control of microbes.pptx
- 1. CONTROL OF MICROBIAL GROWTH
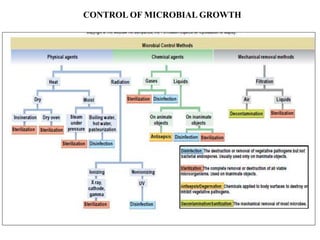
- 3. Moist heat sterilization: 1- At normal pressure (at or below 100°C) a- Pasteurization: It is the use of mild heat to reduce the number of microorganisms in a product or food. In the case of pasteurization of milk, the time and temperature depend on killing potential pathogens that are transmitted in milk. Pasteurization kills many spoilage organisms, as well, and therefore increasesthe shelf life of milk especially at refrigeration temperatures (2°C). It is not a method of sterilization, rather a method for preventing spoilage of food. Milk is usually pasteurized by heating, typically at 62°C for 30 minutes, then cooling to 7°C (holding process) or at 72°C for 15 seconds then rapid cooling to 4°C (flash process), to kill bacteria and extend the milk's usable life.
- 4. b- Boiling in water: 100o for 30 minutes. Kills everything except some endospores. It also inactivates viruses. For the purposes of purifying drinking water, 100o for five minutes is a "standard". c- Steaming: The process is carried out in a double walled box with the inner one perforated to allow the flow of steam (Koch sterilizer) d- Tyndalization: Three 30-minute intervals of boiling, followed by periods of cooling. Kills bacterial endospores. First heat treatment kills vegetative cells and during incubation spores germinate which are in turn killed by the second and third heat treatment.
- 5. 2- At high pressure (above 100°C) To obtain temperature over 100°C, water must boil under pressure, where temperature is directly proportional to pressure. Sterilization by moist heat usually involves the use of steam at a temperature between 115-134°C. a- Pressure cooker: Used in emergency for sterilization of surgical tools in minor operations. Heat treatment is usually at 115-121°C for 20- 25 min. b- Autoclave: Autoclaving is the most effective and most efficient means of sterilization. All autoclaves operate on a time/temperature relationship. These two variables are extremely important. Higher temperatures ensure more rapid killing. The usual standard temperature/pressure employed is 121ºC for 15 minutes.
- 6. Dry heat sterilization 1. Incineration: Burning of inoculating needles, blades, forceps etc. 2. Hot air oven: 1700 C for around 2 hr.
- 12. Chemical agents are generally not intended to achieve sterilization. Most reduce the microbial populations to safe levels or remove pathogens from objects. An ideal disinfectant or antiseptic (chemical agent) kills microorganisms in the shortest possible time without damaging the material treated. CHEMICAL METHODS OF CONTROL
- 13. Phenol. One of the first chemicals to be used for disinfection was phenol. First used by Joseph Lister in the 1860s, it is the standard for most other antiseptics and disinfectants. Phenol derivatives called phenolics contain altered molecules of phenol useful as antiseptics and disinfectants. The phenolics damage cell membranes and inactivate enzymes of microorganisms, while denaturing their proteins. They includecresols, such as Lysol, as well as several bisphenols, such as hexachlorophene, which is particularly effective against staphylococci
- 14. Halogens. Among the halogen antiseptics and disinfectants are chlorine and iodine.Iodine is used as a tincture of iodine, an alcohol solution. Combinations of iodine and organic molecules are called iodophors. They include Betadine and Isodyne, both of which contain a surface active agent called povidone. Iodine combines with microbial proteins and inhibits their function. Chlorine also combines with microbial proteins. It is used as sodium hypochlorite (bleach). As calcium hypochlorite, chlorine is available to disinfect equipment in dairies, slaughterhouses, and restaurants. The chloramines contain chlorine together with ammonia. They are used to sanitize glassware and eating utensils and are effective in the presence of organic matter. Chlorine is also used as a gas to maintain a low microbial count in drinking water.
- 15. Alcohols. Alcohols are useful chemical agents when employed against bacteria and fungi, but they have no effect on bacterial spores. The type of alcohol most widely used is 70 percent ethyl alcohol (ethanol). Isopropyl alcohol (rubbing alcohol) is also useful as an antiseptic and disinfectant. Because alcohols evaporate quickly, they leave no residue and are useful in degerming the skin before injections Heavy metals. A number of heavy metals have antimicrobial ability. For example,silver is used as silver nitrate in the eyes of newborns to guard against infection byNeisseria gonorrheae. It is also used to cauterize wounds. Copper is used as copper sulfate to retard the growth of algae in swimming pools, fish tanks, and reservoirs. Zincis useful as zinc chloride in mouthwashes and as zinc oxide as an antifungal agent in paints. The heavy metals are believed to act by combining with sulfhydryl groups on cellular proteins.
- 16. Soaps and detergents. Soaps and detergents decrease the surface tension between microorganisms and surfaces, and thereby help cleanse the surface. Soaps emulsify the oily film on the body surface, carrying the oils, debris, and microorganisms away in a degerming action. The cationic detergents are quaternary ammonium compounds.They solubilize the cell membranes of microorganisms. Among the popular compounds are Zephiran (benzalkonium chloride) and Cepacol (cetylpyridinium chloride) Aldehydes. Two aldehydes, formaldehyde and glutaraldehyde, inactivate microbial proteins by crosslinking the functional groups in the proteins. Formaldehyde gas is commonly used as formalin, a 37 percent solution of formaldehyde gas. Glutaraldehyde is used as a liquid to sterilize hospital equipment. However, several hours are required to destroy bacterial spores
- 17. Ethylene oxide. Sterilization can be achieved with a chemical known as ethylene oxide (ETO). This chemical denatures proteins and destroys all microorganisms, including bacterial spores. It is used at warm temperatures in an ethylene oxide chamber. Several hours are needed for exposure and flushing out the gas, which can be toxic to humans. ETO is widely used for plastic instruments such as Petri dishes, syringes, and artificial heart valves Food preservatives. Foods can be preserved by using a number of organic acids to maintain a low microbial population. Sorbic acid is used in a number of acidic foods, including cheese, to prevent microbial growth. Benzoic acid also inhibits fungi and is used in acidic foods and soft drinks. Calcium propionic acid prevents the growth of mold in breads and bakery products.
- 18. Understanding the mechanisms of resistance has become a significant biochemical issue over the past several years and nowadays there is a large pool of information about how bacteria can develop drug resistance. Biochemical and genetic aspects of antibiotic resistance mechanisms in bacteria are shown in Fig. 1. DRUG RESISTANCE
- 20. 1. Antibiotic inactivation The defence mechanisms within the category of antibiotic inactivation include the production of enzymes that degrade or modify the drug itself. Biochemical strategies are hydrolysis, group transfer, and redox mechanisms. Antibiotic inactivation by hydrolysis Many antibiotics have hydrolytically susceptible chemical bonds (e.g. esters and amides). Several enzymes are known to destroy antibiotic activity by targeting and cleaving these bonds. These enzymes can often be excreted by the bacteria, inactivating antibiotics before they reach their target within the bacteria. The classical hydrolytic amidases are the b-lactamases that cleave the b-lactam ring of the penicillin and cephalosporin antibiotics. Many Gram- negative and Gram-positive bacteriaproduce such enzymes, and more than 200 different b-lactamases have been identified.
- 21. Antibiotic inactivation by group transfer The most diverse family of resistant enzymes is the group of transferases. These enzymes inactivate antibiotics (aminoglycosides, chloramphenicol, streptogramin, macrolides or rifampicin) by chemical substitution (adenylyl, phosphoryl or acetyl groups are added to the periphery of the antibiotic molecule). The modified antibiotics are affected in their binding to a target. Antibiotic inactivation by redox process The oxidation or reduction of antibiotics has been infrequently exploited by pathogenic bacteria. However, there are a few of examples of this. One is the oxidation of tetracycline antibiotics by the TetX enzyme. Streptomyces virginiae, producer of the type A streptogramin antibiotic virginiamycin M1, protects itself from its own antibiotic by reducing a critical ketone group to an alcohol at position 16.
- 22. 2. Target modification The second major resistance mechanism is the modification of the antibiotic target site so that the antibiotic is unable to bind properly. Peptidoglycan structure alteration The peptidoglycan component of the bacterial cell wall provides an excellent selective target for the antibiotics. It is essential for the growth and survival of most bacteria. Consequently, enzymes involved in synthesis and assembly of the peptidoglycan component of the bacterial cell wall provide excellent targets for selective inhibition. The presence of mutations in the penicillin-binding domain of penicillin-binding proteins (PBPs) results in decreased affinity to b-lactam antibiotics. Alterations among PBPs result in ampicillin resistance among Enterococcus faecium, and penicillin resistance among Streptococcus pneumoniae
- 23. Vancomycin inhibit cell wall synthesis of Gram-positive bacteria by binding C-terminal acyl-D-alanyl-D-alanine (acyl-D-Ala-D-Ala)-containing residues in peptidoglycan precursors. Resistance is achieved by altering the target site by changing the D- -Ala-D-Ala to D-alanyl-D-lactate (D-Ala-D-Lac) or D-alanyl- -D-serine (D-Ala-D-Ser) at the C-terminus, which inhibits the binding of vancomycin.
- 24. Protein synthesis interference A wide range of antibiotics interfere with protein synthesis on different levels of protein metabolism. The resistance to antibiotics that interfere with protein synthesis (aminoglycosides, tetracyclines, macrolides, chloramphenicol, fusidic acid, mupirocin, streptogramins, oxazolidinones) or transcription via RNA polymerase(the rifamycins) is achieved by modification of the specific target. Mutations in the 16S rRNA gene confer resistance to the aminoglycosides. Chromosomally acquired streptomycin resistance in M. tuberculosis is frequently due to mutations in the rpsL gene encoding the ribosomal protein S12. Microorganisms that produce aminoglycosides have developed mechanism of high level antibiotic resistance by posttranscriptional methylation of 16S rRNA in the aminoglycoside binding site. This mechanism of resistance has recently been reported in human pathogens from nosocomial infections and animal isolates.
- 25. DNA synthesis interference Fluoroquinolones interact with the DNA gyrase and topoisomerase IV enzymes and prevent DNA replication and transcription. Resistance is conferred by mutations in specific regions of the structural genes that sufficiently alter these enzymes preventing the binding of antibiotics . The most common mutations in this region cause resistance through decreased drug affinity for the altered gyrase–DNA complex
- 26. 3. Efflux pumps Efflux pumps affect all classes of antibiotics, especially the macrolides, tetracyclines, and fluoroquinolones because these antibiotics inhibit different aspects of protein and DNA biosynthesis and therefore must be intracellular to exert their effect. Efflux pumps vary in both their specificity and mechanism . Although some are drug-specific, many efflux systems are multidrug transporters that are capable of expelling a wide spectrum of structurally unrelated drugs thus contributing significantly to bacterial multidrug resistance (MDR)
- 27. 4. Outer membrane (OM) permeability changes Gram-negative bacteria possess an outer membrane consisting of an inner layer containing phospholipids and an outer layer containing the lipid A moiety of lipopolysaccharides (LPS). This composition of the outer membrane (OM) slows down drug penetration, and transport across the OM is achieved by porin proteins that form water-filled channels. Drug molecules can penetrate the OM employing one of the following modes: by diffusion through porins, by diffusion through the bilayer or by self-promoted uptake. Antibiotics such as b- - lactams, chloramphenicol and fluoroquinolones enter the Gram-negative outer membrane via porins. As such, changes in porin copy number, size or selectivity will alter the rate of diffusion of these antibiotics