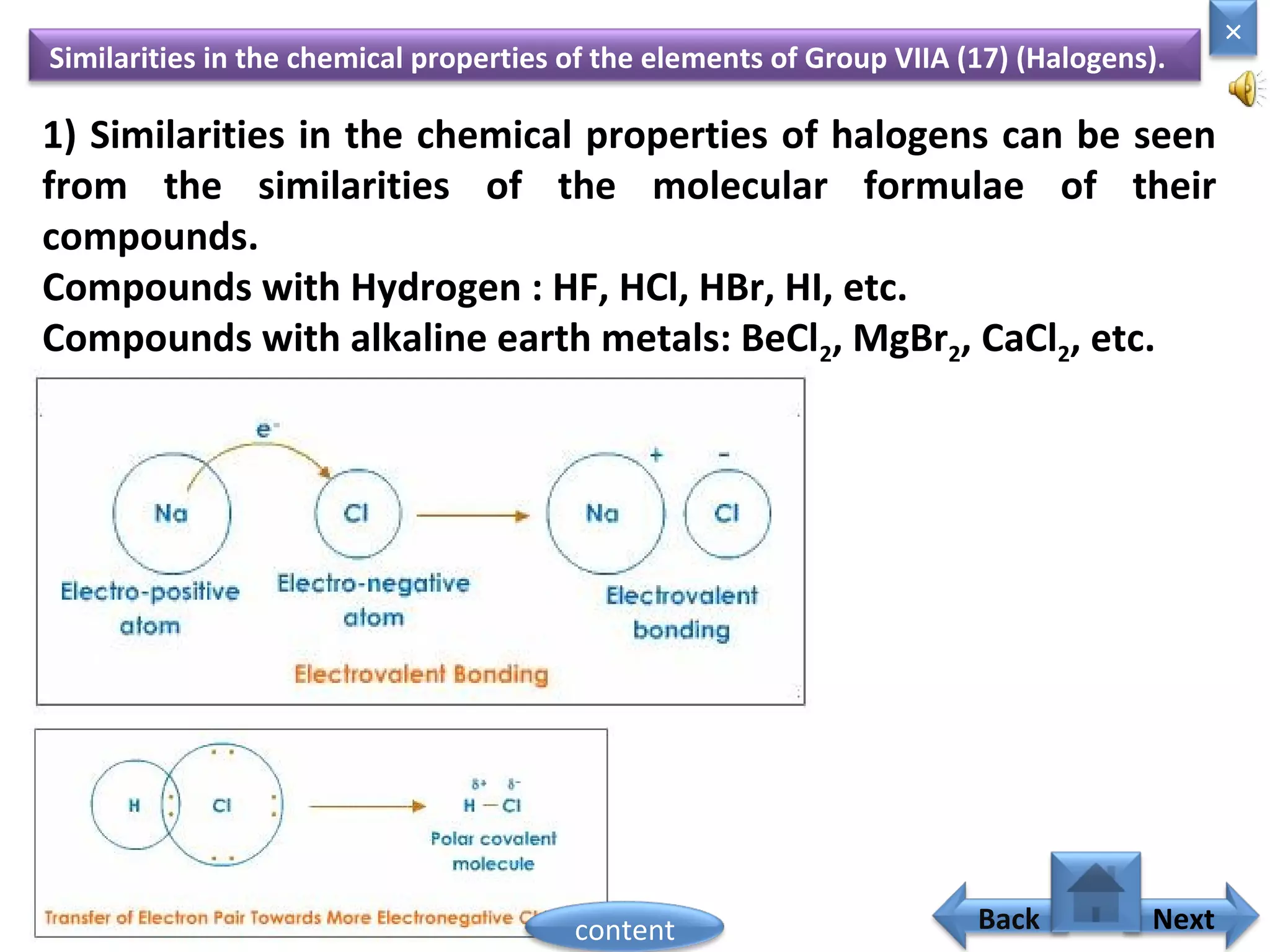
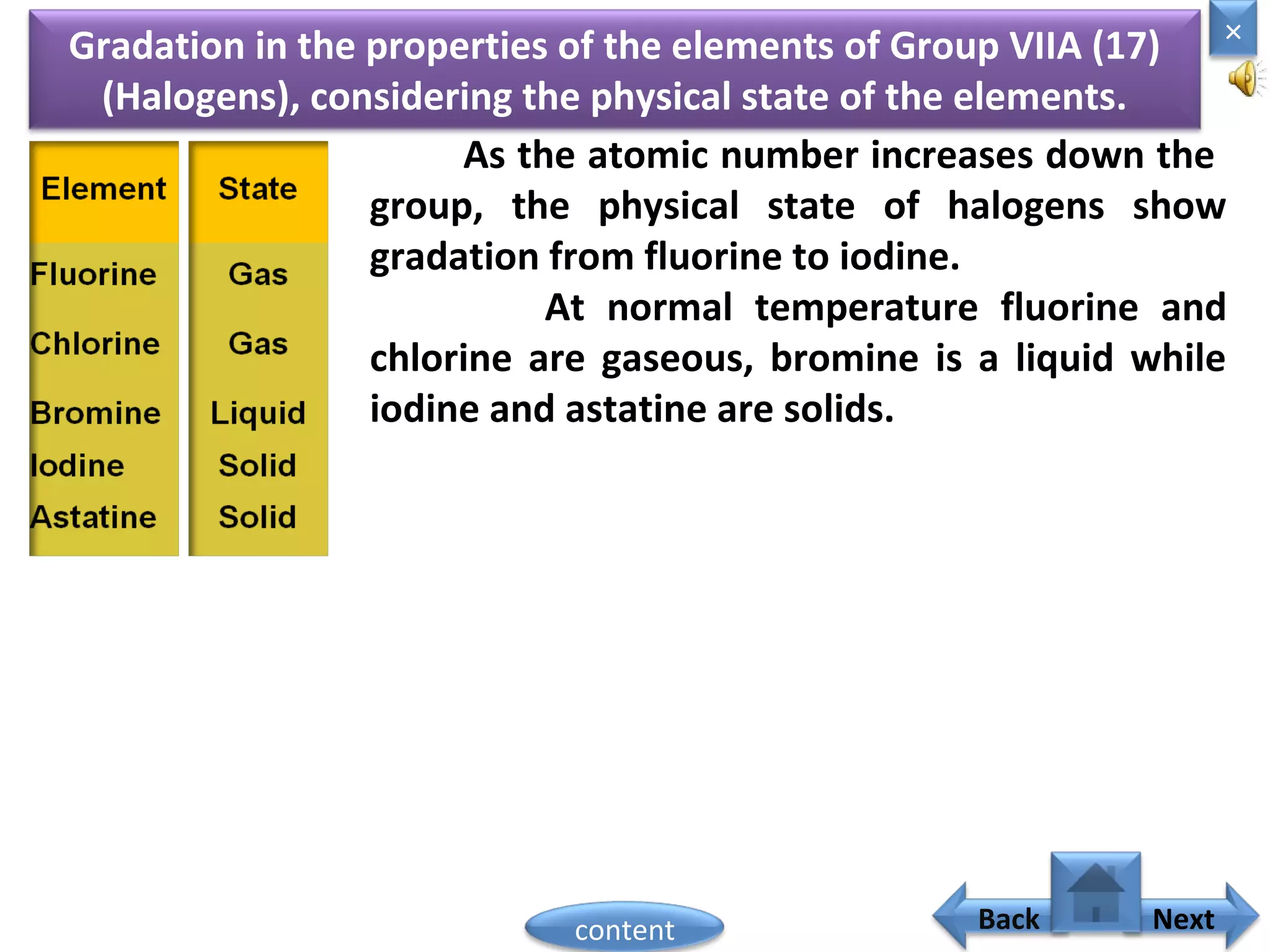
1) Moseley showed that the atomic number of an element is equal to the number of protons in its nucleus. He suggested atomic number is a more fundamental property than atomic weight for classifying elements.
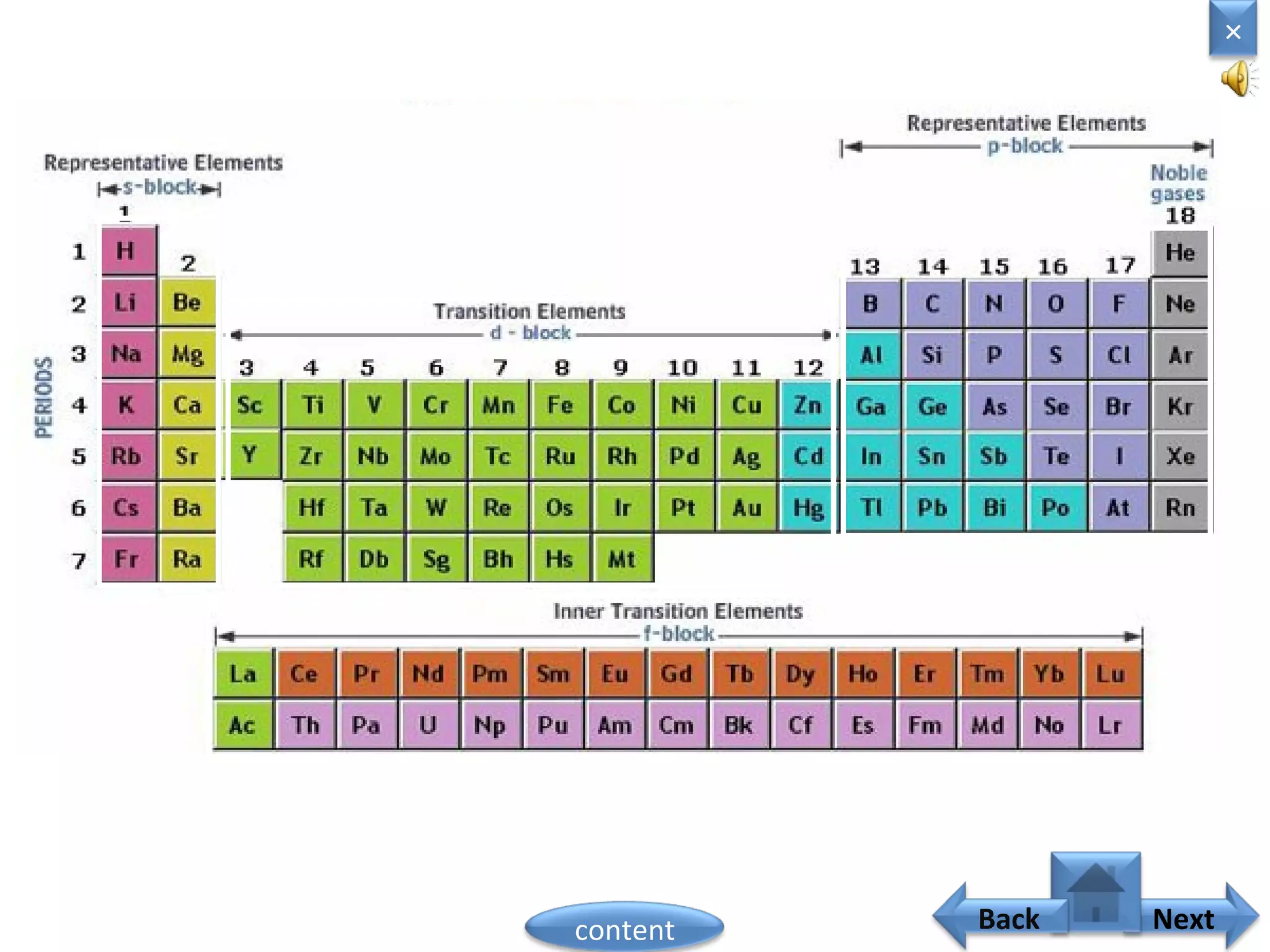
2) In the modern periodic table, elements are arranged in order of increasing atomic number in a more systematic way based on Moseley's work.
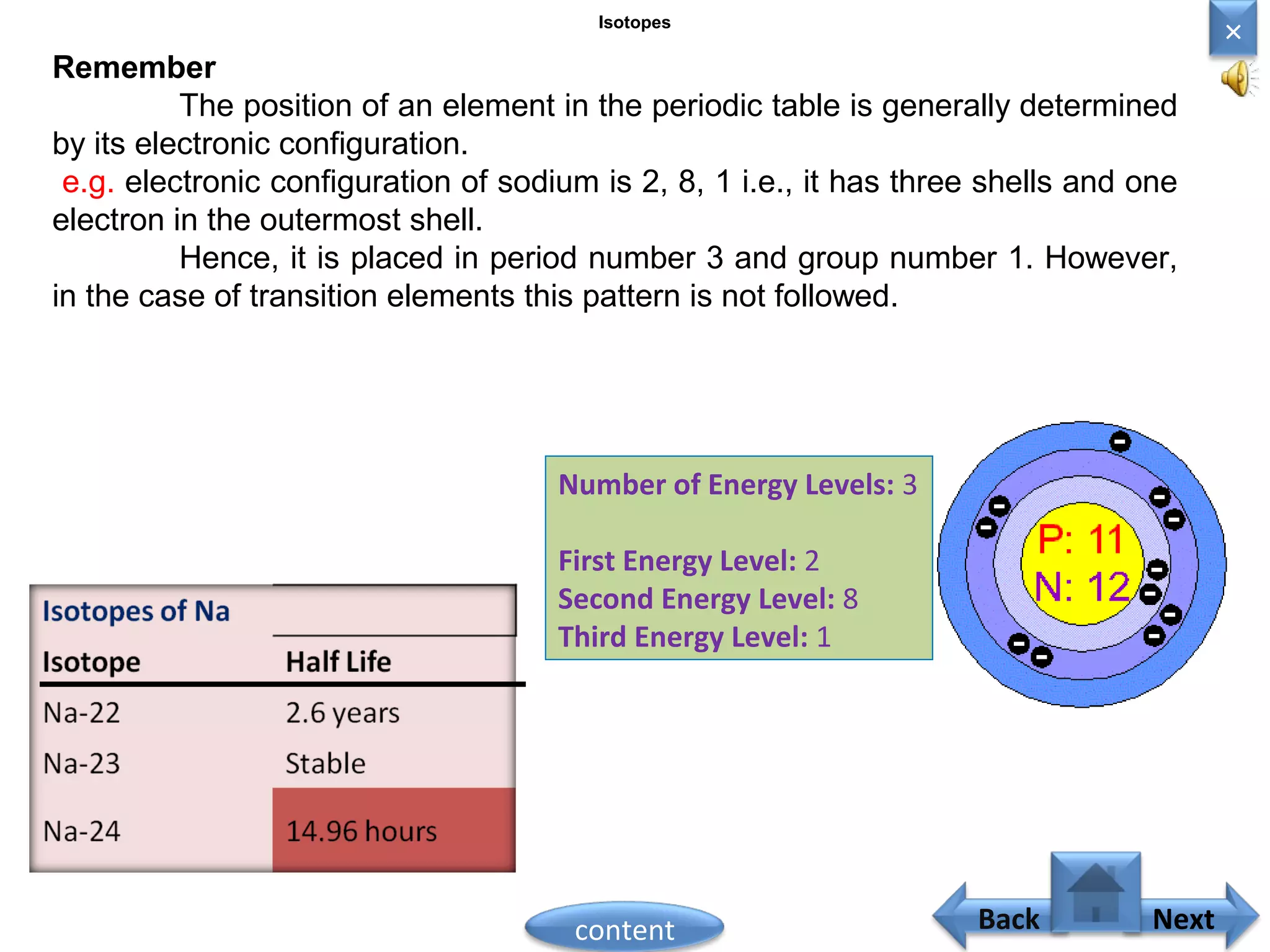
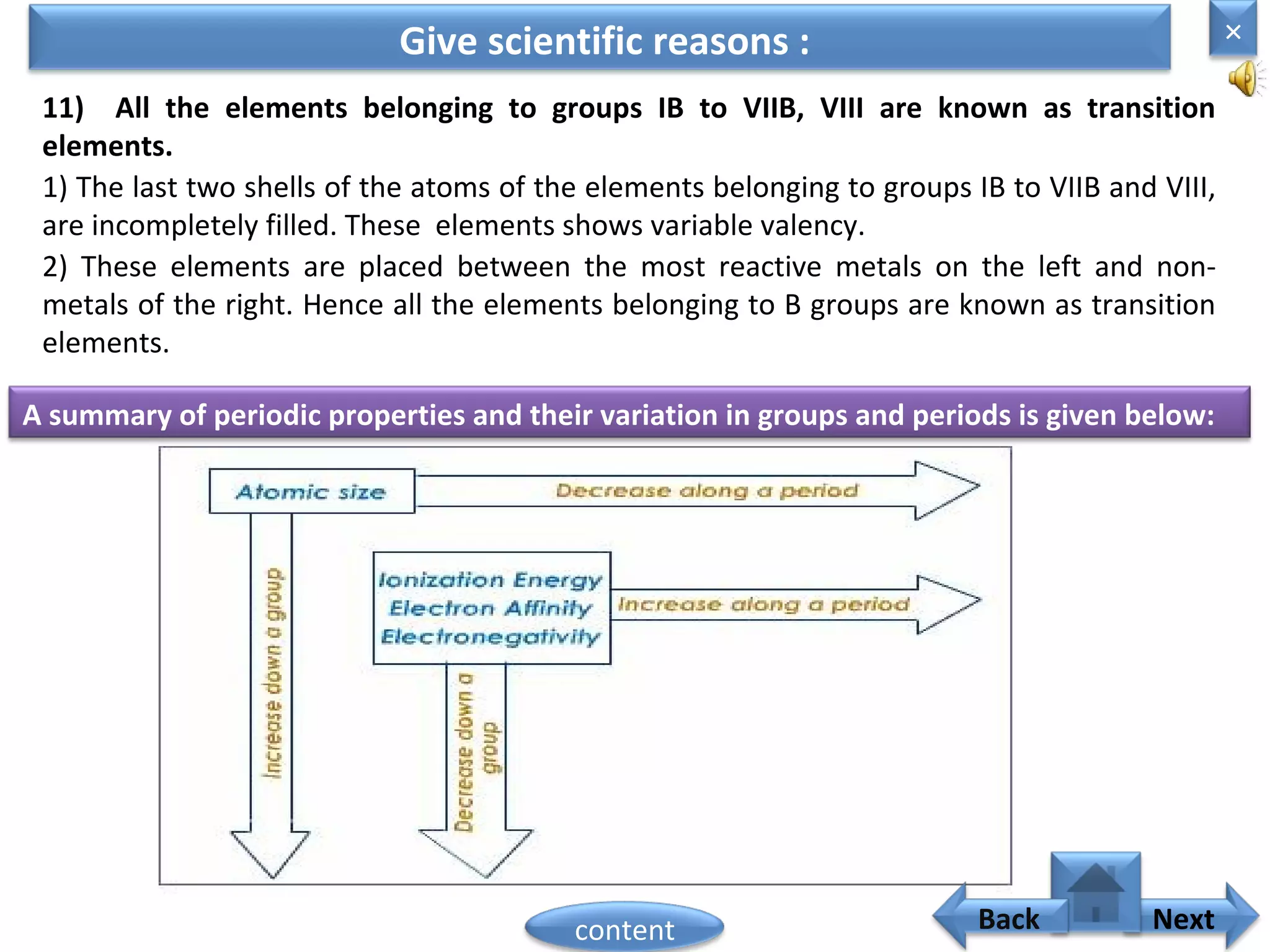
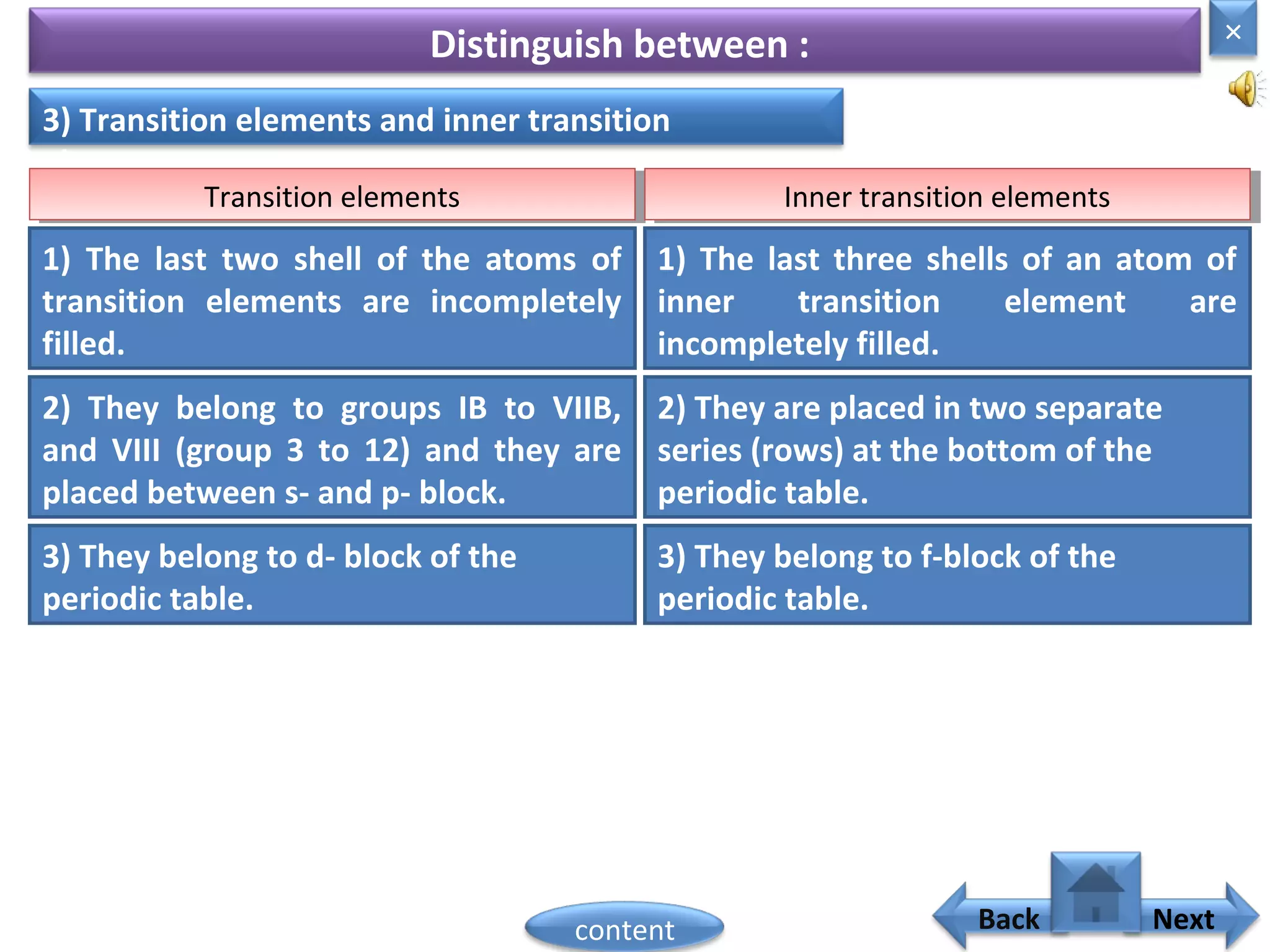
3) The electronic configuration of an element is directly related to its atomic number. The modern periodic law states that the properties of elements are periodic functions of their atomic numbers.