Pankaj omvs biochem symposium
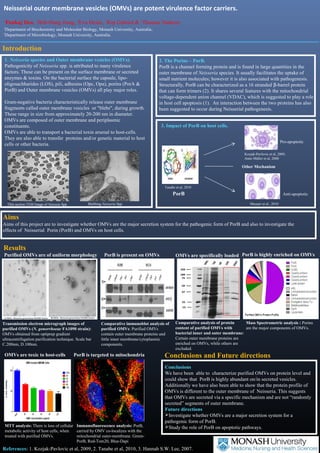
- 1. Neisserial outer membrane vesicles (OMVs) are potent virulence factor carriers. 1Pankaj Deo, 2Jhih-Hang Jiang, 2Eva Heinz, 1Kip Gabriel & 1Thomas Naderer 1Department of Biochemistry and Molecular Biology, Monash University, Australia. 2Department of Microbiology, Monash University, Australia. Introduction 1. Neisseria species and Outer membrane vesicles (OMVs). Pathogenicity of Neisseria spp. is attributed to many virulence factors. These can be present on the surface membrane or secreted enzymes & toxins. On the bacterial surface the capsule, lipo-oligosachharides (LOS), pili, adhesins (Opc, Opa), porins (PorA & PorB) and Outer membrane vesicles (OMVs) all play major roles. Gram-negative bacteria characteristically release outer membrane fragments called outer membrane vesicles or "blebs", during growth. These range in size from approximately 20-200 nm in diameter. OMVs are composed of outer membrane and periplasmic constituents. OMVs are able to transport a bacterial toxin arsenal to host-cells. They are also able to transfer proteins and/or genetic material to host cells or other bacteria. 2. The Porins – PorB. PorB is a channel forming protein and is found in large quantities in the outer membrane of Neisseria species. It usually facilitates the uptake of small nutrient molecules; however it is also associated with pathogenesis. Structurally, PorB can be characterized as a 16 stranded b-barrel protein that can form trimers (2). It shares several features with the mitochondrial voltage-dependent anion channel (VDAC), which is suggested to play a role in host cell apoptosis (1). An interaction between the two proteins has also been suggested to occur during Neisserial pathogenesis. 3. Impact of PorB on host cells. Tanabe et al, 2010 PorB Other Mechanism Thin section TEM Image of Neisseia Spp Blebbing Neisseria Spp Aims Aims of this project are to investigate whether OMVs are the major secretion system for the pathogenic form of PorB and also to investigate the effects of Neisserial Porin (PorB) and OMVs on host cells. Conclusions and Future directions Pro-apoptotic Conclusions We have been able to characterize purified OMVs on protein level and could show that PorB is highly abundant on/in secreted vesicles. Additionally we have also been able to show that the protein profile of OMVs is different to the outer membrane of Neisseria. This suggests that OMVs are secreted via a specific mechanism and are not “randomly secreted” segments of outer membrane. Future directions Investigate whether OMVs are a major secretion system for a pathogenic form of PorB. Study the role of PorB on apoptotic pathways. Results Purified OMVs are of uniform morphology PorB is present on OMVs PorB is targeted to mitochondria References: 1. Kozjak-Pavlovic et al, 2009, 2. Tanabe et al, 2010, 3. Hannah S.W. Lee, 2007. Kozjak-Pavlovic et al, 2009, Anne Müller et al, 2000 Anti-apoptotic Massari et al., 2010 OMVs are specifically loaded Comparative immunoblot analysis of purified OMVs: Purified OMVs contain outer membrane proteins and little inner membrane/cytoplasmic components. Transmission electron micrograph images of purified OMVs (N. gonorrhoeae FA1090 strain): OMVs obtained from optiprep gradient ultracentrifugation purification technique. Scale bar C.200nm, D.100nm. Comparative analysis of protein content of purified OMVs with bacterial inner and outer membrane: Certain outer membrane proteins are enriched on OMVs, while others are excluded. OMVs are toxic to host-cells Immunofluorescence analysis: PorB, carried by OMV co-localizes with the mitochondrial outer-membrane. Green- PorB, Red-Tom20, Blue-Dapi MTT analysis: There is loss of cellular metabolic activity of host cells, when treated with purified OMVs. PorB is highly enriched on OMVs Mass Spectrometric analysis : Porins are the major components of OMVs.
