Polar Molecules Interact Ions Salt Dissociation Equations
•Download as PPT, PDF•
0 likes•196 views
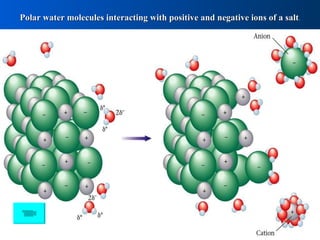
When salts dissolve in water, the polar water molecules interact with the positive and negative ions of the salt, attracting them and breaking apart the crystal structure. Electrolytes separate into ions that move freely in solution while maintaining net neutral charge. Dissociation equations represent electrolytes dissolving as solute separating into aqueous ions. These equations must be balanced and show correct ionic charges and physical states. Dissociation refers to ionic compounds breaking apart while ionization describes molecules breaking into separate particles.
Report
Share
Report
Share
More Related Content
What's hot
What's hot (20)
Chem 2 - Acid-Base Equilibria IX: Salts that Hydrolyze in Water to Affect pH

Chem 2 - Acid-Base Equilibria IX: Salts that Hydrolyze in Water to Affect pH
F.Sc. Part 1 Paper Faisalabad Board 2009 (Malik Xufyan)

F.Sc. Part 1 Paper Faisalabad Board 2009 (Malik Xufyan)
Viewers also liked
Viewers also liked (8)
Similar to Polar Molecules Interact Ions Salt Dissociation Equations
Similar to Polar Molecules Interact Ions Salt Dissociation Equations (20)
B.tech. ii engineering chemistry unit 5 A electrochemistry

B.tech. ii engineering chemistry unit 5 A electrochemistry
More from Kenton Hemsing
More from Kenton Hemsing (10)
Polar Molecules Interact Ions Salt Dissociation Equations
- 1. Polar water molecules interacting with positive and negative ions of a salt .
- 7. ► Practice, p.198 # 2